Quarterly insights: Pharma IT
Validation tech provides critical protection and a compelling opportunity

The proliferation of new technologies and capabilities in healthcare introduces new risks. How can companies keep error from creeping into big data sets and AI output? How can companies deploy new technologies and capabilities so they interact effectively with legacy systems? Validation processes are an unglamorous but critical key to addressing these challenges.
The FDA mandates critical systems, including software, used in manufacturing, storing and testing products be validated and kept in a validated state throughout their life cycles. Recently, the FDA has been encouraging life science companies to adopt computer system validation (CSV) software that enables a risk-based approach to validating computer systems.
As a result, the market for CSV software is seeing robust growth. We highlight CSV vendors we see making inroads in the market with software solutions that better meet life science companies’ CSV needs.
TABLE OF CONTENTS
- Technology’s benefits come with challenges in making sure it works as intended
- What is validation?
- Computer system validation
- Examples of systems requiring CSV
- The FDA wants companies to use software for more efficient, accurate CSV
- CSV market dynamics
- Competitive landscape
- Another CSV software user base: Outsourced validation service providers
- Another technology tool for building a better healthcare ecosystem
- Pharma IT indexes lose momentum, barely positive in past year
- Pharma IT M&A: Notable transactions include Pro-ficiency and Model N
- Pharma IT private placements: Notable transactions include CRIO and ProofPilot
Technology’s benefits come with challenges in making sure it works as intended
The ongoing digital transformation of the life science industry has created significant opportunities to improve healthcare for patients worldwide. With developments like big data and artificial intelligence (AI), life science companies are now equipped to address complex challenges and bring innovative treatments to market more swiftly. But the proliferation of new technologies and capabilities also introduces new risks. How can companies keep error from creeping into big data sets and AI output? How can companies deploy new technologies and capabilities so they interact effectively with legacy systems across the large, diverse healthcare ecosystem? These are just a few of the challenges that have come with the digital transformation of the life science industry. Validation software is one of the keys to addressing these challenges.
What is validation?
Under FDA regulations that encompass good manufacturing practice (GMP) and related best practices (collectively known as GxP), it is essential to “validate” systems that directly or indirectly affect products and patients before they are used in production environments. These GxP-related systems include quality management systems, electronic document management systems, batch record systems, laboratory information management systems, clinical systems, product safety and compliance reporting systems, environmental monitoring systems, and systems for transferring and analyzing production data.
Validation is defined as the collection and evaluation of data that establishes evidence that a process is capable of consistently delivering quality product. Validation is a compliance matter and is the responsibility of life science companies’ quality teams. These teams ensure their organizations remain compliant with regulations, particularly FDA and European Medicines Agency regulations. They are responsible for maintaining data, documentation, and auditability over entire product life cycles, which often span 20 years and sometimes longer. Validation is a specialized field that requires extensive expertise.
Computer system validation
The FDA mandates all critical systems used in manufacturing, storing and testing products be validated and kept in a validated state throughout their life cycles. This mandate encompasses both drugs and medical devices and includes clinical trials. Computer system validation covers a large portion of the broader validation domain. FDA 21 CFR Part 11 requires companies to ensure computer systems are suitable for their intended purposes by rigorously testing and documenting that their features and functions operate accurately, reliably, consistently, and in compliance with relevant regulatory standards. CSV is integral to the life science industry.
Apart from being a regulatory requirement, CSV is also very important because it helps identify and close any gaps in companies’ computer systems and ensure data is accurate and secure. If CSV efforts fail to achieve these objectives, companies risk costly consequences, including receiving Form 483 from the FDA, receiving warning letters from the FDA and incurring monetary fines.
Examples of systems requiring CSV
The following are descriptions of some of the major categories of software and systems life science companies must validate and what CSV does.
- Laboratory information management systems (LIMS): LIMS are crucial for handling laboratory workflows, data and sample tracking and for ensuring data integrity and traceability. CSV confirms accurate data recording and sample tracking and compliance with testing and reporting procedures.
- Electronic document management systems (EDMS): EDMSs manage electronic documents, records and workflows. They are essential for organized documentation like standard operating procedures, batch records and regulatory submissions. CSV ensures secure storage, accessibility and compliance with version control.
