Quarterly insights: Pharma IT
Industry forums highlight 5 key themes in pharma IT

Two recent industry events, the First Analysis Healthcare Technology Summit and SCOPE, highlighted and provided insights on five key themes underpinning investment and transaction activity in pharma IT.
First, transaction activity appears likely to accelerate in 2026, but with discipline and structural caution. Second, platforms that emphasize infrastructure are driving pharma IT investment. Third, rather than positioning AI as a standalone solution, companies are increasingly embedding it into existing clinical and operational workflows and using it to support decisions incrementally rather than automate them outright.
Fourth, strategic acquirers’ focus continues to converge around real-world evidence and trial enablement, as these capabilities address inefficiencies in drug development and commercialization while offering tangible return on investment. Lastly, the importance of operational discipline and compliance is increasing, accelerating demand for enabling technologies.
TABLE OF CONTENTS
Two pivotal industry forums
The First Analysis Healthcare Technology Summit and the SCOPE Summit provided a concentrated view of current priorities, with a meaningful number of companies actively evaluating transactions or capital raises.Transaction activity to reaccelerate, but with discipline and structural caution
Transaction activity is expected to increase gradually, with buyers and investors remaining disciplined on valuation, diligence and structure and continuing to favor scaled, de-risked assets.Platforms that emphasize infrastructure are driving pharma IT investment
Investment is increasingly directed toward infrastructure-driven platforms that support discovery, development, commercialization and care delivery and are viewed as essential to day-to-day operations.AI is helpful but not a silver bullet
The idea that AI can independently replace human expertise in pharma has largely faded, replaced by a more pragmatic view that lasting impact depends on structure, iteration and experienced oversightReal-world evidence remains a strategic priority
Strategic acquirers continue to focus on real-world evidence and trial-enablement capabilities that address inefficiencies in drug development and commercialization and provide tangible return on investment.Operational discipline and compliance demand accelerating
The importance of operational rigor, validation and auditability is increasing, driving demand for technologies designed to natively support inspection readiness, standardized documentation, clear audit trails and traceable decision-making.First Analysis Healthcare Technology Summit 2026: Life science & pharmacy private company snapshot
A curated group of 37 private companies provides a focused view of the life science and pharmacy technology landscape represented at the summit.Pharma IT public market performance, M&A and private placements
Overview of pharma IT public market performance and valuation, sector M&A activity — including IQVIA’s acquisition of Throtle and AstraZeneca’s acquisition of Modella AI — and private placements, including financings by Lindus Health and Paradigm Health.
Two pivotal industry forums
We recently participated in two pivotal healthcare industry forums that yielded insights on a handful of key themes for pharma IT.
In January, First Analysis hosted its 12th Annual Pharma IT/Healthcare Technology Summit. Each year, the event brings together a highly curated group of pharma IT and healthcare technology companies, leading growth equity investors and strategic acquirers.
The First Analysis summit has become one of the most valuable gatherings held alongside J.P. Morgan’s annual healthcare conference in San Francisco, an anchor event that makes the city a healthcare-sector hub for back-to-back meetings, events and informal conversations. Each year the First Analysis summit delivers a highly productive day of meetings and conversations. It uses a speed-dating format, giving host companies the opportunity to conduct nine 20-minute, one-on-one meetings with no set agenda, allowing each conversation to go wherever it is most productive. This low-pressure structure creates an ideal environment for open, candid dialogue and meaningful early stage relationship building while also giving groups considering next steps an efficient way to assess alignment.

