Quarterly insights: Pharma IT
Mind’s role in therapeutics, wellness means opportunity for cognitive assessment technology

The increasing burden and costs associated with Alzheimer’s disease and other neurodegenerative diseases have made early diagnosis and intervention a national priority. However, conducting clinical trials to develop therapies for diseases like Alzheimer’s is particularly challenging, contributing to higher costs than for many other conditions. Cognitive assessment technology is an important solution to this challenge.
Applications for cognitive assessment technology extend beyond neurodegenerative diseases. Trial sponsors, regulators and physicians are increasingly interested in measuring and monitoring cognitive function during the drug development process to assess safety and risk for a broad spectrum of drugs. We think digital cognitive assessments, which trial sponsors have recently adopted out of necessity (often due to the pandemic) will continue to grow in popularity.
We highlight some of the companies providing cognitive assessment technology to trial sponsors for a variety of applications. Their capacity to improve understanding of cognition in healthcare offers enormous promise for technology providers, pharmaceutical companies, and quality of life.
TABLE OF CONTENTS
Includes discussion of Cogstate (ASX: CGS), Cambridge Cognition (AIM: COG), WCG Clinical (WCGC) and seven private companies
- Alzheimer’s and other neurodegenerative diseases dominate the cognitive health challenge
- Beyond hardware and chemistry: Key role for cognitive assessments in the neurodegenerative challenge
- Compelling case for cognitive assessments in drug development
- Aduhelm Alzheimer’s approval a vanguard of widening opportunity for cognitive assessments
- Opportunity for cognitive assessment technology beyond Alzheimer’s
- Rich opportunity for cognitive assessment technology providers and society as a whole
- eClinical index return far ahead of Nasdaq, S&P 500; commercialization index lags all
- Pharma IT M&A activity for Q2 and Q3 within two-year range
- Pharma IT private placements slow in Q2, Q3
Alzheimer’s and other neurodegenerative diseases dominate the cognitive health challenge
Cognition is defined as the mental action or process of acquiring knowledge and understanding through thought, experience and the senses. It involves various brain functions and processes such as knowledge, attention, memory, language, reasoning, judgement, comprehension, decision making and visuospatial function. Cognitive impairment refers to persistent deficits in the brain’s ability to function effectively. Cognitive impairment can have significant impact not only on the daily lives of patients but also their families. Depending on severity, signs of cognitive impairment, such as memory loss, confusion and difficulties carrying out routine day-to-day tasks, can be pronounced or subtle. Cognitive impairment is associated most notably with neurodegenerative diseases such as Parkinson’s disease, amyotrophic lateral sclerosis, Huntington’s disease and Alzheimer’s disease.
Alzheimer’s, the most common neurodegenerative disease, is a progressive brain disorder that slowly destroys memory and thinking skills, and eventually the ability to carry out simple tasks. According to statistics from the Alzheimer’s Association, an estimated 6.2 million Americans age 65 and older are currently living with Alzheimer’s. In addition to being the sixth-leading cause of death in the United States, it is costly. The current total national cost of caring for patients with Alzheimer’s other dementias is about $355 billion annually, not including an estimated $257 billion in unpaid caregiving. Without progress, the number of patients suffering from Alzheimer’s is expected to double to about 13 million by 2050, with annual costs projected to eclipse $1 trillion.
Beyond hardware and chemistry: Key role for cognitive assessments in the neurodegenerative challenge
The increasing burden and rising costs associated with Alzheimer’s have made early diagnosis and intervention an urgent national priority. According to the Alzheimer’s Association, advances in early diagnosis could save the United States an estimated $7.9 trillion. The problem is that Alzheimer’s is difficult to diagnose. There is no single test for Alzheimer’s. Physicians use a variety of tools to help diagnose the disease such as imaging (magnetic resonance imaging, positron emission tomography scans and single-photon emission computed tomography scans with specific radiotracers) and cerebrospinal fluid tests for various disease-associated proteins such as amyloid or tau. Unfortunately, neuroimaging and biomarker-based detection can be costly or inaccessible for patients.