- Quality management systems (QMS): QMSs manage quality-related events such as deviations, corrective and preventive actions, change controls and audits. They are critical for compliance and tracking corrective actions. CSV ensures these processes are consistent and well documented.
- Manufacturing execution systems (MES): MESs manage manufacturing processes, batch records, equipment and personnel, ensuring operations are controlled and compliant with GMP requirements. CSV helps prevent errors in batch records, ensuring product consistency and quality.
- Enterprise resource planning (ERP) systems: ERP systems integrate various business processes, including inventory management, procurement, finance and human resources. In regulated industries, ERP systems track materials, ensure accurate financial reporting and maintain regulatory compliance. CSV ensures data accuracy and integrity within ERP modules.
- Clinical trial management systems (CTMS): CTMSs manage clinical trial data, including patient enrollment, study protocols and regulatory submissions. These systems ensure the integrity and accuracy of clinical trial data, which is essential for regulatory submissions and patient safety. CSV safeguards the reliability of clinical trial data.
- Pharmacovigilance systems: These systems handle adverse event reporting and safety surveillance for pharmaceutical products, which is crucial for patient safety and regulatory compliance. CSV ensures adverse event data is accurately captured, assessed and reported.
- Regulatory information management (RIM) systems: RIM systems manage regulatory submissions, approvals and compliance information, supporting timely submission of regulatory documents and compliance. CSV ensures regulatory information is accurate and current.
- Process control systems: These systems, which are used in manufacturing environments to monitor and control critical process parameters, ensure consistent product quality and adherence to GMP requirements in industries like pharmaceuticals and biotechnology. CSV safeguards the accuracy of process control data.
- Data analysis and reporting software: Any software used for data analysis, reporting and decision-making in regulated environments requires CSV. This includes statistical analysis tools, data visualization software and reporting tools used for generating data-driven insights.
The FDA wants companies to use software for more efficient, accurate CSV
CSV is a time-consuming and labor-intensive process, still predominantly done on paper. But manually transferring data between documents and physically moving and storing paper documents can lead to data integrity issues, compliance risks, and logistical challenges, potentially resulting in significant regulatory issues, fines and product recalls. Of all FDA warning letters issued to pharmaceutical companies in 2021, 65% related to data integrity, up from 51% in 2020.
To address these problems, the FDA has been encouraging life science companies to adopt a “least burdensome approach to validation.” In 2022, the FDA released its latest proposed guidance for CSV, titled Computer Software Assurance for Manufacturing, Operations, and Quality System Software (CSA). This draft guidance aims to clarify the FDA’s expectations for software validation and promote a more agile, iterative approach to validating software. The guidance encourages moving away from exhaustive paper-based validation processes to adopting software that enables a risk-based approach to CSV. While the guidance is non-binding and not a final rule, it signals the FDA’s intention to finalize the rule, pushing the industry toward more efficient and streamlined compliance processes.
CSV market dynamics
The CSV market has seen robust growth in recent years, with annual growth estimated in the 15-20% range. This surge has been fueled by regulatory demands, the rapid adoption of cloud-based applications in the pharmaceutical industry, and the shift toward continuous integration/continuous delivery practices. As pharmaceutical companies expand and implement more systems with frequent updates, their CSV workloads increase significantly. Each system may require from three to 12 mandatory software updates annually, depending on the technology used.
According to the State of Validation 2023 report, CSV is the most prevalent work process in day-to-day life science operations, and roughly two-thirds of 153 survey respondents (mainly consisting of professionals in the life science sector) indicated CSV regularly consumes more than 8% of total project spend. Considering industry leader Veeva Systems’ (VEEV) total addressable market (TAM) estimate of $20 billion for the life science software market, we estimate a $1.6 billion TAM for CSV. In terms of market penetration, we estimate around 25% of life science companies have adopted a fully digital CSV solution. This indicates a substantial opportunity for CSV software providers to capture new business, as we expect CSV workloads and budgets will continue to rise over the next five years.