In February, we attended the SCOPE Summit (Summit for Clinical Operations Executives), the flagship annual conference in Orlando, Florida, focused on clinical research and clinical trial operations. SCOPE brings together leaders across the life science ecosystem to share insights, strategies, innovations and best practices that help advance how clinical trials are designed and run.
The mood across both conferences felt decisively action-oriented: A meaningful number of companies we met with were in the process of evaluating transactions or capital raises. Below, we highlight five themes that were prominent at both conferences along with additional insights that help shape our thinking for 2026.
1. Transaction activity to reaccelerate, but with discipline and structural caution
Transaction activity appears likely to increase in 2026, but the recovery is shaping up to be gradual, rather than explosive as it was in the 2020-2021 time frame. The market has shifted away from expectations of a sharp rebound in transactions toward a more practical rebuild, with buyers remaining disciplined on valuation, diligence and transaction structure. This stands in contrast to 2025, when optimism often outpaced reality and transaction volume ultimately came in below forecasts.
TABLE 1: Biopharma venture capital transactions by quarter (dollars in billions)
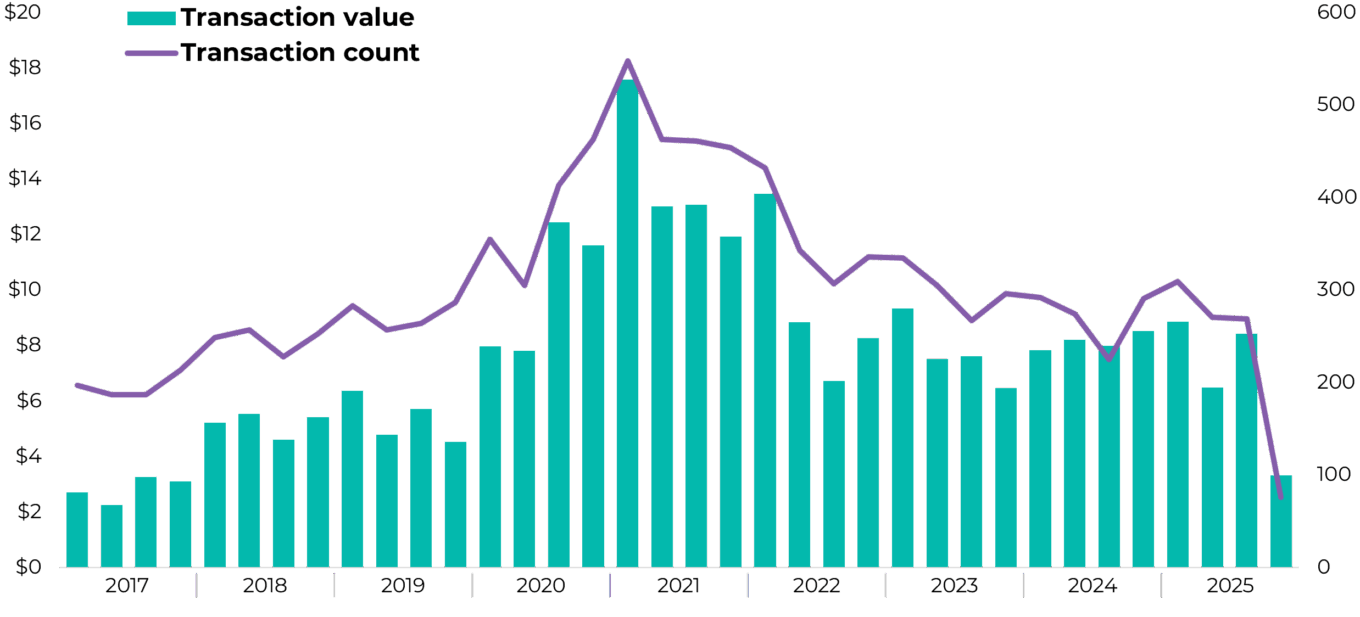
Source: PitchBook.
An important barometer we use to help gauge the state of the market is biopharma venture capital investment: the number of transactions and the amount of capital invested in biopharma businesses. Historically, there has been a strong association between dollars invested in biopharma and increased adoption of digital solutions to run clinical trials.
TABLE 2: Biopharma venture capital transaction value by stage (dollars in billions)
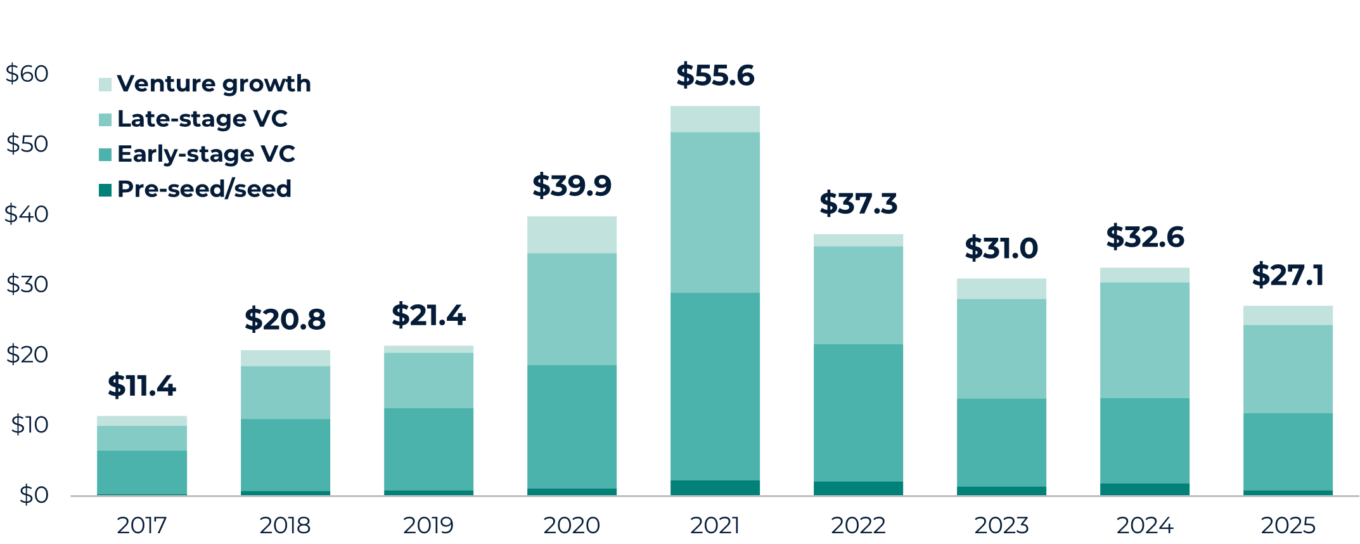
Source: PitchBook.
While transaction counts in 2025 were muted relative to the prior five years, transaction values have shown modest improvement, driven by a handful of larger financings and later-stage rounds. Investors continue to favor clinical-stage and de-risked assets, reflecting lingering caution following the capital misallocation of the 2020-2021 funding surge. Early stage activity remains lower, with pre-seed and seed transactions representing a smaller share of funding than historical averages as capital continues to concentrate around assets with clearer paths to commercialization.
A similar pattern is evident in life science private equity, where declining transaction counts in 2025 were paired with a rebound in total buyout value. Fewer transactions are getting done, but the ones that clear the market are larger and more deliberate, reflecting higher conviction and tighter underwriting standards. Growth and expansion activity has also remained muted, reinforcing the broader shift away from volume-driven investing toward scale, durability and cash-flow visibility.