TABLE 1: 2021 Alzheimer’s drug development pipeline
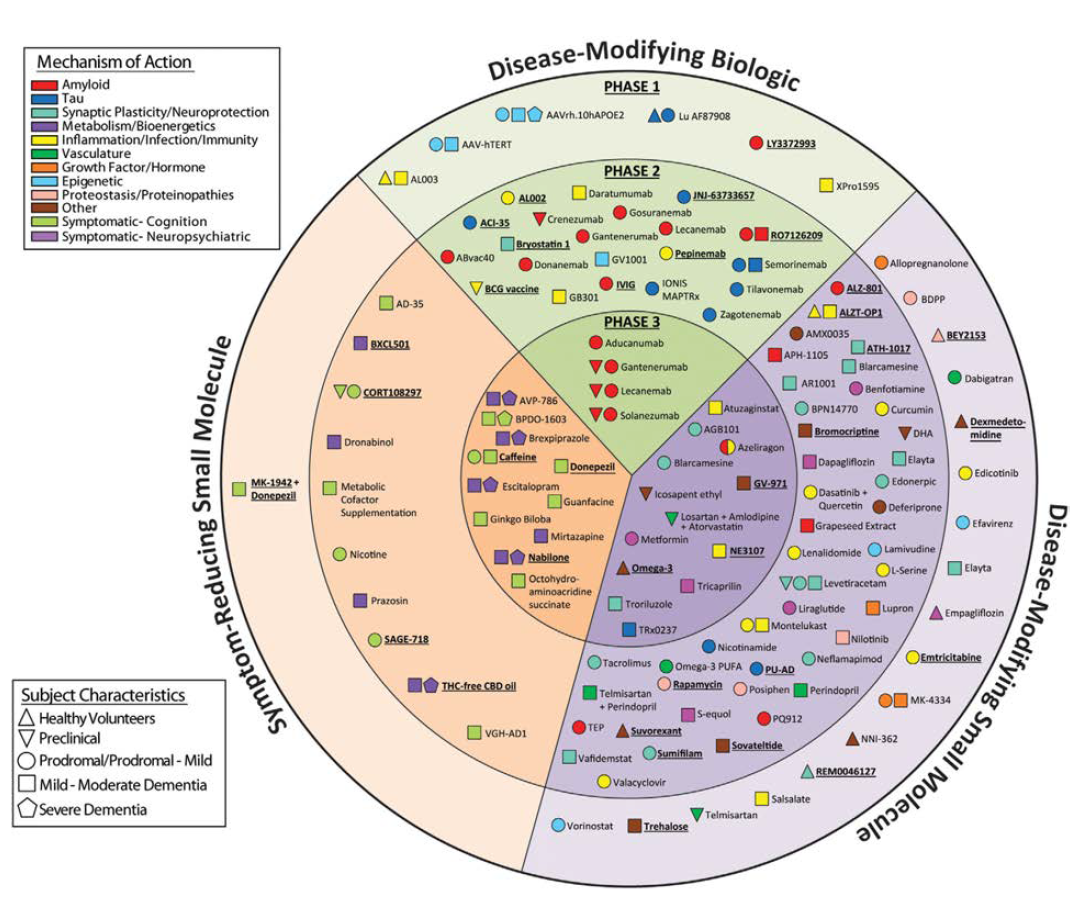
Source: Wiley Periodicals, May 2021. Alzheimer’s & Dementia: Translational Research & Clinical Interventions
As a result, cognitive assessments are being used to assist with early screening, diagnosis, treatment selection, patient monitoring and clinical research. Cognitive assessments are tests used to measure a patient’s ability to perform tasks and establish the severity of cognitive impairments over time. Cognitive assessments are typically administered in a battery of tests assessing domains including memory, attention, processing speed, visuospatial function and problem-solving. Traditionally, cognitive assessments are performed on-site in a clinical setting with pencil and paper or on a computer; however, the on-site assessment model tends to be burdensome for patients who are typically elderly and already struggling with mobility issues. In addition, they tend to be time consuming, requiring a trained specialist to administer, score and report on the results.
Compelling case for cognitive assessments in drug development
It takes an average of $2.6 billion, including cost of failures, and 10 to 15 years to discover, develop and win approval for a new drug. Additionally, only one of every 10 drugs that starts trials ends up being approved by the Food and Drug Administration (FDA). Alzheimer’s drug development costs are substantially higher than for other therapeutic areas, costing an average $5.6 billion, including the cost of failures, and taking over 13 years from study start to approval by the FDA. Between 1998 and 2017, there were 146 unsuccessful clinical trials for Alzheimer’s-related therapies. In that same time frame, only four new drugs were approved, none of which focused on Alzheimer’s underlying pathophysiology but rather on treating symptoms. Despite these setbacks, pharmaceutical companies remain committed to finding new therapies. As of January 2021, there were 126 drugs in 152 trials assessing new therapies for Alzheimer’s.