Competitive landscape
Below we highlight CSV vendors we see making inroads in the market with software that better meets life science companies’ CSV needs.
Publicly traded in Canada, Kneat is considered the market leader in validation. Kneat was founded by former industry executives in Limerick, Ireland, in 2006. It introduced its current validation solution, Kneat Gx, in 2014, and went public in 2016. At the end of the first quarter, Kneat reported annual recurring revenue (ARR) of approximately $42.1 million, up 57% year-over-year. In June, the company signed a partnership agreement with Körber, a Germany-based life science tech-enabled services provider. The agreement expands Kneat’s go-to-market effort by enabling Körber to resell and implement Kneat Gx and offer managed services to its customers using the Kneat Gx platform.
The Kneat dashboard provides a user-friendly interface
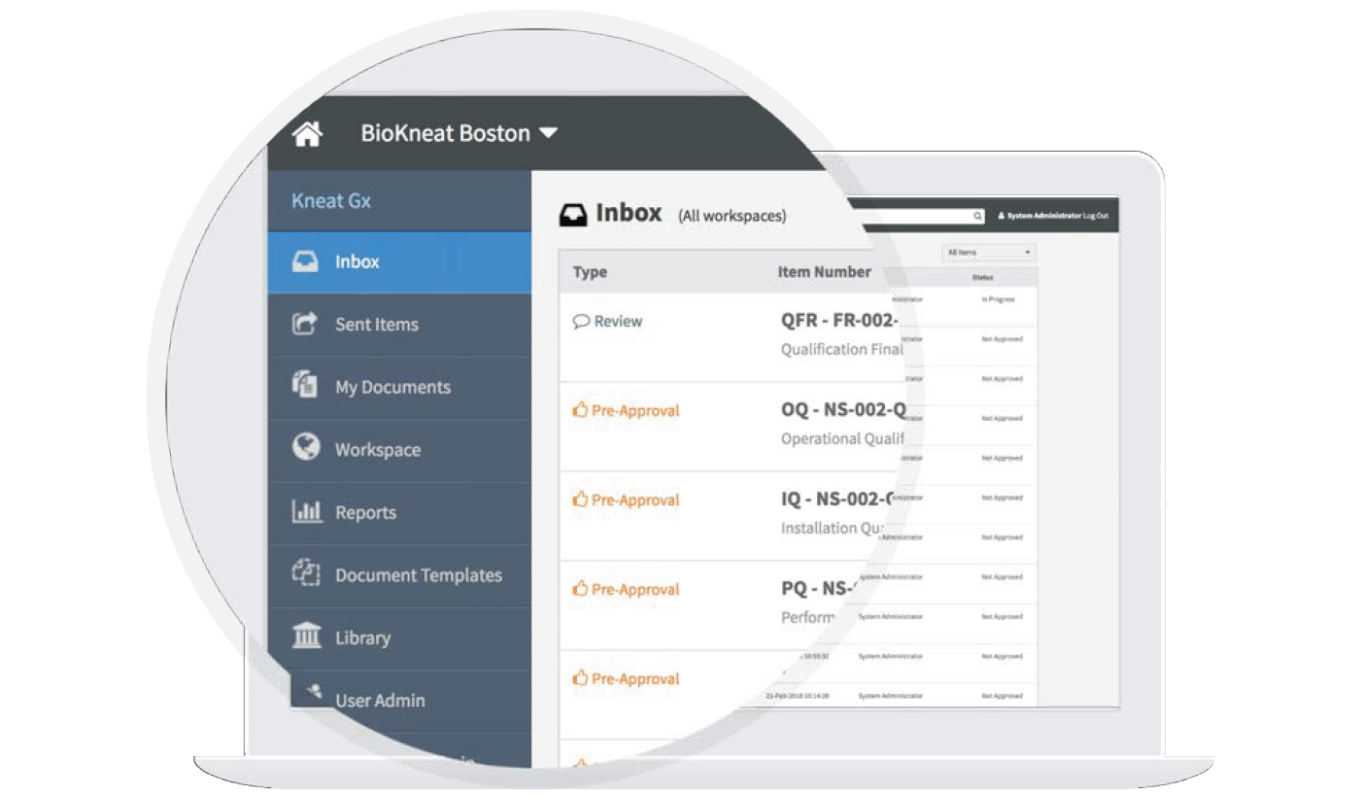
Source: Kneat.