These dynamics help explain two trends from 2025 that are likely to carry into 2026: a continued bias toward add-on acquisitions versus new platform creation and longer holding periods across private equity portfolios. With financing costs still elevated and valuation gaps unresolved, sponsors are prioritizing expanding existing platforms through tuck-in acquisitions and operational improvement while delaying exits. As a result, transaction activity is expected to remain focused on lower-risk, incremental transactions rather than aggressive platform building, even as overall volume begins to recover.
TABLE 3: Global life science private equity transaction count and value by type (dollars in billions)
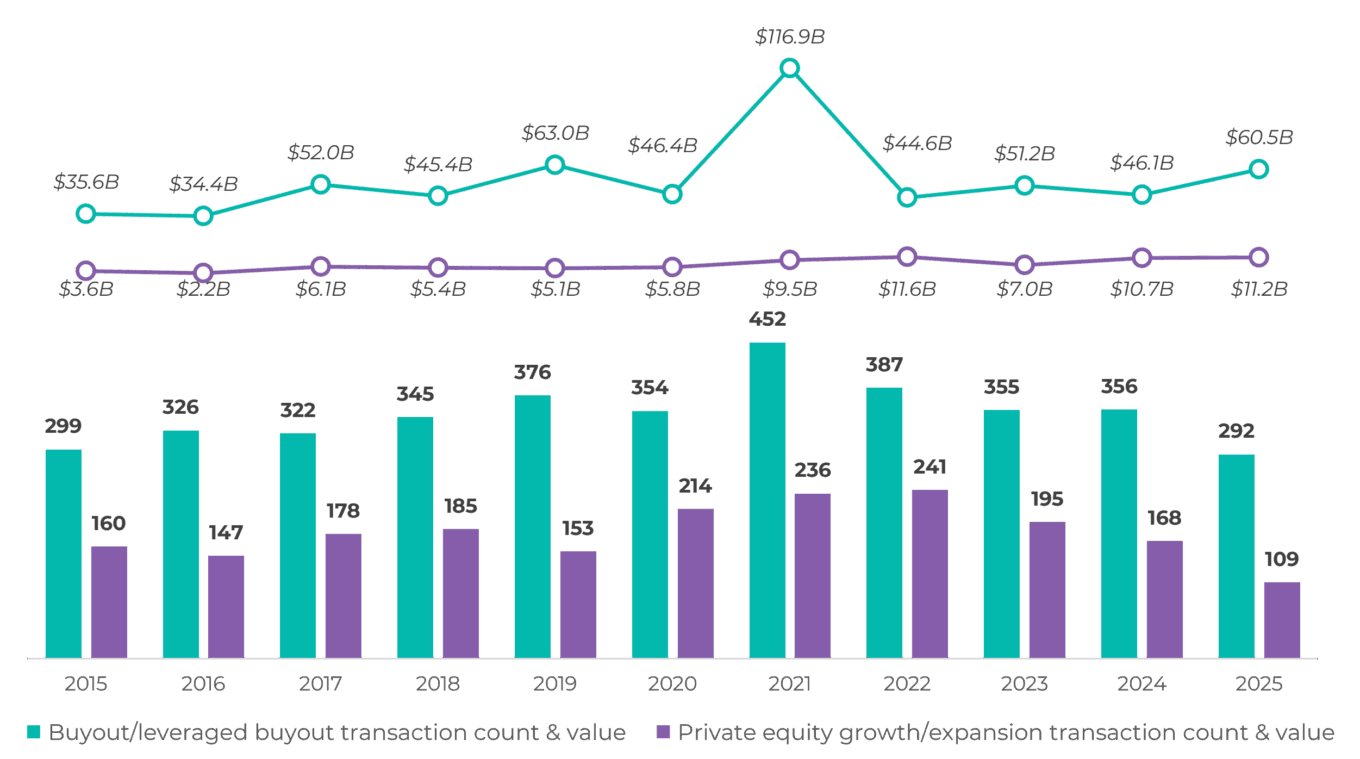
Source: PitchBook.
Strategic buyers are also expected to remain disciplined and selective in 2026, with merger and acquisition activity skewing toward capability-driven tuck-in acquisitions rather than large, transformational transactions. Strong balance sheets will likely keep select strategics active, but most are likely to use acquisitions to fill product gaps, accelerate AI and digital initiatives and enter attractive adjacencies without taking on outsized integration risk or capital intensity.
2. Platforms that emphasize infrastructure are driving pharma IT investment
A key current theme is the move toward infrastructure-driven platforms: platforms that support discovery, development, commercialization and care delivery that are viewed as essential and drive efficiencies in day-to-day operations. This shift reflects a clear preference among buyers and investors for business models built around predictability, extensibility and repeatable value creation, particularly in an environment where execution risk is closely scrutinized. These platforms enable pharma and healthcare organizations to accelerate timelines, improve decision making and standardize processes across programs, while keeping clinical and regulatory risk concentrated within the asset portfolio rather than the technology layer.
This shift toward infrastructure-driven platforms is being reinforced by increasing convergence across the healthcare ecosystem. Traditional boundaries between payers, providers, pharma IT and digital health continue to blur, driving demand for platforms that can operate horizontally across stakeholders and use cases. Value is increasingly accruing to technologies that connect data, workflows and decision makers end-to-end, supporting everything from research and development and market access to care coordination and outcomes measurement. In this environment, platform depth has become the defining attribute. Solutions that combine technical scale, domain expertise and integration readiness are better positioned to grow with customers as priorities shift, indications expand and market conditions evolve, making them structurally more resilient than point solutions designed for narrow use cases.
3. AI is helpful but not a silver bullet
There is a clear pullback from broad AI hype toward a more execution-focused mindset. While AI can be effective in well defined, discrete tasks, it continues to struggle in environments that require contextual judgment, cross-functional coordination and accountability for high-stakes decisions. The idea that AI can independently replace human expertise in pharma has largely faded, replaced by a more pragmatic view that lasting impact depends on structure, iteration and experienced oversight.