Several factors make conducting Alzheimer’s trials particularly challenging, contributing to the higher cost: Patients who are candidates for Alzheimer’s drug trials often refuse an invasive lumbar puncture to assess levels of proteins in the cerebrospinal fluid, and imaging can be expensive and difficult to schedule, especially during the pandemic. This makes it hard to screen, enroll and retain Alzheimer’s trial participants, leading to long and expensive clinical trials.
Cognitive assessments are an important tool in overcoming this challenge. Trial sponsors can use cognitive assessments in place of or in conjunction with physical assessments to make screening, enrolling and stratifying patients, detecting adverse events, and measuring outcomes more efficient and less costly. Cognitive assessments become even more powerful when combined with technology to automate and administer them remotely.
It can be challenging for clinical trial sites to hire and train qualified raters needed for manually administered tests, and it can be difficult to get Alzheimer’s patients to a centralized trial site. Administering cognitive assessments in decentralized clinical trials (DCTs) addresses these challenges. Decentralized trials are trials conducted either completely or partially outside traditional trial-site settings using digital tools that enable direct-to-patient support and remote monitoring. These digital tools include telemedicine, wearable devices and sensors, and electronic clinical outcome assessments (eCOAs).
Table 2: Cost and duration of each aspect of Alzheimer’s drug development
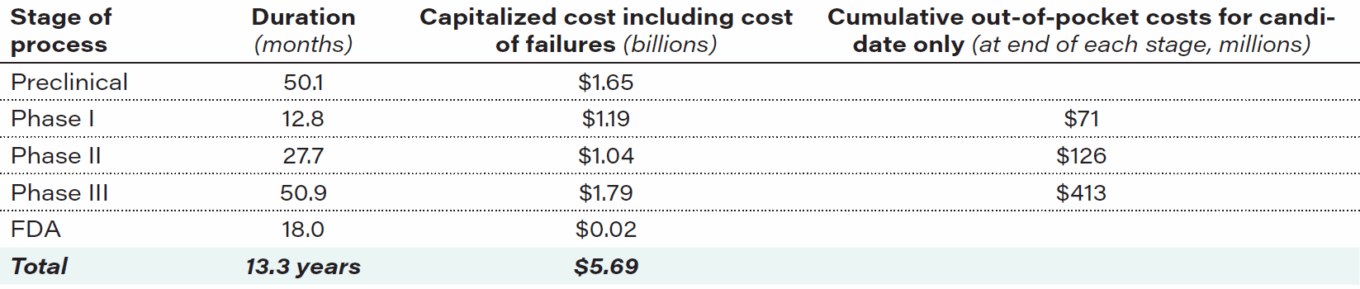
Source: Alzheimer’s & Dementia: Translational Research & Clinical Interventions, 2018: The price of progress: Funding and financing Alzheimer’s disease drug development.
The FDA describes a clinical outcome assessment (COA) as an assessment that directly or indirectly measures how patients feel or function and can be used to determine whether a drug has been demonstrated to provide a treatment benefit. Historically, paper was predominantly used to collect COA data in clinical trials, but electronic COA, or eCOA, has become the preferred method of data collection, particularly when COA data is being used as the primary or secondary endpoint for a clinical trial. Types of COAs include:
- Patient-reported outcome (PRO): A measurement based on a report that comes directly from the patient about the status of a patient’s health condition, without amendment or interpretation of the patient’s response by a clinician or anyone else.
- Observer-reported outcome (ObsRO): A measurement based on a report of observable signs, events or behaviors related to a patient’s health condition by someone other than the patient or a health professional. Generally, observer-reported outcomes are reported by a parent, caregiver or someone else who observes the patient in daily life, and they are particularly useful for patients who cannot report for themselves.
- Clinician-reported outcome (ClinRO): A measurement based on a report that comes from a trained healthcare professional after observation of a patient’s health condition. Most clinician-reported outcome measures involve clinical judgment or interpretation of the observable signs, behaviors or other manifestations related to a disease or condition.
- Performance outcome (PerfO): A measurement based on standardized tasks undertaken by a patient according to a set of instructions. A performance outcome assessment may be administered by an appropriately trained person or completed by the patient independently. Cognitive assessments fall into this bucket.