MasterControl, based in Salt Lake City, provides life science software focused on quality and operations. In 2017, MasterControl CEO Jon Beckstrand spearheaded development of the Validation Excellence Tool (VxT) as part of the company’s overall initiative to improve software validation. VxT is a cloud-based application designed to streamline risk-evaluation processes. It evaluates multiple risk areas, such as software usage, impact of failure, regulatory requirements, variation from best practices and testing mitigations. VxT was designed with the assistance of a former director of IT for the FDA to ensure it satisfies regulatory requirements and meets regulatory standards.
Sware, a First Analysis venture capital portfolio company based in Chelsea, Massachusetts, was founded in 2015 as a managed services business and released its digital CSV platform (Res_Q) in 2019, shifting the business to being a software-as-a-service (SaaS) provider. Res_Q provides a streamlined workflow through an intuitive interface that automates, unifies and accelerates software validation in a growing and complex digital compliance environment for life science companies. Res_Q unifies over 30 industry-supported applications from a list of vendors that includes Veeva, Nuvolo, Okta, Docusign, SAP, MasterControl, TrackWise, Tracelink, Oracle and OpenClinica, to name a few. CEO Ellen Reilly sees “an opportunity to transform a heavily regulated industry’s approach to software validation. Our risk-based technology allows companies to simplify continuous SaaS product validation using automated workflows and AI-supported functionality. This approach is fundamentally disruptive in an industry that has primarily relied on document management and outside consulting for nearly two decades.”
Sware dashboard
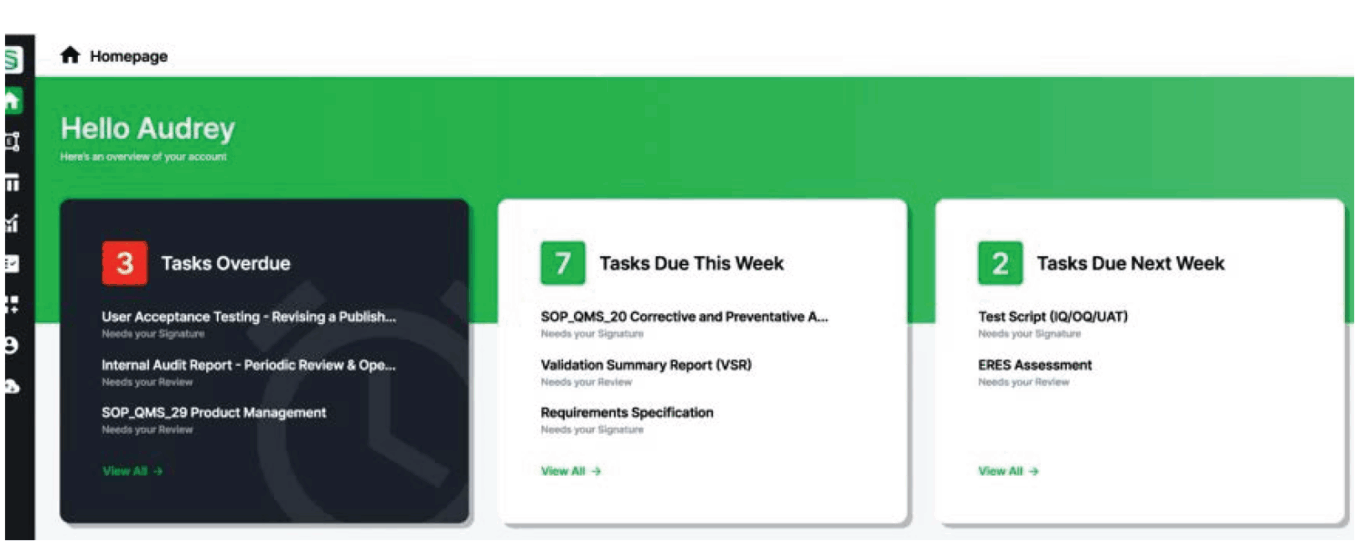
Source: Sware.
