White paper: Going beyond the pill
Digital solutions driving pharma commercialization and improving patient outcomes

Economic and regulatory pressures, pricing scrutiny, new industry stakeholders, and the explosion of real-world data have caused life sciences companies to rethink their commercialization strategies.
By leveraging data-driven digital technologies, life sciences companies can generate the knowledge and insights needed to improve commercialization and patient outcomes.
In this report, we explore the dynamic and rapidly evolving array of digital technology solutions related to the drug commercialization process, highlighting numerous promising opportunities in this burgeoning emerging sector
TABLE OF CONTENTS
Includes profiles of 134 public and private companies
- Data is the new gold
- Pharma commercialization: Changing the way drugs are marketed
- Transforming the legacy commercialization model
- The emergence of the CRM: Improving sales-force effectiveness
- Going beyond the CRM: Unifying databases through master data management systems
- Storing the data: Challenges pharma faces with data integration
- Data sources: Which data is useful and which data is acceptable for regulatory approval
- Analytics: Harnessing the power of real-world data to lower costs, recruit patients
- Emerging digital channels: Leveraging prescription data through virtual reality education
- Background on KOLs: How key opinion leaders are used to validate drugs
- Digital KOLs: Emergence of digital platforms to promote research and new products
- Patient centricity: Understanding the patient experience to improve outcomes
- Direct-to-consumer advertising: Another way for pharma to reach physicians
- Point-of-care advertising: Another way for pharma to reach patients
- mHealth: Using mobile and wireless devices to improve health outcomes
- Digital therapeutics: Software that aids in diagnosis, treatment, and coordination of care
- Medication adherence & compliance: Software that engages with patients to increase compliance
- Patient communities & social media: Where patients can share information and receive advice
- Hub services: Where patients and HCPs go for benefit verification and prior authorization of specialty drugs
- Engaging payers: The emergence of outcomes-based payment models
- Featured company profiles
Includes discussion and profiles of the following companies: 23andMe, AB Cube, ActiGraph, ACTO, Advera Health Analytic, AiCure, Aktana, AliveCor, AllazoHealth, Amazon, Analytical Wizards, Ancestry, Anju Software, Apple, ArcheMedX, ArisGlobal, Arpeggio Biosciences, Asembia, Atomwise, Auransa, Axtria, Backpack Health, BenevolentAI, BERG, Biodesix, Cardinal Health, CareMetx, Click Therapeutics, closerlook, ConnectiveRx, CoPilot, Cota, Covance, Crossix (Veeva), Dassault Systèmes, Datavant, Decision Resources Group, Deep 6 AI, Doximity, Eularis, Evidation Health, Exscientia, Facebook, Fitbit, Genomenon, Genpact, GNS Healthcare, Google, Health Union, HealthEconomics.com, HealthLink Dimensions, HealthPrize, Hexi.ai, Human API, IBM, ICON, ImmersiveTouch, Innoplexus, Inovalon, Insilico Medicine, Inspire, IntegriChain, inVibe Labs, IQVIA, KloudScript, Komodo Health, Koneska Health, Level Ex, Life Image, Liquid Grids, McKesson, Medidata, Medisafe, MedPro Systems, Mendel.ai, MMIT, Mu Sigma, MyMeds&Me, NeoGenomics, Numerate, OM1, Omada Health, Omron, OptimizeRx, Oracle, Orig3n, Outcome Health, Owkin, Oxford Nanopore, Parexel, PatientPoint, PatientsLikeMe, Pear Therapeutics, Phenomic AI, Phreesia, PhysIQ, PPD, PRA Health Sciences, Prescriber360, Prognos, QPharma, Qrativ, Reltio, ResMed, ReviveMed, rMark Bio, Roche, RxS, Saama, Scientist.com, Sermo, SHYFT Analytics, Skipta, StayinFront, Surescripts, Swoop, Syapse, Syneos Health, Synergistix, TARGET PharmaSolutions, Tempus, TikaMobile, TrialCard, TriNetX, trUStr, Twitter, TwoXAR, UnitedHealth, Validic, Veeva, VIDA, WEGO Health, XtalPi, ZS Associates
Data is the new gold
The life sciences industry is experiencing a great transformation in customer and market dynamics. The emergence of specialty segments, increased regulatory constraints, the focus on subgroups in various therapeutic areas, and a recent explosion of information have all added to the complexity of the industry, while drug pricing regularly makes the headlines, with patient advocacy organizations, healthcare providers (HCPs), and politicians weighing in on the discussion. With years of painstaking R&D and new drugs costing pharmaceutical companies hundreds of millions to billions of dollars, the entire business model of pharmaceutical companies hinges on their ability to monetize the drugs they develop and bring to market. The increased focus on cost from stakeholders has prompted the life sciences industry to demonstrate value with supporting data.
TABLE 1: Sources of real-world data
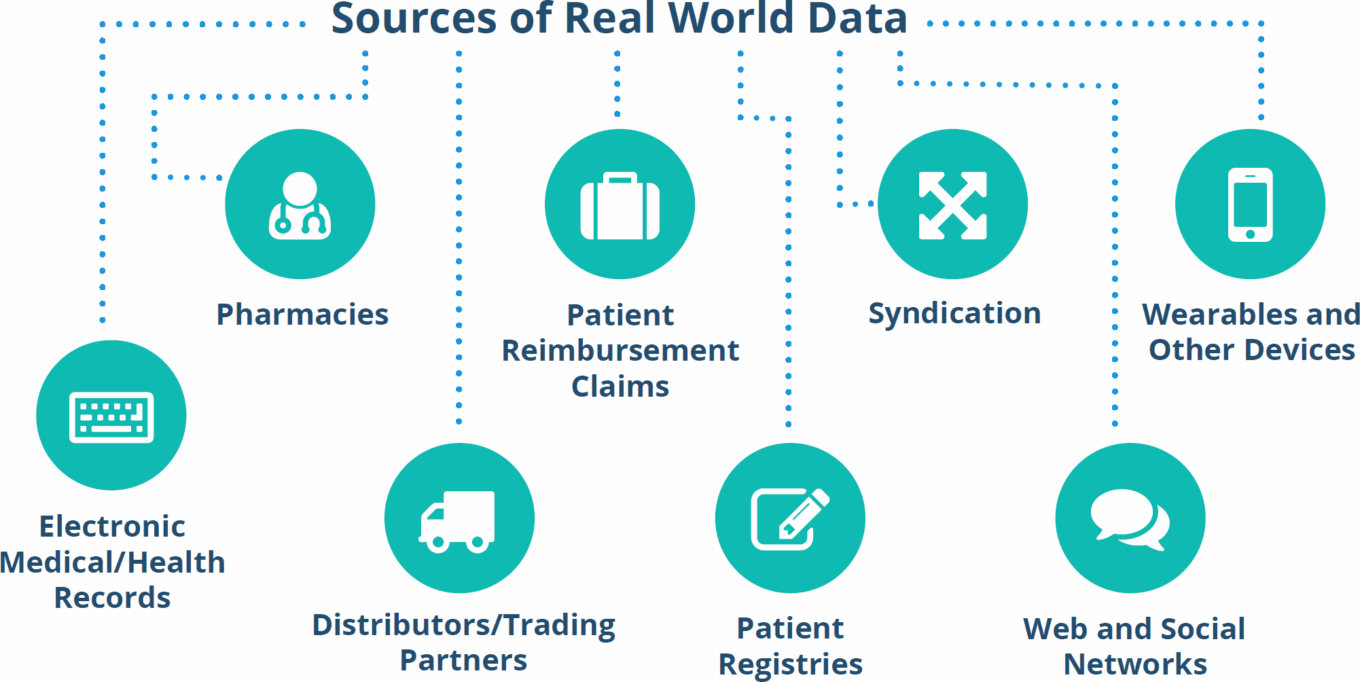
Source: Saama Technologies.
In one form or another, the topic of data has rippled through the life sciences industry for years. At first, it was the term ‘big data’ that attracted the attention of industry executives. Now, real-world data (RWD) and real-world evidence (RWE) are front and center. While RWD and RWE are often used interchangeably, the U.S. Food and Drug Administration (FDA) provides a distinction. It defines RWD as data collected from sources outside of traditional clinical trials. RWE is generated through the analysis and/or synthesis of RWD to identify the effects of health interventions, such as benefits, risks, or resource uses that are not collected during a clinical trial. What unites these concepts is digital technologies. Digital technologies provide the ability to rapidly and easily collect, store, analyze, and transmit data on a large scale, unlocking huge potential in drug discovery, clinical development, commercialization, value-based care, and patient safety. Drug launch is no longer the finish line, but rather the beginning of the race.