Using technology such as smartphones and tablets to remotely assess cognition brings some risks. Research has shown that test anxiety can have a significant effect on cognitive performance, and older patients using remote technology to complete cognitive assessments may experience anxiety and stress if they don’t understand test instructions, struggle to complete a task, or are generally uncomfortable using technology.
Additionally, participants may be embarrassed about their declining cognitive abilities and perform differently than they would in an on-site face-to-face testing environment, or they may be more tempted to give up rather than risk a wrong answer or forgetfulness. Caregivers may unintentionally interfere by repeating words or instructions or directing a participant’s attention (such as by pointing or signaling); indeed, the caregiver’s presence alone may affect participant performance.
Nonetheless, we think digital cognitive assessments, which trial sponsors have recently adopted out of necessity, often due to the pandemic, will continue to grow in popularity while a subset of more difficult and sensitive cognitive assessments will remain on-site.
Aduhelm Alzheimer’s approval a vanguard of widening opportunity for cognitive assessments
In June, the FDA approved Aduhelm, the first disease-modifying therapy for Alzheimer’s. It was the first new treatment for Alzheimer’s to be approved by the FDA in nearly 20 years and is the first therapy that targets the fundamental pathophysiology of the disease rather than the symptoms. Approvals of the first drug in a new category tend to invigorate the field, increase investment in new treatments and encourage greater innovation. To the extent this is true of Aduhelm, we expect adoption of cognitive assessment technology to gain further momentum.
TABLE 3: Cogstate Brief Battery

Source: Cogstate.
Aduhelm, jointly developed by Biogen (BIIB) and publicly traded Japanese pharma company Eisai, is a human-made antibody designed to remove toxic clumps of the protein beta-amyloid, which is thought to drive the death of brain neurons in Alzheimer’s patients. Australia-listed cognitive assessment technology provider Cogstate is a direct beneficiary of Aduhelm’s approval. Cogstate is a neuroscience-focused provider of digital cognitive assessments used in clinical research and clinical treatment. Its Cogstate Brief Battery (CBB) is expected to be used for screening and monitoring Aduhelm candidates. Cogstate CEO Brad O’Connor notes, “Cogstate’s solutions are about removing friction.” In October 2020, Cogstate entered into an exclusive 10-year agreement with Eisai for the global development rights and exclusive commercialization rights of all cognitive function tests developed by Cogstate.
Opportunity for cognitive assessment technology beyond Alzheimer’s
Current and future applications for cognitive assessment technology extend well beyond Alzheimer’s. Trial sponsors, regulators and physicians are increasingly interested in measuring and monitoring cognitive function during drug development to assess safety and risk for a broad spectrum of drugs. Some drugs can exert a cognitive effect at any time across a patient’s life. This includes not only central nervous system (CNS) drugs but also non-CNS therapies (oncology drugs, pain drugs, antihistamines, cardiovascular drugs including statins, AIDS drugs, and antimuscarinics).
For example, in 2012 a post-marketing surveillance study of statins uncovered several adverse effects tied to cognitive performance and psychological symptoms, including memory loss, amnesia, altered mood, irritability, sleep disruption and confusion. As a result, the FDA mandated warnings for all statin drugs for possible adverse effects on cognition.
Chemotherapy is often associated with cognitive impairment known as chemobrain. A recent study focused on the effects of physical activity and fatigue on cognitive performance in breast cancer survivors found that physical activity reduced fatigue and had a positive impact on cognition. The researchers used an innovative mobile platform from BrainBaseline that includes self-administered cognitive and motor assessments capable of detecting neurocognitive impairments. According to CEO Joan Severson, “Regardless of indication, BrianBaseline’s platform provides trial sponsors with a novel quality-of-life understanding in the real world by capturing cognition in the wild.”
In another example, BrainBaseline is developing digital biomarkers for Parkinson’s disease through the Watch-PD clinical trial sponsored by the University of Rochester in collaboration with Biogen and Takeda. In our view, the observational study’s design allows for more unbiased real-world measurement across the different populations who suffer from Parkinson’s disease and the variety of symptoms they exhibit. The study is expected to be completed in 2022. Once validated, the biomarkers are expected to be used to help assess other movement and mobility disorders.