Founded in 1999 in Santa Barbara, USDM Life Sciences was primarily a managed services provider helping biotech, medical device and pharmaceutical companies with GxP compliance until 2021, when it launched ProcessX, a fully validated GxP process automation software solution built on the ServiceNow platform. ProcessX is designed to manage the entire software development life cycle from inception to decommissioning with data-driven application life cycle management.
USDM Life Sciences’ ProcessX dashboard
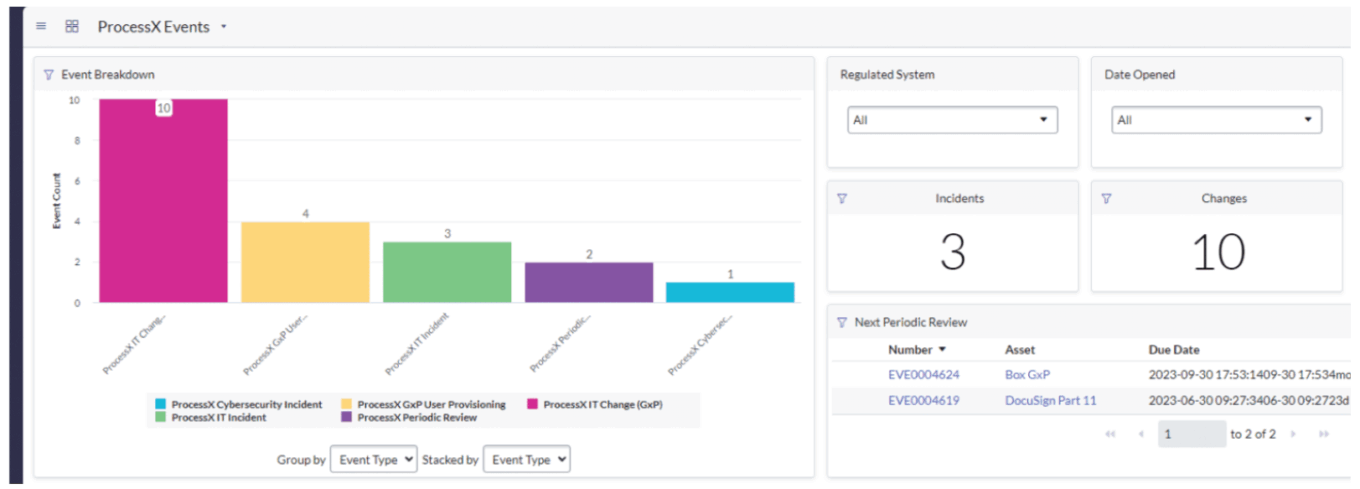
Source: USDM Life Sciences.
Founded in 2005 and headquartered in Fremont, California, ValGenesis offers its flagship Validation Lifecycle Management System (VLMS) to help life science companies enforce compliance and enable standardization with digital, risk-based validation software. In early June, the company announced its launch of VLMS 5.0 with a redesigned user interface focused on simplicity, usability and data integrity. The platform natively supports risk-based testing, automated and unscripted testing methodologies, and assurance activities needed for regulatory compliance. In July, the company announced a strategic reseller partnership with Validator K.K., a well-known Japanese validation product and service company. The partnership leverages Validator’s on-site validation service model alongside ValGenesis VLMS for the Japanese life science sector.