Our recent conversations reinforced our perception of this shift toward AI realism. Rather than positioning AI as a standalone solution, teams are increasingly embedding it into existing clinical and operational workflows and using it to support decisions incrementally rather than automate them outright. The emphasis is on practical integration, where AI surfaces insights and reduces friction while practitioners exercise judgment and retain ownership. AI adoption is concentrated in enterprise technology, research and development, and medical functions where workflows are relatively structured, data is well defined, and ownership is clear, rather than in areas that require heavy cross-functional judgment and open-ended decision making.
TABLE 4: Percentage of leaders planning to streamline operations with AI agents, by workflow type
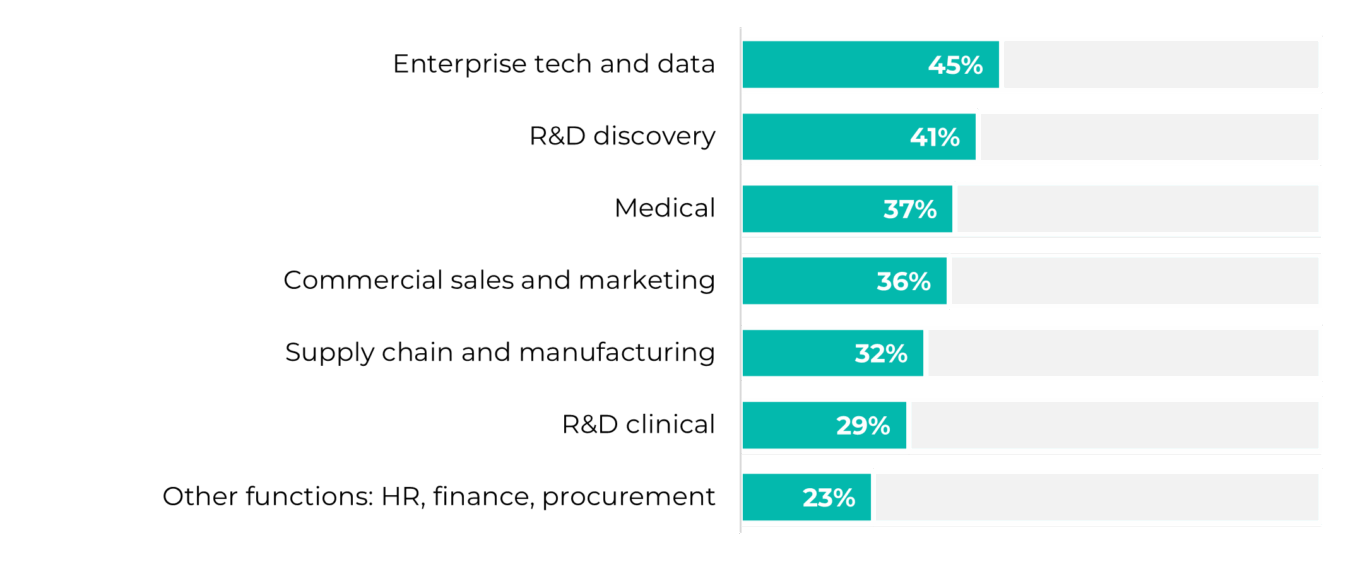
Source: ZS Associates: Scaling AI in pharma and biotech: 2026 outlook from ZS’s CDIO research, Nov. 3, 2025.
Taken together, these discussions point to a broader move away from sweeping AI narratives toward narrower, execution-driven uses grounded in domain expertise. The most credible approaches pair AI with proprietary data and workflows designed for adoption rather than disruption.
4. Real-world evidence remains a strategic priority
Strategic acquirers’ focus continues to converge around real-world evidence (RWE) and trial enablement, as these capabilities address inefficiencies in drug development and commercialization while offering tangible return on investment across research and development, medical affairs and market access. These platforms create value without direct reimbursement exposure, support multiple use cases, and drive long-term stickiness across client organizations. Real-world evidence is already institutionalized inside pharma, with 87% of top pharmaceutical companies now maintaining dedicated RWE departments, highlighting how central these capabilities have become.
That internal prioritization is now showing up externally as well. Our conversations with industry participants over the past few weeks suggest RWE and trial enablement are increasingly viewed as horizontal infrastructure rather than niche analytics tools. Strategics we spoke with are seeking companies with proprietary datasets that support specific workflows for stakeholders across the drug life cycle.
TABLE 5: Research draws from diverse data sources, including real-world data generated by health system operations
Real-world evidence is derived by curating, standardizing and analyzing real-world data to produce high-quality, reliable insights from diverse, complex sources. It can inform every phase of treatment discovery and development. However, thus far, it has been used most often to inform the questions and data in early development decisions and postmarketing safety surveillance or comparative effectiveness studies. By contrast, clinical development and review have tended to use idealized, tightly controlled data sources for efficacy trials.
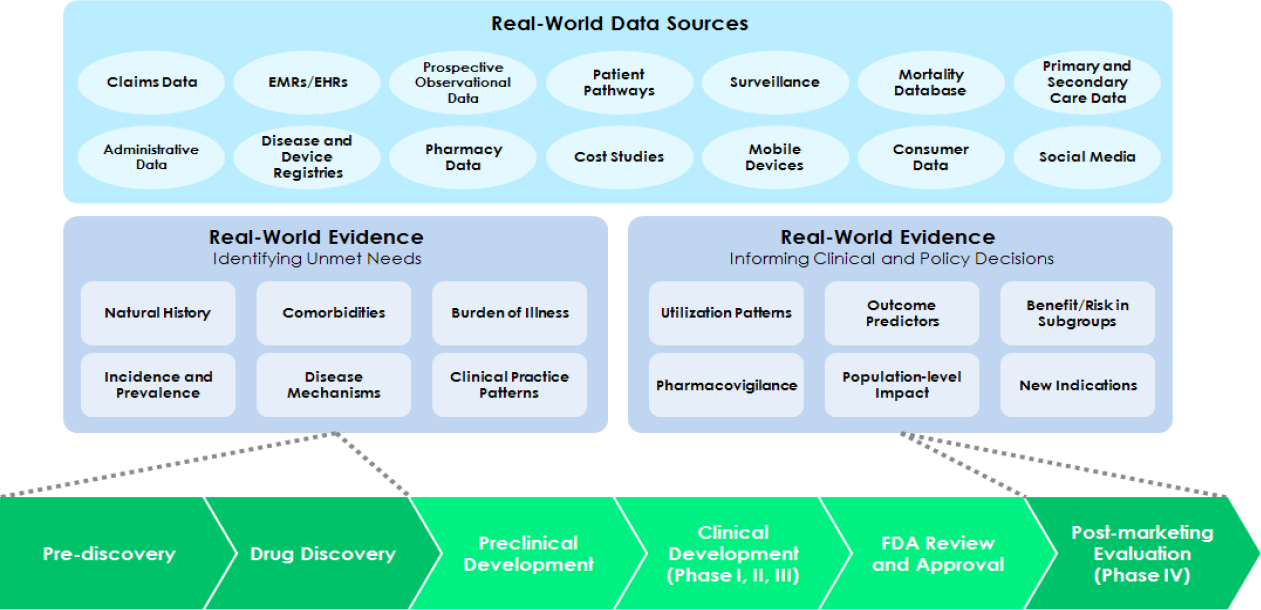
Source: National Academy of Medicine.