TABLE 2: Prescriptions dispensed: 2014-2018
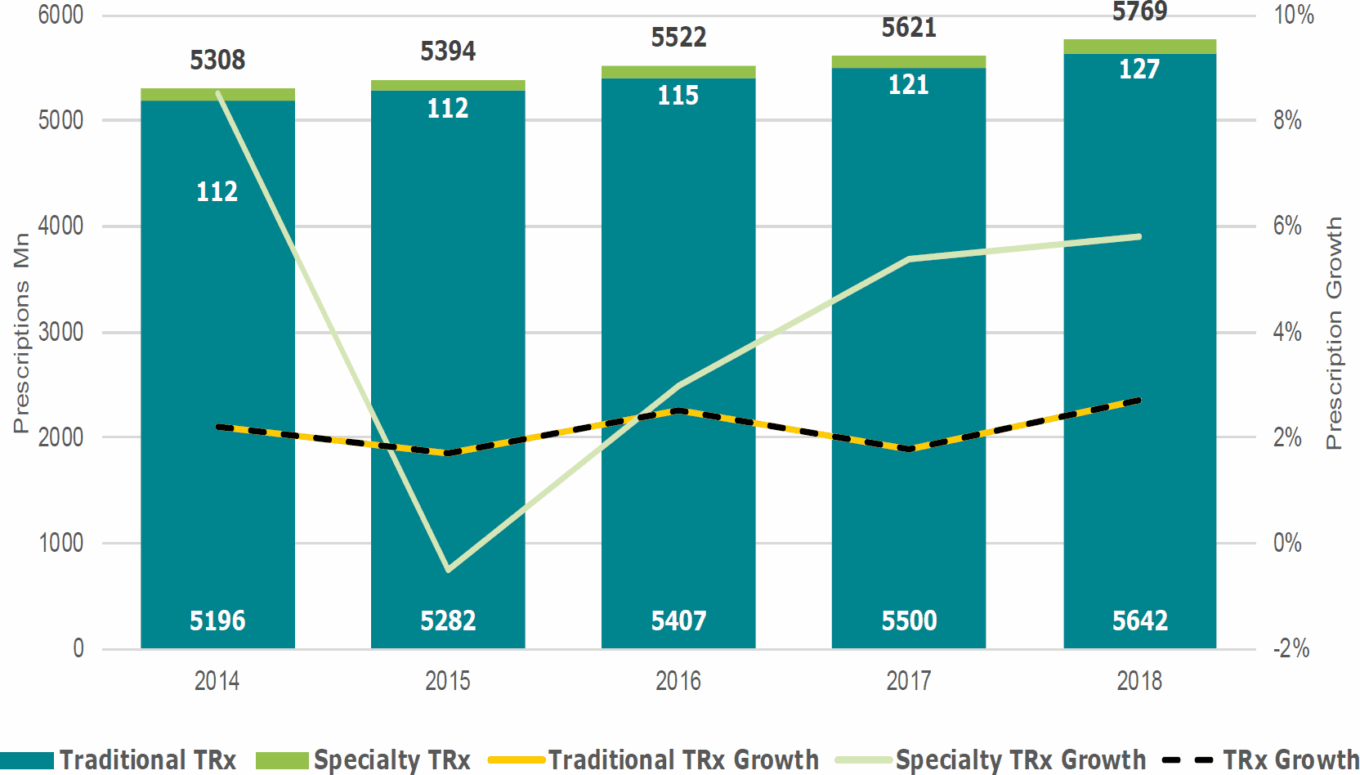
Source: IQVIA Institute for Human Data Science.
The life sciences industry is swimming in data, from internally generated clinical data to outsourced vendor-provided prescription data. This is partly the result of new digital technologies such as wearables, tablet computers used for engagement, and medical devices with built-in features that facilitate data collection, patient-reported outcomes data, sales and marketing data, and other kinds of information. It’s also the result of digital platforms such as electronic medical records (EMRs) and CRM (customer relationship management) software that make the collection and analysis of data possible. As the hardware and software to manage large data sets continue to evolve, so too have the sources of data grown. Privately held databases, as well as public data provided by government agencies such as the Centers for Medicare & Medicaid Services (CMS), have become essential components of the clinical development and commercialization process. If data is the new gold and pharma companies are the new miners, then data collection/management tools and analytics are the new picks and shovels.
Pharma commercialization
The U.S. healthcare system is the costliest in the world, with national health expenditures accounting for 18% of U.S. GDP, or $3.9 trillion. According to the CMS Office of the Actuary, national health spending is projected to outpace GDP growth from 2018-2027 by almost a full percentage point at an average rate of 5.5% per year and reach $6.0 trillion by 2027, driven by aging populations, improved and earlier disease diagnosis, insurance coverage expansion, and an increasing shift toward unhealthy lifestyles.
TABLE 3: Pharma R&D returns
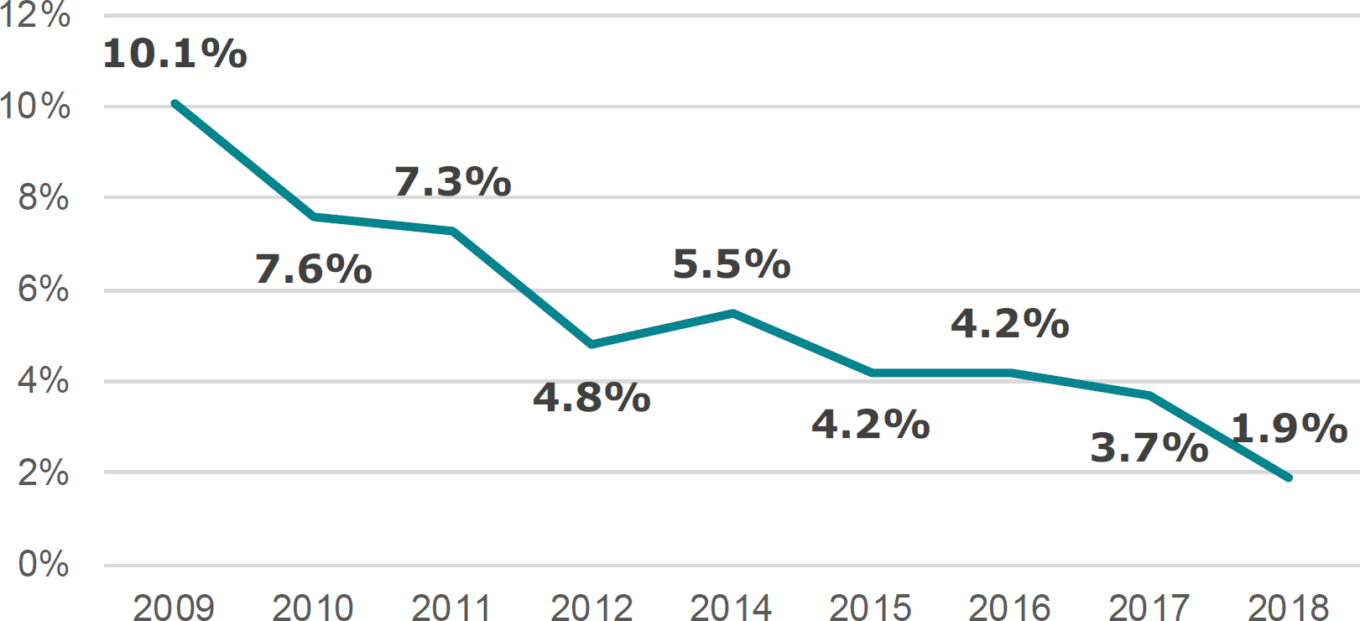
Source: Deep Knowledge Analytics.
As a result, the pharmaceutical industry is well-positioned to be a prime beneficiary, with total prescription drug spending in the United States expected to grow 6% annually from 2020-2027 and reach $780 billion (from $453 billion in 2017). The biggest driver of this growth is expected to be the launch of new drugs, as R&D efforts across the pharmaceutical industry result in new drug approvals and uptake.