ValGenesis’s Validation Lifecycle Management System dashboard
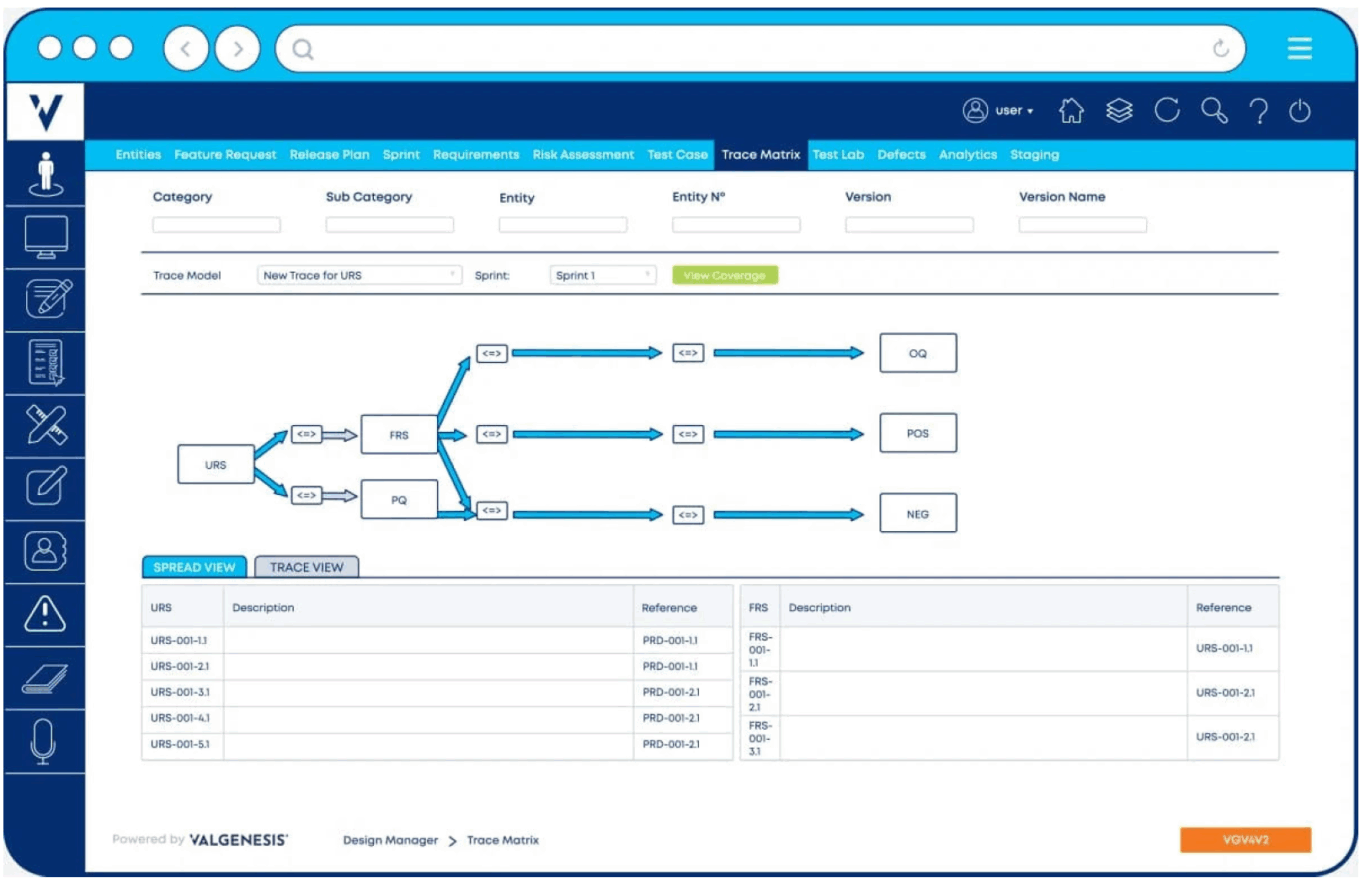
Source: ValGenesis.
Veeva Systems introduced its validation product, Vault Validation Management, in 2021. Vault Validation Management is an application in the Vault Quality Suite designed to manage and execute paperless validation across multiple validation areas including CSV; facility, equipment, and utility; process validation; cleaning validation; and method validation. Integrated with Vault QualityDocs and Vault QMS, Vault Validation Management centralizes tracking system inventory, requirements, and project deliverables, with the aim of simplifying workflows for users.