Even more broadly, regulators and payers are placing greater emphasis on RWE to support label expansions, post-market surveillance and value assessments, pushing pharma companies to invest earlier in evidence generation outside traditional trials. At the same time, advances in data integration and AI have made it easier to translate fragmented clinical and claims data into actionable insights, further increasing the strategic value of RWE and trial enablement platforms in 2026.
5. Operational discipline and compliance demand accelerating
Pharma buyers are increasingly treating operational rigor, auditability and compliance readiness as core product features rather than downstream services or optional add-ons. Platforms that can natively support inspection readiness, standardized documentation, clear audit trails and traceable decision making are seen as significantly lower risk and easier to scale across the enterprise. As we outlined in our prior work on regulation and risk driving quality management system modernization, regulatory frameworks are converging around shared expectations for validated systems, risk-based governance and digital traceability, raising the baseline for compliance as the industry heads into 2026.
This shift is reshaping vendor expectations and product roadmaps. Buyers want systems designed around how pharma organizations actually operate, including formal review cycles, role-based permissions and clear accountability structures that align with inspector expectations under frameworks such as 21 CFR Part 11 and Annex 11. Quality and operational leaders are increasingly wary of tools that rely on patchwork compliance layers or extensive customization, particularly as inspections become more data-driven and continuous. Products built with validation, life cycle governance and audit readiness embedded at the core are better positioned to expand across functions and geographies, while those that require procedural workarounds face longer sales cycles and limited enterprise penetration.
Looking ahead to 2026, several forces are reinforcing this shift. Regulatory scrutiny is expanding beyond manufacturers to include contract development and manufacturing organizations, data partners and digital vendors, increasing the need for shared, interoperable quality infrastructure across the ecosystem. At the same time, inspection models are moving toward more continuous oversight, with regulators expecting real-time system access and consistently traceable documentation rather than periodic, manual audits. Investor and partner diligence is also placing greater weight on operational maturity, making quality systems a visible proxy for execution risk. Together, these dynamics are pushing compliance out of the cost-center bucket and into the role of strategic infrastructure that supports scalability, resilience and long-term confidence.
Reading the tea leaves for 2027 event conversations
The prospect for a disciplined and cautious reacceleration of transaction activity in pharma IT, combined with strong secular trends driving increased operational efficiency and optimized therapeutic development, suggest pharma IT companies face bright prospects in 2026. Our conversations at these two early 2026 industry forums support this perspective. We anticipate next year’s conversations will highlight substantial progress within these themes.
First Analysis Healthcare Technology Summit 2026: Life science & pharmacy private company snapshots

