Quarterly insights: Process technology
The war on coronavirus and innovation in process technology and services
We believe the worldwide effort to develop treatments and vaccines for COVID-19 will help transform global vaccine capabilities and be a catalyst to drive new, more productive technology platforms forward to commercialization.
The effort to approve and produce vaccines and treatments will provide an additional tailwind on top of already healthy growth in the pharma service and technology sector.
Given the urgent nature and large volume of any resulting vaccines, we assume they will face extreme price pressure and limited margins. Therefore, on a risk adjusted basis we think the companies best positioned to grow and profit as a result of these efforts are the providers of services and technology – the picks and shovels of the efforts – where productivity and technology tend to be rewarded.
TABLE OF CONTENTS
- Who are the players?
- Wars and races drive innovation and opportunity
- But it’s not just vaccines
- Select COVID-19 partnerships in the sector
- Interesting times ahead
- Process technology outperforms major indices, rallies to annual high
- Process technology M&A loses steam
- Process technology private investment slows during 1H20
Introduction
In our first Process Technology Quarterly Insights report, it is nearly impossible to not review the healthcare oriented players in the space and how they contribute to the war on COVID-19. The current effort to address the pandemic serves as an example of the power and flexibility of the evolving pharma technology and services sector, from fundamental research to packaging of drug product to meet healthcare needs. In addition, we believe innovation and adoption of new ways of operating prompted by these desperate times bodes well for the sector participants both in the short and long runs.
Who are the players?
The participants in the sector span the drug development and commercialization gamut from contract science organizations (CSOs) such as Viva Biotech to contract research organizations (CROs) such as Syneos and IQVIA to contract manufacturing organizations (CMOs) such as Cambrex to fully integrated contract development and manufacturing organizations (CDMOs) exemplified by Catalent and Lonza.
The common theme is that they provide services to pharmaceutical and biotech firms. The other participants in the segment are platform technology providers, which sell equipment, consumables and technical expertise to both the contract service providers above as well as directly to pharma and biopharma.
Historically, this group included players such as Sigma Aldrich and Millipore (together now part of Merck KGaA), Pall and GE Life Science (both now part of Danaher), Invitrogen (now part of ThermoFisher) and current independents such as Codexis, Repligen and Dyadic.
TABLE 1: Representative players by primary segment

Source: First Analysis.
Wars and races drive innovation and opportunity
With the new coronavirus well on its way to infecting 7 million people and killing a reported 400,000, the pandemic is focusing the efforts and attention of the global community like few events in history. We believe these types of events transform industries and technology.
Compare military capabilities in 1914 prior to World War I to the conclusion of World War II in 1945. The world essentially moved from calvary charges to radar, aircraft carriers, jets and the nuclear age. Another less brutal example is the space race between the Soviet Union and the United States. It yielded innovations in computing, solar cells, air and water purification and robotics (not to mention the major impact of Tang powdered drink mix).
More recent events of this nature squarely related to healthcare and this sector include the race to sequence the human genome and the fight against HIV/AIDS, which have fundamentally changed our ability to understand and use genomics and advanced our understanding of the immune system and virology due to the focused global effort. As we move to battle COVID-19, the knowledge from the Human Genome Project and the ongoing HIV/AIDS fight has put us in much better shape to address the new coronavirus.
While we don’t claim COVID-19 rises to the level of world war, landing on the moon or the complexity of HIV, we do see the focus and investment relating to COVID-19 as opening up opportunities for new technologies and regulatory paths and growth of companies in what until recently was somewhat of a backwater in healthcare: vaccines.
But it’s not just vaccines
The World Health Organization, which published a list as of April 20 of vaccine candidates, shows five in clinical trials and 71 in preclinical stages; a summary from the New England Journal of Medicine is shown in Table 2. The Milken Institute lists 160 vaccine programs. What strikes us about this activity is the diversity of platforms – it’s no longer a world of developing seasonal flu vaccines in chicken eggs, the long-standing basis of vaccine production.
While vaccines rightly receive the most attention, there are numerous other treatments addressing COVID-19; the Milken Institute lists 228 non-vaccine therapies, including 64 antibodies, 23 antivirals and 15 cell therapies.
The diversity of programs is important for the sector as the global effort requires technology and capacity relevant to areas ranging from biologicals (vaccines and antibodies) to small molecules such as antivirals. This means there is a broad base of business across the sector, from research to packaging and from biologics to cell therapy to small molecules.