TABLE 4: BrainBaseline’s mobile platform for assessing motor and cognitive aspects of Parkinson’s disease

Source: Biogen, BrainBaseline.
Another company, eClinical solutions provider ERT, has partnered with Cogstate to expand ERT’s industry-leading eCOA solution with digital cognitive endpoint measurement to enhance safety and efficacy assessment in clinical trials that include capabilities for at-home testing. (The Cogstate-Eisai agreement specifically excludes clinical trials.)
EMA Wellness focuses on ecological momentary assessments (EMA). According to EMA Wellness Chief Scientific Director Phil Harvey, “The company’s technology allows for fundamentally better outcomes by capturing the variability in trial participant behaviors.” EMAs collect data in real time and in real-world settings to avoid retrospective biases and memory distortions, to collect ecologically valid data, and to study behavioral processes over time. An EMA enables more frequent sampling (often multiple times a day), so time-series analysis can be performed, providing a deeper understanding of processes at work versus static snapshots at distant time points.
The premise is that single-time-point assessments fail to capture variability in human cognitive performance affected by a host of factors, including mood, stress and time of day. EMAs have been used in numerous clinical trials, including to identify the best predictors of substance use, a way to identify anxiety symptoms and behaviors, and as a way to track elderly people’s activities. For example, the company’s EMA smartphone app was recently used to track the course of symptoms in a depression trial. Trial participants received daily queries throughout the screening and treatment phases of the study. Participants were queried twice daily, including on the in-person clinic visit days.
Gamification can be an important element of cognitive assessment technology. Gamification involves extracting the elements of gameplay that trial participants find compelling and using them to drive trial engagement and compliance without undermining scientific value. Datacubed Health’s mobile eCOA platform Linkt incorporates behavioral science tools such as reward systems, progress and feedback to provide participants with an intrinsically motivating experience that promotes participant retention and compliance in longitudinal studies.
Datacubed Health Chairman and founder Paul Glimcher explained, “It is a basic tenet of user experience science that, if the technology can be well aligned with a participant’s identity, it can significantly improve the quality and timeliness of compliance.” Linkt has a library of cognitive assessments, can collect audio recordings of patients performing speech and language tasks, and includes a survey builder. The app also integrates passive data, including global positioning system and pedometer data.
TABLE 6: Examples of Linkt app mobile user interface

Source: Datacubed.
Signant Health’s SmartSignals eCOA is described as a validated and flexible cognitive assessment platform designed for repeated use in clinical trials. It comprises a range of tests assessing essential cognitive domains relevant to everyday life activities such as attention, working memory, episodic memory and executive function.
London-traded Cambridge Cognition has developed a suite of computerized neuropsychological tests called the Cambridge Neuro-psychological Test Automated Battery (Cantab). Cantab is widely used in academic, clinical and pharmacological research and has been published in over 2,000 peer review papers. Cantab tests include a suite of cognitive assessments measuring aspects of cognitive functioning in therapeutic areas including attention and psychomotor speed, executive function, memory, emotion and social cognition. Users initially administered Cantab on personal computers, but the company now offers a tablet-based Cantab mobile application. Cambridge Cognition also offers an online platform for trial recruitment that pre-screens patients using its cognitive assessment instruments.
Decentralized clinical trial platform provider Thread offers virtual visits via telehealth technology that enables trial staff to instruct, administer and monitor assessments remotely. Thread’s platform is agnostic to the cognitive assessment being used during the trial and can partner with vendors offering their own proprietary battery of tests. According to Thread CEO John Reites, “It’s not the what, but the how it’s being captured” that’s critical to Thread.
eClinical solutions provider WIRB-Copernicus Group (WCG) has acquired several cognitive assessment technology companies. In May 2017, the company acquired MedAvante and ProPhase, and in July 2021, it acquired VeraSci (formerly NeuroCog), a cognition and clinical assessment solutions company. Via its Pathway technology platform, VeraSci enables remote collection of high-quality data that can be used to assess symptoms, cognition and functional capacity in a wide range of therapeutic trials including CNS-related trials.
Rich opportunity for cognitive assessment technology providers and society as a whole
Cognitive assessment technology makes it easier and faster to achieve improved understanding of cognition to advance healthcare. Prospects for increased use of cognitive assessment technology in drug development, and ultimately in healthcare more broadly, offer enormous promise for technology providers, pharmaceutical companies, and quality of life throughout our society.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