Despite the attractive industry tailwinds, new marketplace dynamics are changing the way drugs are marketed, prescribed, and paid for. One important factor influencing the changing healthcare landscape is the Affordable Care Act of 2010, which continues to impact the U.S. healthcare system through expanded insurance coverage, reform of healthcare delivery and payment systems, and new measures that transfer more responsibility for cost and quality from payers to healthcare providers, with a renewed focus on value.
As a result, new stakeholders are increasingly gaining influence in the purchasing process as the definition of “customer” has evolved from the prescriber to include payers and providers. Individual HCPs still make key therapeutic decisions, but they do so in an industry where payers – including patients, integrated delivery networks (IDNs), accountable care organizations (ACOs), pharmacy benefits managers (PBMs), and the government – are exerting more influence. In addition, data and analytics are making new insights possible to every stakeholder within healthcare, including providers, patients, and payers.
TABLE 4: Drug launch timeline
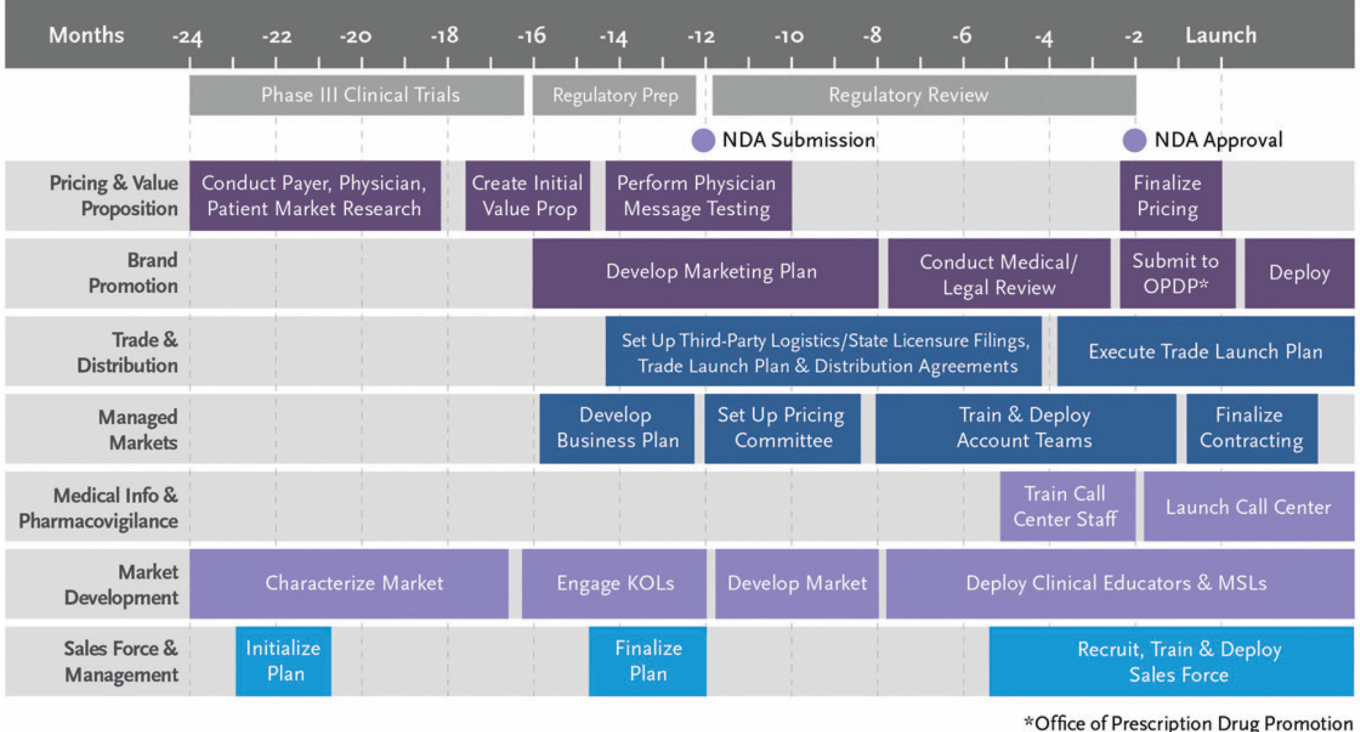
Source: IQVIA.
On top of these changes, pharma companies are faced with competitive pricing pressure from generics, patent expiration, and rising regulatory burdens. These issues have caused pharmaceutical companies to rethink their traditional commercialization models to identify ways to create value “beyond the pill.” This involves the adoption of digital technologies that drive HCP and patient engagement, as well as those that collect and extract outcomes data needed for value-based pricing.
TABLE 5: R&D by function, PhRMA member (companies 2018, $ in millions)
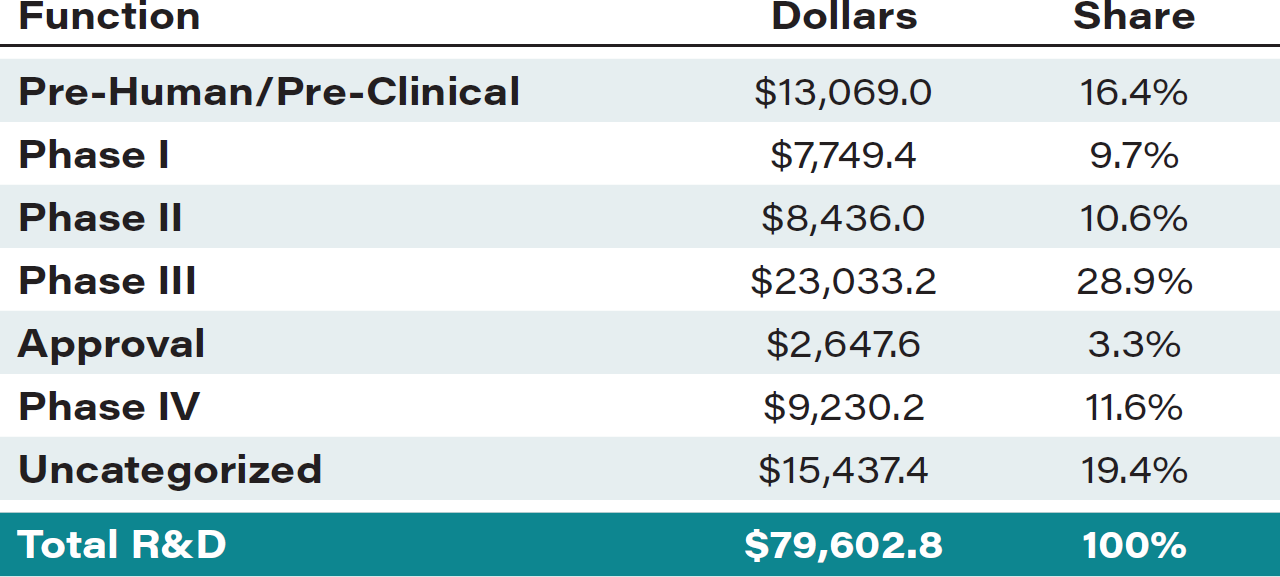
Source: Pharmaceutical Research and Manufacturers of America, PhRMA Annual Membership Survey, 2019.
Notes: All figures include company-financed R&D only. Total values may be affected by rounding.
As we discussed in a previous white paper, eClinical Solutions: Streamlining the introduction of new drugs and medical technology, the drug R&D process is inefficient, complex, bureaucratic, and, above all, expensive. Furthermore, the return on R&D expenditure has fallen from an industry average of approximately 20% in the late 1990s to just ~2% today. According to the Tufts Center for the Study of Drug Development, it takes an average of $2.6 billion, including cost of failures, and 10-15 years to discover, develop, and win approval for a new drug. Additionally, roughly only one of every 10 drugs that starts the trials process ends up being approved by the FDA.
Pharmaceutical companies that have spent a decade or more investing in drug development and clinical trials need to understand the product lifecycle in order to drive the greatest possible ROI before patent expiration and generic competition drive down pricing. Though drug patents in the United States last 20 years by law, pharmaceutical companies typically apply for them before clinical trials. Thus, the exclusivity of an on-the-market drug before generic varieties emerge varies. For some drugs, the commercial duration of the patent protection could be as short as seven years. As a result, a drug’s launch phase is the critical moment in a product’s lifecycle.