Acclinate is redefining diversity in clinical trials, building trust with underrepresented communities through its NOWINCLUDED platform. The system engages participants with culturally relevant education, events and storytelling, while its AI and machine learning engine, e-DICT, interprets real-world behavioral signals to optimize trial participation. This combination of community engagement and predictive analytics accelerates recruitment timelines, mitigates operational risk and helps ensure trials reflect the populations they aim to serve.

AdviNOW provides an intelligent assistant that supports patients’ healthcare interactions. From digital registration and self-scheduling to augmented reality exams and automated documentation, the platform streamlines workflows and captures structured patient data in electronic health records. With AdviNOW, practices can achieve increased throughput, reduce administrative burden and improve coding accuracy, freeing providers to focus on delivering high-quality care.
Areti Health offers a 24/7 AI Coordinator that manages patient outreach, pre-screening, scheduling and follow-up across multiple channels. The platform also reviews medical records to identify treatment eligibility, flag contraindications and inform care plans. By automating routine workflows while maintaining empathetic engagement, Areti Health enhances compliance, improves appointment conversions and eases operational pressure on clinical teams.

basys.ai uses agentic AI to transform prior authorization, utilization management and clinical auditing. Its platform accelerates clinical decision making while maintaining strict compliance standards, reducing the time and manual effort typically required. Providers and payers gain streamlined workflows that lower administrative burden without compromising accuracy or regulatory requirements.