TABLE 2: Vaccine platforms, their attributes, and the status of vaccine candidates*
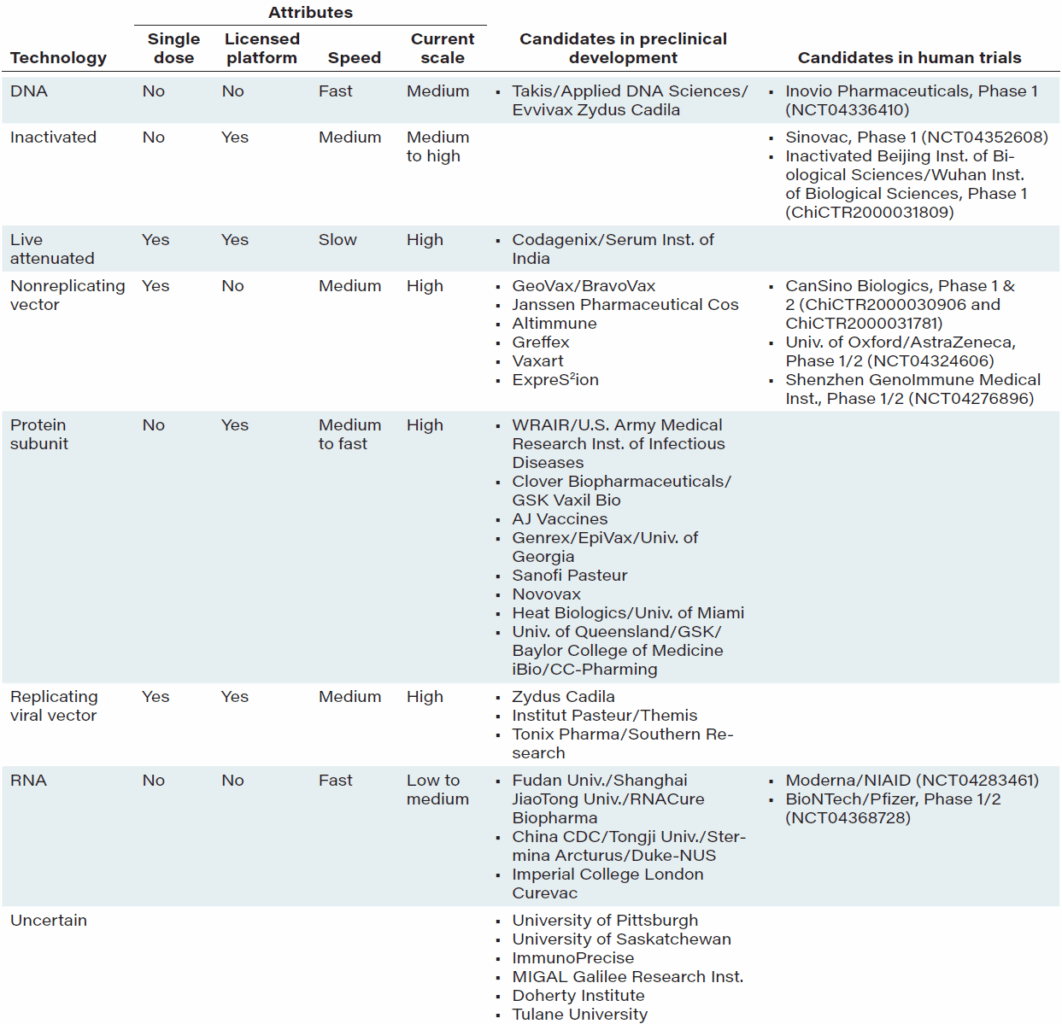
Source: New England Journal of Medicine, Developing Covid-19 Vaccines at Pandemic Speed, May 21, 2020.
Notes: Attributes refer to general attributes of the platform, and assessments are not intended as inferences about a particular candidate. NIAID denotes National Institute of Allergy and Infectious Diseases, and WRAIR denotes Walter Reed Army Institute of Research.
Select COVID-19 partnerships in the sector
Corden / Moderna: The Luxembourg-based CDMO Corden Pharma announced on May 28 an amendment to its existing manufacturing agreement with Moderna that will enable it to manufacture large-scale volumes of Moderna’s lipid excipients to be used in making Moderna’s mRNA-1273 vaccine.
Emergent BioSolutions / J&J: Emergent and J&J have entered an agreement for Emergent to support manufacturing for J&J’s lead vaccine. Emergent is to provide drug substance manufacturing. The contract was reported to be worth $135 million.
Additionally, Emergent has been issued a task order under an existing contract with the Biomedical Advanced Research and Development Authority (BARDA), part of the Office of the Assistant Secretary for Preparedness and Response (ASPR) at the U.S. Department of Health & Human Services, to deploy its contract development and manufacturing capacities, capabilities, and expertise in support of the U.S. government’s efforts to accelerate delivery of COVID-19 vaccines. Total value of the task order is $628 million.
PolyPeptide Group / Novavax: The PolyPeptide Group, a global CDMO for peptide, peptidomimetic and other drug substances, has partnered with Novavax, a late-stage biotechnology company developing next-generation vaccines for serious infectious diseases, on large-scale good manufacturing practice (GMP) production of a critical component of Novavax’s coronavirus vaccine candidate, NVX-CoV2373.
Catalent / Ennaid: Catalent, a leading global provider of advanced delivery technologies, development and manufacturing solutions for drugs, biologics, cell and gene therapies and consumer health products, announced on May 13 a collaboration with Ennaid Therapeutics to develop an oral antiviral treatment targeted at COVID-19. Under the terms of the agreement, Catalent will develop a powder-in-capsule formulation of Ennaid’s ENU200 program, a patent-pending, repurposed oral antiviral drug.
Dyadic / Israel Institute for Biological Research: This research collaboration combines IIBR’s renowned scientific capabilities and current good manufacturing practice (cGMP) facilities with Dyadic’s patented and proprietary C1 gene expression platform, which is known to shorten development cycles, lower manufacturing costs and improve the performance of vaccines and monoclonal antibodies.
Interesting times ahead
While the pace of transactions in the space has slowed for the moment, particulary versus the past couple years, we expect increased fundraising and M&A going forward. We expect the global injection of intellectual and economic capital into drug development and manufacturing will seed new companies and reveal capability gaps among current players, setting the stage for a robust cycle of investment and consolidation.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