TABLE 6: Patent challenges by generic companies
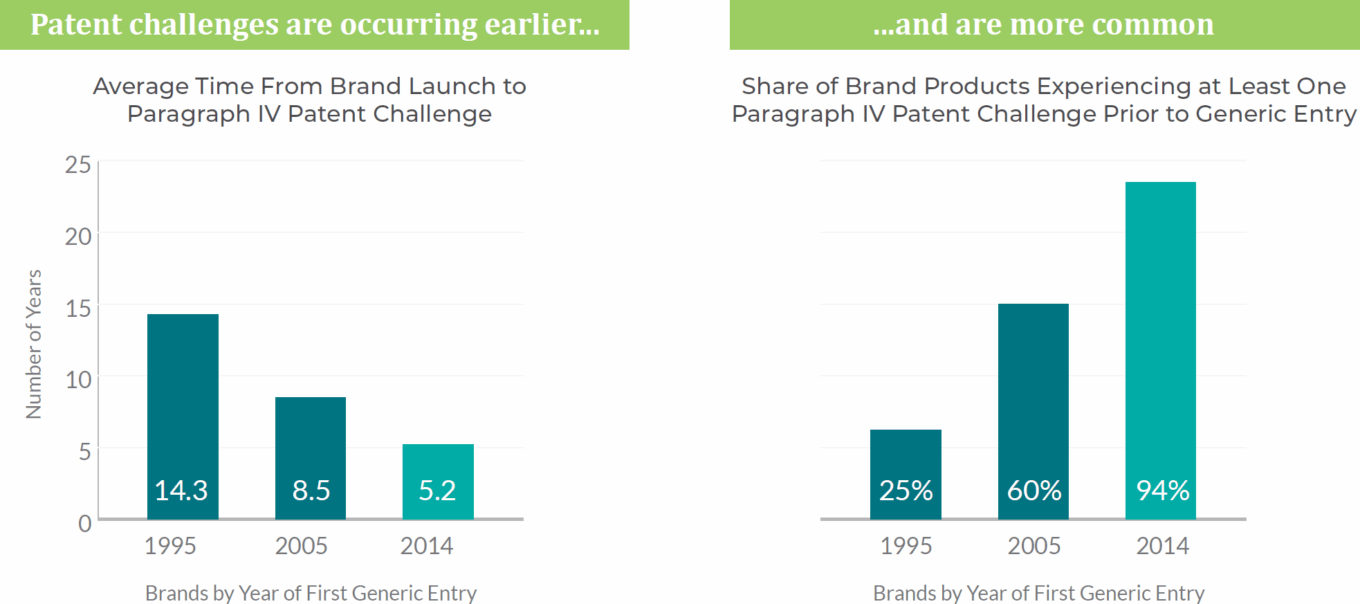
Source: PhRMA.
Drug launches face more intense competition today than a decade ago. The average window of time in which a drug remains on the market before competing products arrive is currently only 2-4 years, down from ~8 years in the 1990s. According to the Pharmaceutical Research and Manufacturers of America (PhRMA), competition from generics and biosimilars is expected to reduce brand sales in the U.S. by $105 billion from 2019 to 2023. Despite the potential headwinds from generics and biosimilars, new drug launches are expected to account for 40-50% of industry revenue over the next five years.
TABLE 7: Drug lifecycle

Source: PhRMA.
A new drug’s sales trajectory is set in the first six months of sales. Nearly 66% of all drug launches fail to meet prelaunch sales expectations for their first year on the market, and those that fall short typically continue to underdeliver for the next two years. In addition to being risky, launching a drug is also expensive. The average single-product pharma company spends >$125M in SG&A in the three years leading up to launch. When it comes to pharma companies launching novel first-in-class products, they spend even more, with three-year accumulated costs of ~$160M. Given the high costs and the complexity of the launch process, pharma companies need to have a robust plan in place to ensure they make informed and effective spending decisions when it comes time to commercialize their drug.
Transforming the legacy commercialization model
Despite the vast amount of capital pharma has invested in R&D and its pressing need to drive market adoption, most pharma commercialization teams are still running antiquated playbooks when it comes to one of the most challenging aspects of the overall pharma commercialization process, engaging HCPs.
TABLE 8: Parties involved in the prescription process (standard Rx)
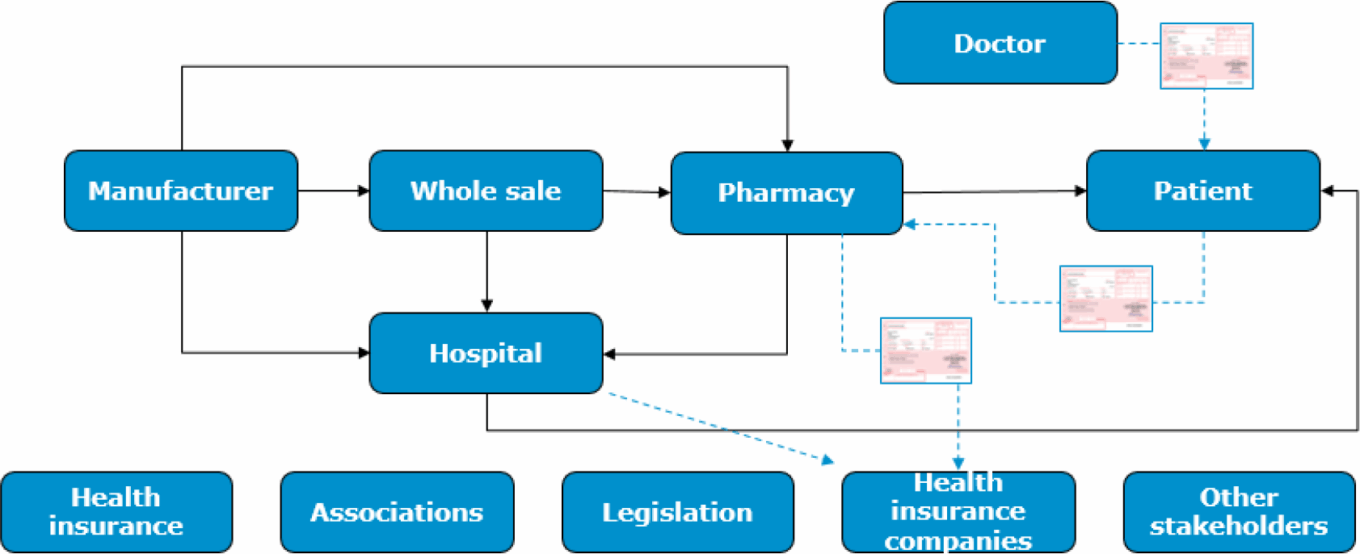
Source: Gregor Börner.
Historically, when launching a new drug, pharmaceutical companies relied heavily on teams of sales representatives for promotional activities, with commercial success largely dependent on coverage, frequency, selling skills, and HCP relationships. A medium-sized pharmaceutical company might have a sales force of 1,000 representatives, while a large pharma company might have tens of thousands of representatives globally.
Pharma has employed this model extensively since the 1960s. It relies on teams of sales reps who meet with HCPs, provide information on a drug, and offer free drug samples to convince HCPs to write prescriptions and eventually become advocates for a drug. Spending on product detailing to HCPs (typically face-to-face) was $5 billion in 2016, while spending on free drug samples was $13.5 billion.