BEKhealth offers the BEKplatform, an AI-driven solution for patient identification in clinical trials. By analyzing structured and unstructured electronic health record data, including physician notes, the platform identifies candidates eligible for protocols with high precision. Research teams can reduce manual chart reviews, improve feasibility studies, accelerate enrollment timelines and improve patient selection.

Dash Bio merges technology with bioanalytical expertise to deliver fast assay development and sample analysis. It helps automate, scale and deliver services including ELISA (enzyme-linked immunosorbent assay), MSD (meso scale discovery), liquid chromatography-mass spectrometry, and qPCR (quantitative polymerase chain reaction) with transparent timelines and pricing. This approach allows sponsors to access reliable bioanalytical results across all stages of drug development, accelerating decision making and time-to-market.

Delve Health combines digital patient-reported outcomes with real-time human concierge support to improve trial compliance and retention. The platform monitors participant engagement, addresses protocol deviations and supports multilingual communication. Sponsors and sites benefit from integrated digital tools and personalized human outreach, improving diary completion and study retention rates.

Drug Intelligence provides prescribing analytics and market intelligence for specialty pharmaceuticals. Its platform delivers patient- and treatment-level insights, contextual analysis and consulting expertise. With these tools, biopharma teams can gain a deeper understanding of market trends, optimize brand strategy and make informed clinical and commercial decisions.

Everest by LOG10 accelerates document creation for life science teams, transforming regulatory, clinical and commercial deliverables with AI-driven workflows. With Everest, protocols, submissions, study reports and market strategy documents can be generated quickly. Contract research organizations and consultancies can also white-label Everest to scale their expertise into AI-powered software.

Genomenon turns complex genomic literature into actionable insights for diagnostics and precision medicine. Its platforms like Mastermind and Cancer Knowledgebase organize millions of variants, supporting accurate interpretation of germline and somatic mutations and helping clinicians and researchers gain confidence in diagnoses, therapeutic development and real-world evidence generation.

Hawthorne Health brings clinical trials closer to patients by leveraging community-based research sites and home visit solutions. Its network of experienced investigators and their therapeutic expertise enable faster enrollment, improved retention and broader trial participant representation. This approach can expand patient access to trials while generating high-quality, patient-centered data.

HumanTrue translates complex trial protocols into structured content and actionable workflows. Its Study Assistant and automation tools help sponsors, contract research organizations and sites understand protocols quickly, generate compliant documents and integrate data into existing systems. The platform can reduce amendments, enhance cross-team alignment and accelerate study start-up.

InsightRX uses model-informed precision dosing software to individualize therapy in clinical trials and patient care. Bayesian modeling, pharmacokinetic and pharmacodynamic analytics, and real-world data combine to optimize dosing. The platform reduces adverse events and improves efficacy, helping clinicians make informed, data-driven decisions at the point of care.

Keona Health provides an AI-powered patient access platform that automates calls, scheduling and triage. Routine tasks are handled automatically, while complex interactions guide staff in real time. By centralizing patient touchpoints, Keona Health reduces errors, improves efficiency, and helps patients access care smoothly.

Kivo Health delivers a virtual pulmonary rehabilitation platform for patients with chronic obstructive pulmonary disease. Its personalized at-home kits, one-on-one coaching and small group programs support symptom management, increased activity and overall wellness. By integrating clinical guidance, exercises, and ongoing support, it encourages patient treatment adherence and enhances long-term outcomes.

Latica offers a secure, unified real-world data platform that integrates structured and unstructured sources such as lab results, imaging and clinical notes. It helps life science and healthcare teams collaborate, analyze data and accelerate AI development and research insights. This environment breaks down data silos while maintaining privacy and compliance.

LighthouseAI simplifies state licensing and regulatory compliance for pharmaceutical supply chains. Its platform centralizes license tracking, monitors renewals and provides alerts for manufacturers, distributors and third-party logistics providers. Its expert support helps ensure filings, inspections and Drug Enforcement Adminstration compliance are managed accurately, reducing risk across all jurisdictions.

MassiveBio connects cancer patients to appropriate clinical trials. Its AI platform analyzes hundreds of medical data points and combines insights with physician expertise to provide personalized trial recommendations. The approach streamlines pre-screening, matching and enrollment while supporting a seamless, compliant and patient-centered experience.

MedaSystems helps life science teams manage expanded access and compassionate use programs efficiently. Its cloud platform centralizes provider requests, supports regulatory compliance and captures real-world data. The system enables collaboration across teams and accelerates patient access to investigational treatments.

Narrativa Navigator automates creating complex regulatory and clinical documents using AI. The platform generates study reports, patient narratives and other high-volume content quickly and accurately while ensuring compliance. The platform helps life science teams move from data to submission faster, maintaining consistency and quality across documentation.

Navidence turns real-world health data into actionable evidence using computable operational definitions. Its platform enables precise study design, consistent data queries and fit-for-purpose assessments, improving researchers’ confidence in their data and helping them gain insights faster and make more informed decisions in healthcare and clinical trials.