Veeva’s Vault Validation Management software
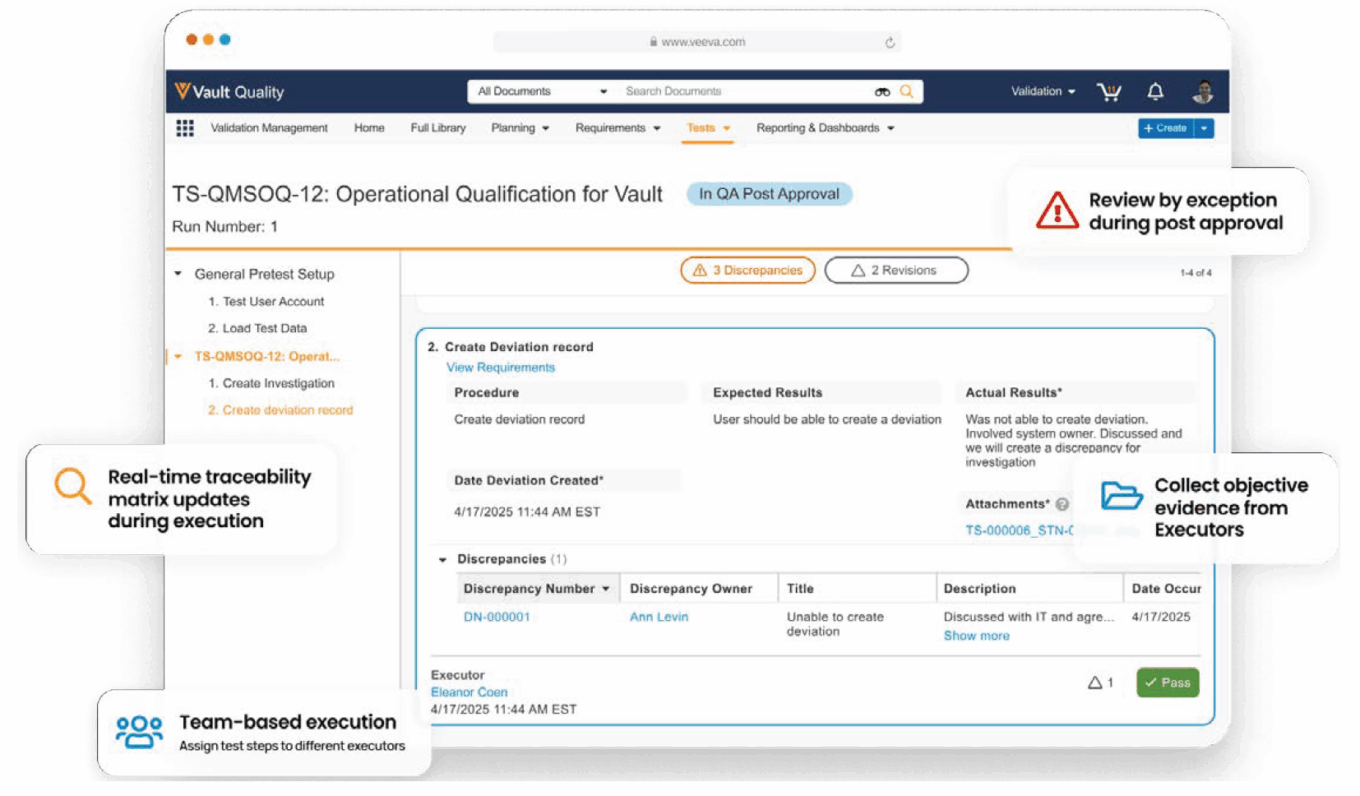
Source: Veeva Systems.
Another CSV software user base: Outsourced validation service providers
Outsourced validation service providers address part of the life science sector’s need for CSV. These providers help life science companies deal with the limited availability of CSV expertise and the variable nature of their CSV needs, which depend on the timing and volume of internal production projects. Although some outsourced validation service providers have attempted to create their own CSV software, the majority seem to be using existing platforms that best serve their clients.
Another technology tool for building a better healthcare ecosystem
It seems there are few realms of healthcare where software is not a powerful tool for improving quality and outcomes, and there are plentiful frontiers where software has yet to reach its full potential in healthcare. We think this is a particularly ripe moment for CSV software to fulfill its potential in the life science sector and, ultimately, to contribute meaningfully to the healthcare ecosystem.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