TABLE 8: Accessible prescribers since 2008
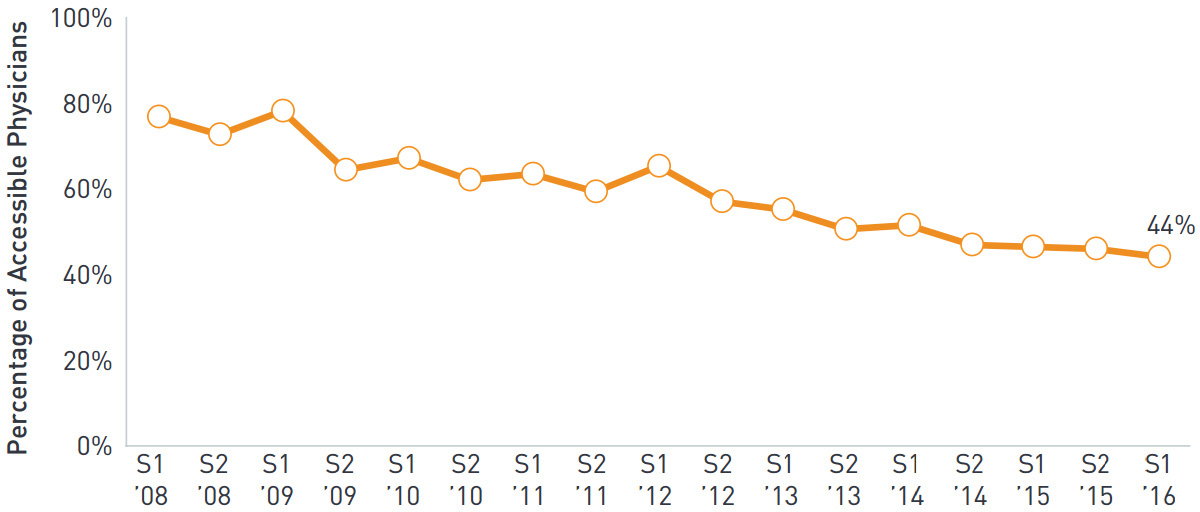
Source: ZS Associates AccessMonitor.
While sales representatives still play a vital role, they no longer dominate the landscape as changing market conditions, like practice consolidation, cost pressures, and new regulatory policies such as the Physician Payments Sunshine Act, have reduced their access to HCPs. Enacted as part of the Affordable Care Act signed into law in 2010 by President Obama to increase transparency around the financial relationships between healthcare providers and drug manufacturers, the Sunshine Act requires manufacturers of drugs, medical devices, and biological and medical supplies covered by the three federal healthcare programs, Medicare, Medicaid, and the State Children’s Health Insurance Program (SCHIP), to collect, track, and report payments and transfers of value they make to “covered recipients,” currently defined as physicians and teaching hospitals. These transfers of value include items such as consulting fees, honoraria for speaking events, and research grants.
In October 2018, President Trump signed the Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act (SUPPORT Act). The SUPPORT Act expands the definition of a “covered recipient” to include physician assistants, nurse practitioners, clinical nurse specialists, certified registered nurse anesthetists, and certified nurse midwives.
Furthermore, many hospitals and physician networks are limiting sales rep access to HCPs in an effort to curb the influence of these reps on prescribing habits. Simultaneously, hospitals and physician networks are challenging HCPs to make their practices more efficient and to increase the patient-to-physician ratio.
The end result has been a significant reduction in access granted to pharmaceutical sales reps over the course of the last decade. According to a recent ZS Associates report examining pharma’s interaction with health-care providers, the percentage of physicians classified as “rep-accessible” has dropped from 80% in 2008 to 44% in 2016. Specialty practices such as oncology have the most restricted access, with 27% being rep-accessible. Additionally, from a peak of 101,000 reps in 2005, the number of pharma reps in the United States has fallen to 65,000, representing $13 billion in annual costs to the industry.
TABLE 10: Breakdown of life sciences marketing budget
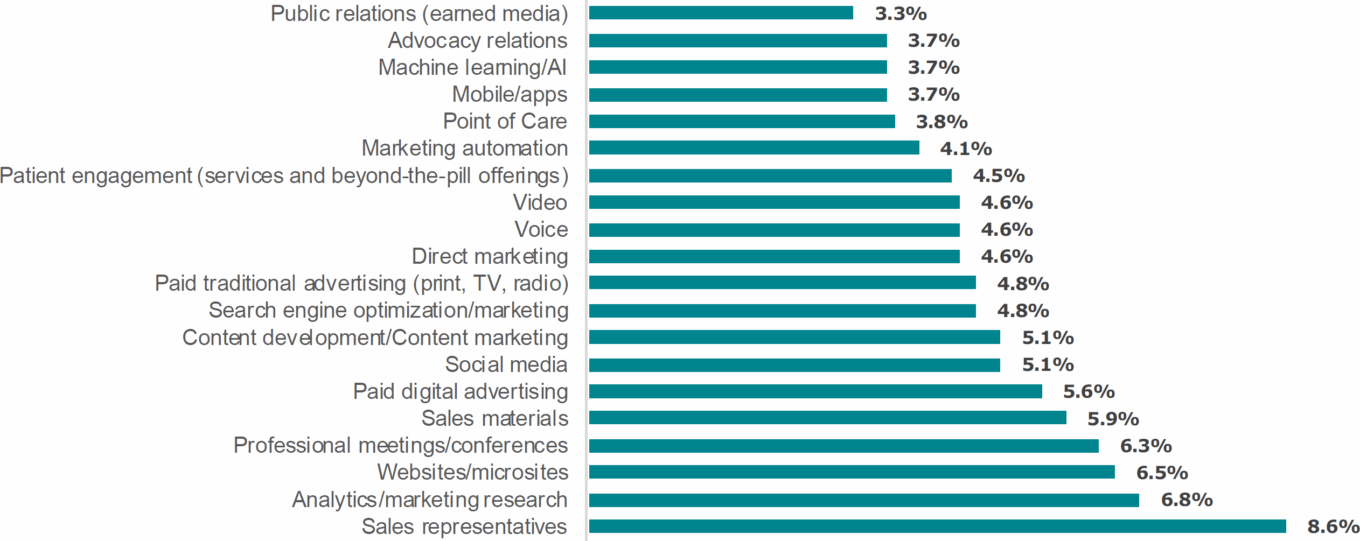
Source: MM&M/Deloitte Healthcare Marketers Trend Report 2019.
Even though doctors are spending less time with sales reps, that does not mean they aren’t interested in the information the reps are presenting to them. Many doctors are just choosing to get the information differently or in ways that are more accommodating to their very busy schedules. According to one industry executive we spoke with, 80% of physicians prefer getting drug information online instead of via sales rep visits. HCPs often get medication information via an expanding array of digital content, which can be found on pharmaceutical-branded websites, professional association web portals, newswires, social media, or blogs.
Historically, only younger tech-savvy HCPs would access medication information via the internet, but today, even older HCPs prefer to receive information digitally. HCPs are spending an average of 84 hours a year on reading digital marketing material. According to a recent study from Accenture, 25% of sales-force interactions have been replaced by digital materials, which, in our view, has resulted in a transformation of the sales rep’s role to that of a curator of content and communication strategies. As a result, pharma companies want to be able to measure how HCPs interact with different digital channels and then use those insights to inform sales reps for their next face-to-face call, personalize web portals and emails, and enhance mobile education.
TABLE 11: Interactions between U.S. HCPs and drug companies
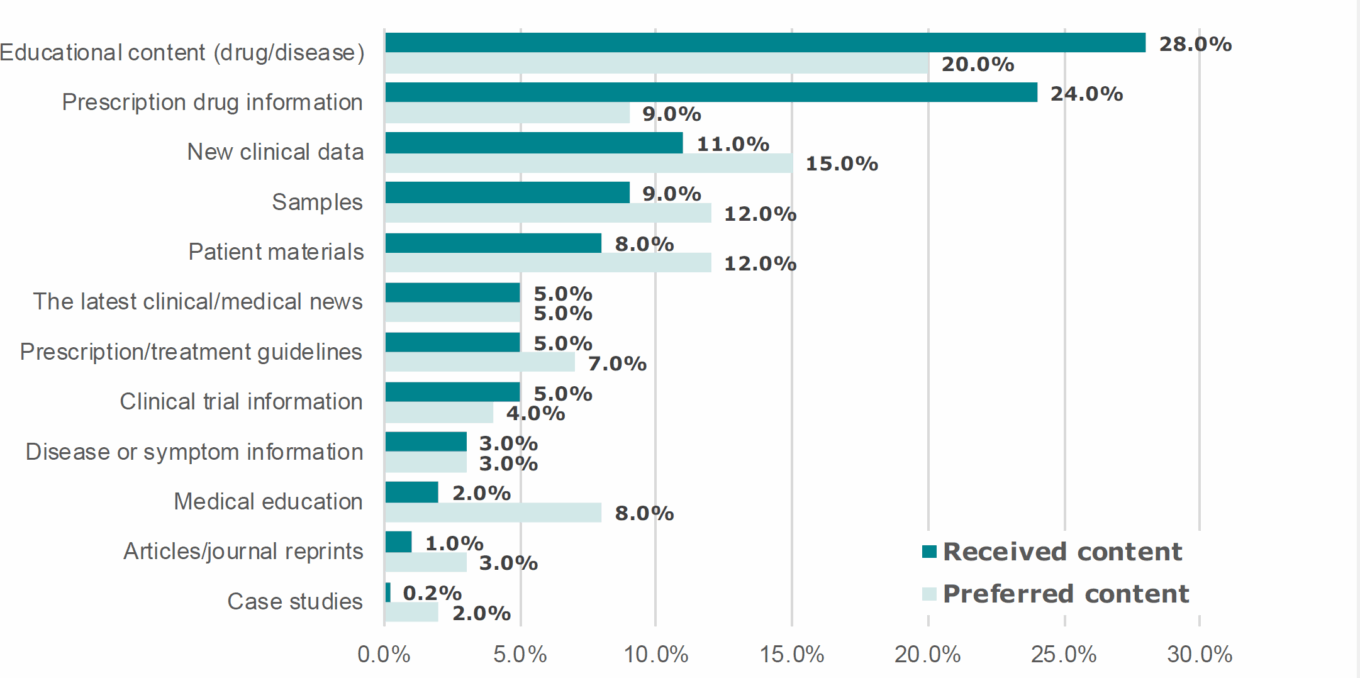
Source: DT Associates/Aptus Health.