Nexus Healthcare uses human digital twin technology to create predictive, model-based evidence for trials, surgical planning and personalized wellness. Simulated patient populations help teams reduce development risk, optimize outcomes and speed regulatory and payer decisions. By combining real-world data with simulations, Nexus supports safer and more-personalized healthcare innovation.

NyquistAI is an intelligence platform for life sciences that automates research and insight generation based on global clinical, regulatory and market data. By leveraging over a billion curated data points, the platform supports strategic decision making, reduces time to market and provides intelligence to medical technology and pharmaceutical teams.

Octozi automates clinical trial data workflows, including data cleaning, review and reconciliation and safety monitoring. Its custom AI models help reduce errors, accelerate database lock and improve oversight across study teams. It can improve sponsors and contract research organizations’ confidence in trial data while lowering costs and shortening timelines.

OmniScience, through its Vivo platform, acts as a control tower for clinical trial operations. It unifies data from trial sites, labs, endpoints and patient-reported outcomes to deliver explainable, real-time insights. It helps teams detect anomalies, forecast enrollment and mitigate risks, enabling faster, cost-efficient decisions across trial portfolios.

Picture Health leverages AI to extract insights from radiology and pathology images for precision oncology. Its Px platform integrates imaging, genomic and clinical data to characterize tumors and their microenvironments. By turning routine imaging into predictive biomarkers, Picture Health helps clinicians make more informed treatment decisions without additional tests.

Pienomial’s Knolens platform unifies clinical, regulatory and market data for life science teams, delivering real-time insights. Its scenario-based simulations, predictive analytics and regulatory-grade evidence support faster decisions across clinical, health economics and outcomes research, market access, regulatory, and strategy teams. The platform helps organizations optimize trial design, regulatory submissions and competitive positioning.

Prognosis Technologies provides cloud-based tools to simplify clinical trial planning and supply management. It helps teams forecast demand, manage inventory, plan cohorts and analyze costs more quickly. The platform supports confident, data-driven operational decisions while reducing complexity and saving time.

Redox offers a healthcare interoperability platform that connects providers, payers, electronic health records and digital health tools through a standardized application programming interface. Its real-time data exchange capabilities enable analytics and AI workflows and help generate operational insights. By providing secure, scalable connectivity, Redox reduces friction and accelerates digital transformation across the healthcare ecosystem.

ScienceMedia delivers a digital platform for clinical training that uses its MindFlow methodology to ensure protocol compliance and scientific competency. Life science sponsors, sites and trial participants access multimodal, evidence-based courses tailored to therapeutic areas. The system improves enrollment, retention and study execution while maintaining high-quality, science-driven training.

Sorcero uses AI to convert fragmented medical and scientific data into actionable insights for life science companies. The platform accelerates medical affairs, content generation, literature monitoring and safety reporting. Sorcero’s medically tuned AI supports faster analysis, regulatory compliance and strategic decision making for better patient outcomes.

Trialynx provides a digital platform to design, document and launch trials efficiently. Its AI-powered document generation, real-time collaboration and data-driven study design capabilities help improve operational feasibility and compliance, leading to fewer trial revisions, reduced institutional review board resubmissions and smoother trial execution.

Univo IRB simplifies institutional review board oversight while prioritizing patient safety and advocacy. Its OneVerse platform centralizes submissions, approvals and regulatory documentation for Phase I to IV biomedical, biotech, medical device and noninterventional studies. It helps sponsors and sites improve efficiency, start studies faster and manage institutional review board workflows.

VIDA accelerates clinical trial imaging with AI-powered digital biomarkers, enabling sponsors to collect, analyze and leverage high-quality data across multiple therapeutic areas. The VIDA Intelligence Platform provides cloud-based access to imaging from over 1,500 sites worldwide. By delivering precise quantitative endpoints and streamlined trial imaging management, VIDA reduces development costs and improves trial efficiency. First Analysis venture capital funds have invested in VIDA.
Videra Health provides an AI-driven platform to automate patient engagement, documentation and monitoring in behavioral health clinics. Its system streamlines workflows, enhances risk identification and increases clinician capacity without adding staff. Videra helps improve patient outcomes and reduce crisis-driven interventions and supports secure, compliant care.

VuuAI helps pharma and medical technology teams efficiently create, validate and optimize content for commercial and medical engagement. Its AI-driven automation accelerates medical, legal and regulatory review, helps protect brand consistency and improves the impact of scientific communications. By integrating with existing asset management systems, it helps teams scale content production, reduce costs and deliver more effective messaging.

Yunu is an end-to-end clinical trial imaging platform that centralizes workflows, data and compliance from initial scans to trial submission. The platform automates protocol-guided assessments, reduces manual tasks by up to 80% and provides real-time oversight. With Yunu, sponsors, conract research organizations and cancer centers can benefit from faster turn-around times, improved accuracy and better collaboration across all trial stakeholders. First Analysis venture capital funds have invested in Yunu.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