That being said, engaging HCPs isn’t as easy as sending them a Google ad or emailing blast messages to every physician who seems likely to prescribe a drug. According to industry statistics, branded email sent to HCPs has a 5%-8% open rate and 4%-6% click-through rate. Mass messaging is costly and inefficient and often results in already busy HCPs getting annoyed and disinterested. According to research from closerlook, 72% of HCPs report poor quality and low relevance as the reasons for opting out of email. In our view, pharma companies need to complement the traditional sales model with digital tools and multi-channel digital strategies that deliver useful and relevant personalized content to HCPs when and how they want it.
The emergence of the CRM
While RWD and advanced data analytics in pharma companies have been used primarily to drive insights in clinical research and trial management (i.e., trial design, patient recruitment, biomarkers), they can be equally impactful in commercial applications. The most common commercial application is to improve sales-force effectiveness using CRM systems. A CRM system is a computer-based application – whether installed on client computers, third-party hosted, or available through Software-as-a-Service (SaaS) – that helps a sales force gather and organize information about its clients to facilitate or improve relationships and interactions with each of those clients. Typically, a sales rep interacts with the CRM system and schedules a call with the HCP. These discussions are captured in the form of call notes and are updated in the CRM system.
A well-designed pharma CRM system will highlight requests for samples or materials, capture questions or actions requested from HCPs, and provide insights on specific practices so customized solutions can be crafted. Implementing a CRM platform requires a significant financial investment and time commitment to set up and maintain. Despite this, the pharma CRM software market has experienced continued growth over the past few years due to the emergence and maturation of cloud technology and new regulations affecting pharma’s traditional go-to-market sales and marketing operations.
Almost every pharma company has implemented a CRM solution. In fact, many companies are moving on to second- and third-generation technology as cloud-based software is now preferred by most sales and marketing organizations, which has led to wide-scale upgrading and replacement of older CRM implementations over the past few years. Most pharma CRM systems are designed with enhanced features designed to work on a variety of mobile technologies, such as tablets and cell phones. However, the user experience and system capabilities of many of the CRM solutions available today significantly differ. Veeva, for example, offers a multichannel CRM solution that includes not only call notes but also approved email, event management, and online meetings. Founded in 2007 by ex-Salesforce.com early-stage employee Peter Gassner and ex-Siebel life sciences employee Matt Wallach, Veeva is currently the global leader in life science CRM market share.
Veeva’s CRM software uses multi-tenant SaaS architecture built on top of the Salesforce.com platform, which allows Veeva to serve multiple customers from a single shared instance of the software application. In a nutshell, only one version of the application is deployed to all customers who share a single common infrastructure and code base that is centrally maintained. Adoption of the Veeva CRM solution has been rapid, especially since the launch of its iPad app in 2011. Currently, we estimate Veeva has 75% share of the global life-sciences CRM market with over 300,000 reps (300+ CRM customers) using the application. Other notable competitors include Synergistix’s CATS, Stayin-Front EdgeRx, Oracle’s Siebel, QPharma’s Titanium, TikaMobile, and IQVIA’s Orchestrated Customer Engagement (OCE) platform, launched in 2017, which has ~30,000 rep users.
Going beyond the CRM
CRM adoption has been revolutionary for the pharma industry, but it’s predominantly used as a backward-looking tool tracking transactional data rather than a forward-looking tool to enhance engagement. In the blockbuster drug era of the 1990s and 2000s, a CRM was considered the go-to commercialization tool with the sales rep at the center because salespeople were the most influential component of the commercialization process.
That is no longer the case, and in today’s environment, sharing insights gathered by different teams within the organization is a well-known and difficult problem across many pharmaceutical companies. For example, insights gained by sales and marketing teams may not always be shared with market access/ health economics and outcomes research (HEOR) and medical teams (and vice versa). Despite not being on the front lines directly engaging HCPs, each of these subgroups is gaining influence and garnering larger roles in shaping the commercialization process. Thus, companies must go beyond the CRM and adopt specific tools and applications backed by external sources of RWD that can break down the traditional pharma-siloed model.
As noted, the role of an HCP has changed dramatically, with payers now exerting their influence on stakeholders involved in purchasing decisions. Thus, it is imperative for pharma to understand the buying process and behavior of a number of different stakeholders in this complex marketplace. The problem is that the data needed to do so is often fragmented and/or scattered across different departments within the organization, making it challenging to manage and interpret both historical and new data. As a result, most pharmaceutical companies have turned to master data management (MDM) systems to unify what could be dozens of internal HCP databases, each of which they compile for a different purpose, such as targeting and segmentation, identifying key opinion leaders (KOLs), aggregate-spend reporting, corporate integrity agreement compliance, or market research; the information is related, but much of it resides in siloes with little cross-communication.
TABLE 12: Physician MDM profile
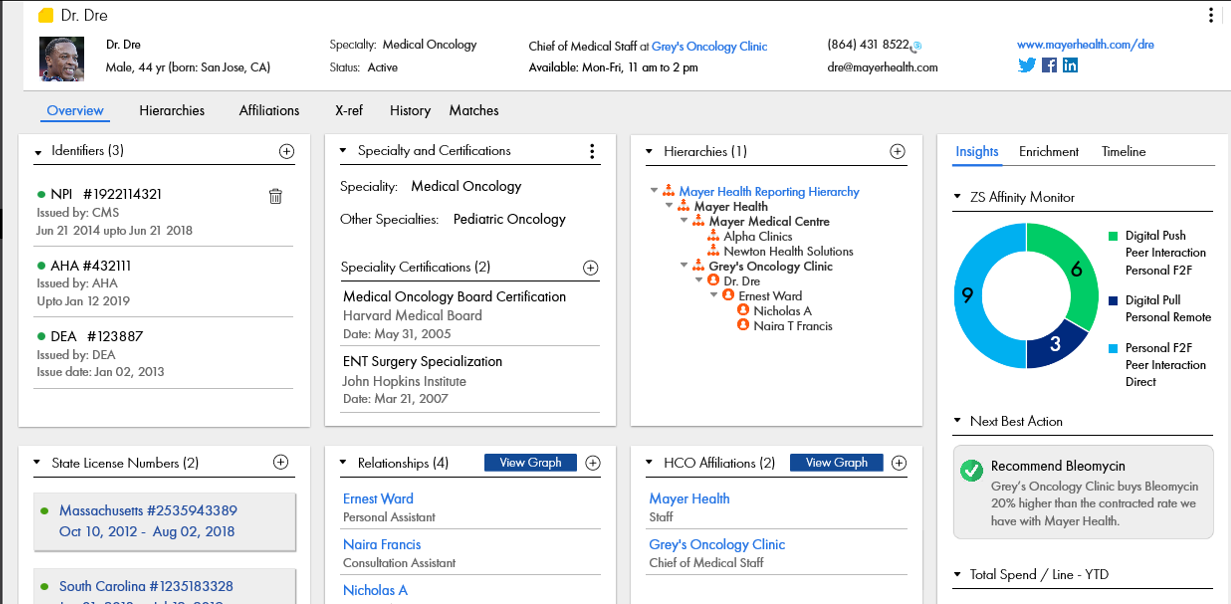
Source: Mayer Health.
The goal of an MDM is to create a single version of the truth or a “golden record” – a master file containing all the necessary and pertinent information about an HCP. An MDM aggregates data from various internal sources including a CRM and enriches it with third-party industry reference data such as physicians’ names, addresses, and contact information, affiliations (i.e., the health systems that HCPs are employed by or affiliated with and, in turn, the insurance providers those health systems use), a HCP’s National Provider Identifier (NPI), Drug Enforcement Administration (DEA) registration numbers, state medical licenses (a significant number of HCPs operate in more than one state), and certifications by medical specialty. The scrubbed updated data and relationships are then provided to users within the organization or sent downstream to a data warehouse.
In addition to matching HCPs with payments and “transfers of value” to them to help pharma comply with the Sunshine Act (in 2018, 1,582 companies made $9.4 billion in payments to 627,000 physicians and 1,180 teaching hospitals) as well as sample accountability and reconciliation to comply with the Prescription Drug Marketing Act (PDMA) and Ohio’s Terminal Distributor of Dangerous Drugs (TDDD) licensing, an MDM can provide pharma with a 360-degree view of the HCP that applications can leverage across the organization. A good MDM system will provide pharma commercial teams with up-to-date customer information to drive face-to-face or digital engagements so they can launch effective marketing campaigns and sales initiatives.
86% of the life sciences organization employees Veeva questioned in its 2018 Customer Reference Data Survey identified customer reference data as a key strategic asset integral to their sales operations. Proper vetting of customer reference data can act as a significant differentiator for campaign management, commercial activities, multichannel engagement, retention, and account-based selling. This requires pharma companies to maintain updated and relevant information about HCPs, healthcare organizations (HCOs) and their affiliation with IDNs, group purchasing organizations (GPOs), key decision-makers for purchases, and key influencers of buying decisions.
TABLE 13: LexisNexis provider data MasterFile
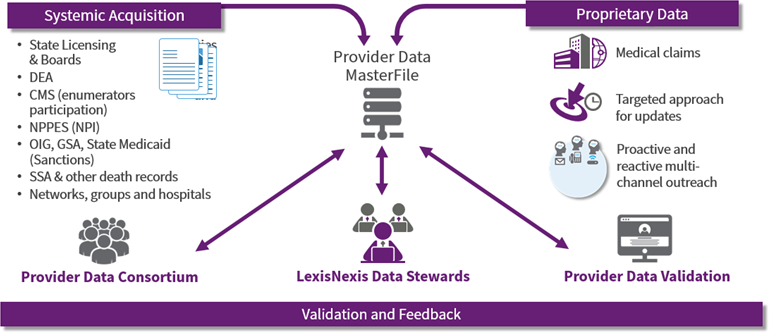
Source: LexisNexis.
We note there is a well-defined difference between syndicated reference data sold by vendors (i.e., the AMA’s Physician Masterfile, which includes current and historical data for more than 1.4 million U.S. physicians, residents, and medical students) and data stewardship/governance, which involves managing and curating how reference data is qualified and updated. Despite the difference, vendors in the space are promoting the idea of a one-stop shop for MDM and reference data. For example, IQVIA via IMS Health (prior to the 2016 merger with Quintiles) has its own reference data service. In 2014, IMS acquired both the OneKey global data service (9.7 million HCPs and greater than 708,000 HCOs) and assets from Cegedim, including its CRM tools and its SK&A Information Services. In 2015, it also acquired Healthcare Data Solutions, another reference data provider.
Veeva got its start solely as a CRM software provider. In 2013, it acquired a reference data provider, AdvantageMS, and it launched Customer Master in 2014. In 2015, it launched a reference-data subscription service, Veeva OpenData (16 million HCPs and HCOs). Data source capabilities are a point of bitter contention between Veeva and IQVIA. Since 2017, Veeva and IQVIA have been in litigation with each other, with IQVIA claiming, among other things, theft of trade secrets, false advertising, and unfair trade practices. IQVIA also prevents its data from flowing into Veeva OpenData. Veeva is countersuing, claiming antitrust violations and unfair competition.
A notable up-and-coming MDM vendor in the pharma space is Reltio. Founded by ex-Informatica executives in 2011, Reltio offers a SaaS MDM platform called Reltio Cloud. Reltio Cloud is a cloud platform that organizes enterprise data and offers the ability to analyze different data sets and find hidden insights, such as relationships between HCPs, the HCOs they are affiliated with, and the treatment regimens being prescribed to patients.
Seeing the value in Reltio’s technology and wanting to modernize its own commercial offering, IQVIA partnered with Reltio in June 2016 to create a multi-domain MDM solution that provides a single platform to manage HCP, HCO, product, and consumer data. The platform connects to all data sources, including data from internal applications or external sources, such as supplier data, social data, and even data from third-party sources. The solution imports, organizes, and unifies the data. Once aggregated and unified, the platform creates master profiles that are correlated with transactional data, reference data, and affiliation data, allowing it to find relationships between patients and physicians, members and payers, hospitals and suppliers, and drugs and patients.
In addition to Reltio, others in the field include LexisNexis Risk Solutions, a global business information service company that acquired Health Market Science (HMS) in late 2014. HMS performs comprehensive reference data research based on medical claims data from more than 8.5 million HCPs and 1 million HCOs in the United States. LexisNexis’s MDM technology compiles HCP affiliations, insurance plan acceptance, and other data, and the company offers an outsourced data-management service for clients.
Other reference-data providers that stand out to us include MedPro Systems, which touts its ability to keep state licensing attributes accurate, not just for HCPs but also retail pharmacies, wholesalers/distributors, and other parts of the pharma supply chain. The MedProID database (22 million HCPs, 800,000 HCOs) is used by a number of life sciences CRM vendors, such as Synergistix and StayInFront, as well as by sample-accountability provider RxS. Another notable reference data provider is HealthLink Dimensions, which boasts 2.7 million HCPs in its database derived from 500+ sources. The company uses proprietary algorithms and methodology to continuously cleanse and verify its compliance grade data.
Storing the data
The U.S. healthcare system generates roughly a trillion gigabytes of data annually, doubling every two years. With data growing exponentially and becoming more fragmented, pharma and life science organizations face significant challenges with data integration, data transformation, and data cleansing. In fact, 70% of pharma data projects involve managing data before the analysis can even begin.
Despite its importance and inherent value, data is often stored in a custom-built on-premises repository that is typically siloed from the rest of the organization. Although this approach serves the near-term needs of a single department, it makes it hard to maximize the value of purchased data sets as they are not shared with the entire company. As a result, companies lack a clear picture of the data sets acquired by different teams and departments, often resulting in departments purchasing redundant data sets stored in on-premises repositories. Furthermore, even if multiple groups are assembling data sets into a single integrated in-house repository, it can be difficult and expensive to store and manage vast quantities of disparate data on premises.
Companies are waking up to this by replacing on-premises repositories with cloud-based data lakes from the likes of Amazon Web Services, Microsoft Azure, and Google Cloud that pool raw data sets for analytics. Data warehouses are different than data lakes in the sense that lakes store raw structured and unstructured data while warehouses, which need to be rebuilt every 3-4 years due to technology obsolescence, store current and historical structured data in an organized fashion. This delays the flow of insights from reaching the business. But because data is loaded in its native format rather than preconfigured, it can be used in ways beyond basic capture.
Pharma can take advantage of data-lake technology’s ability to handle computing-intensive tasks, such as those required to conduct advanced analytics. Data lakes like Strata from Shyft Analytics (a subsidiary of Medidata) are designed for experimentation, as they can load both structured and unstructured data from third-party and proprietary commercial data sources for ad hoc analysis. Veeva is going beyond the data lake and focusing its efforts on cloud-based data warehousing. In 2018, Veeva launched Nitro, a cloud-based commercial data warehouse built on Amazon Redshift that is pre-built and pre-fed with Veeva reference data. Nitro aims to eliminate the time and effort devoted to on-premise custom data warehouse development and maintenance while providing a foundation for advanced analytics.
Data sources
Having accurate and reliable customer data is only half the battle. Access to accurate RWD during the product lifecycle is the other half. Although randomized controlled trial (RCT) data is considered the gold standard, the FDA is authorizing the collection and use of data gathered outside the clinical trial setting as regulators recognize the need for a more flexible framework for evaluating treatments. The 21st Century Cures Act gives the FDA the authority to include and consider real-world patient-generated data in both its review of new drugs and devices as well as new indications.
The act is consistent with a growing understanding among stakeholders that RWD is generated across various stages of the product development lifecycle, from clinical testing to product launch in the market, as well as during the post-marketing phase. Additionally, the constant barrage of information coming from competing products makes it much harder to use phase III clinical trial data alone to differentiate a therapy in the eyes of doctors, regulators, and payers. Sources of RWD are routinely generated and collected from medical and prescription claims, patient and provider surveys, patient registries, social media, wearables, lab reports, scientific/medical literature, and electronic health records. Although the FDA states it does not endorse one type of RWD over another, questions remain among industry stakeholders on how to turn RWD and RWE into data that can be used to support regulatory approval.
TABLE 14: Four forms of business analytics
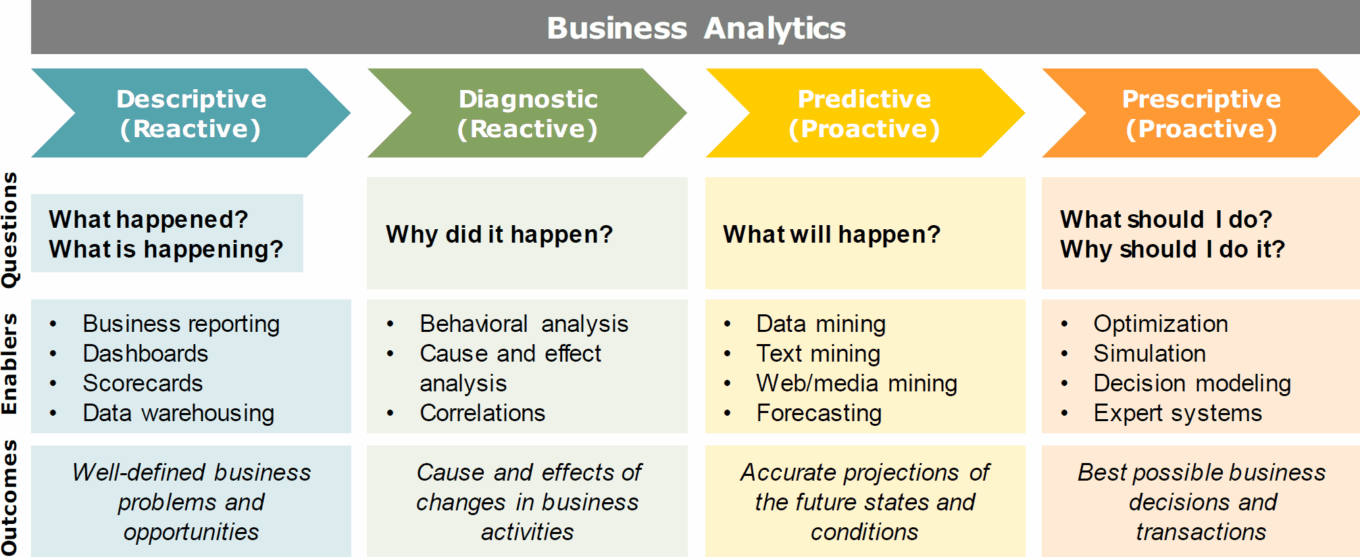
Source: Intelligent Solutions.
Pharma companies recognize RWD is both an opportunity and a challenge. While RWD can significantly improve the effectiveness of strategic decision-making, increasing the likelihood of long-term success, it is growing at a rapid pace such that in the last four years, more data has been created than in all the preceding years of recorded human history.
While HCPs and payers can generate extensive RWD on the use of a particular drug, pharma companies are lagging behind with their internal capabilities. As a result, pharma companies, more than ever, must adopt new digital tools and applications to develop actionable insights and compete more effectively. RWD can lead to more reliable and accurate generation of evidence that increases productivity and reduces costs. A report by IQVIA, for example, looked at a top 10 pharma company with a global budget of $800 million across scientific and commercial domains (excluding RCTs), $400 million of which is in markets where RWE capabilities can be built. It found a savings rate of 25% from use of RWE (vs. exclusive use of primary market research), which would represent $100+ million in potential cost savings.
Given that pharma companies have less internal access to knowledge about how their drugs work in the real world than other stakeholders, they turn to outside vendors for help. For example, a pharmaceutical company’s brand team relies on access to frequently updated third-party utilization data (e.g., prescription new starts, refills, and switches) to track brand performance and make quick adjustments. Individual departments in a pharma company address data gaps by licensing data from external third-party vendors, such as acquiring prescription data from IQVIA, patient/customer data from Crossix (recently acquired by Veeva), disease-based outcomes from TARGET PharmaSolutions, medical imaging data from Life Image, cancer-omics data from NeoGenomics, or medical claims data from Truven Health Analytics (owned by IBM) or Optum (owned by UnitedHealth). Based on our conversations with industry stakeholders, we estimate large pharma companies spend an average of $30 million annually on RWD/RWE.
Analytics
Data integration and aggregation, performance measurement and monitoring, and actionable intelligence are essential to commercialization efforts. Consequently, analytics is an area of great interest and focus for pharma companies. Data analytics tools allow pharma companies to harness the power of RWD generated throughout the product lifecycle, which can provide valuable insights to inform high-level decision-making. We use the terms business intelligence (BI) and analytics interchangeably in our report, but both have broad meaning in terms of the underlying technologies (databases, data integration tools, predictive analytics, and machine learning) and use cases.
TABLE 15: Analytical applications for sales and marketing

Source: First Analysis.
Table 14 shows four basic forms of BI, and while most of the healthcare industry is at Stage 1 (Descriptive) or 2 (Diagnostic), several organizations are moving to Stage 3 (Predictive) or Stage 4 (Prescriptive) and using technologies such as artificial intelligence. AI is an umbrella term for multiple computer science technologies that can be combined in different ways to perform capabilities normally requiring human intelligence. AI is essentially a smart solution for compiling and analyzing data, taking account of more variables than humans are able to, categorizing the new data, predicting trends, and ultimately identifying solutions. These technologies have the capability to sense, comprehend, act, and learn. Sensing technologies such as computer vision and audio processing involve perceiving the environment by acquiring and processing images, sounds, and speech.
Comprehending technologies include natural language processing (NLP), which can enable AI systems to analyze and understand the information in knowledge representation. The act and learn capabilities of AI allow systems to take action through expert systems technologies, machine learning (ML), and inference engines. Currently, we estimate the life science analytics market size is ~$6 billion and growing at a CAGR of 20-30%.
Growing demand for reducing healthcare expenditure, increasing instances of regulatory oversight, and declining R&D productivity are some of the key drivers contributing to market growth. Increasing adoption of analytical solutions for various applications in pharma companies, such as drug discovery, clinical trials, sales and marketing, and risk management, is further expected to boost the market. Table 15 highlights how analytics can be used to improve sales and marketing effectiveness.
The ability of AI to generate significant improvements in cost, quality, and success has encouraged many pharmaceutical companies to explore AI along the R&D value chain in drug discovery and development. Drug discovery is predominantly a trial-and-error process that is typically very time-consuming as it involves the identification and evaluation of thousands of compounds. For every 5,000-10,000 compounds initially evaluated, only five advance to human testing, and only one is ultimately approved for commercialization. Additionally, 15-20% of a drug’s development cost is in the discovery phase, amounting to hundreds of millions of dollars. As a result, reducing the cost and time of drug discovery and increasing the clinical trial success rate have become imperative. One way to lower costs is to use AI to screen compound libraries virtually instead of physically testing every compound in a given library. This offers pharma R&D a faster and more efficient method of finding lead compounds that have the potential to become effective drugs.
TABLE 16: Drug discovery and development timeline
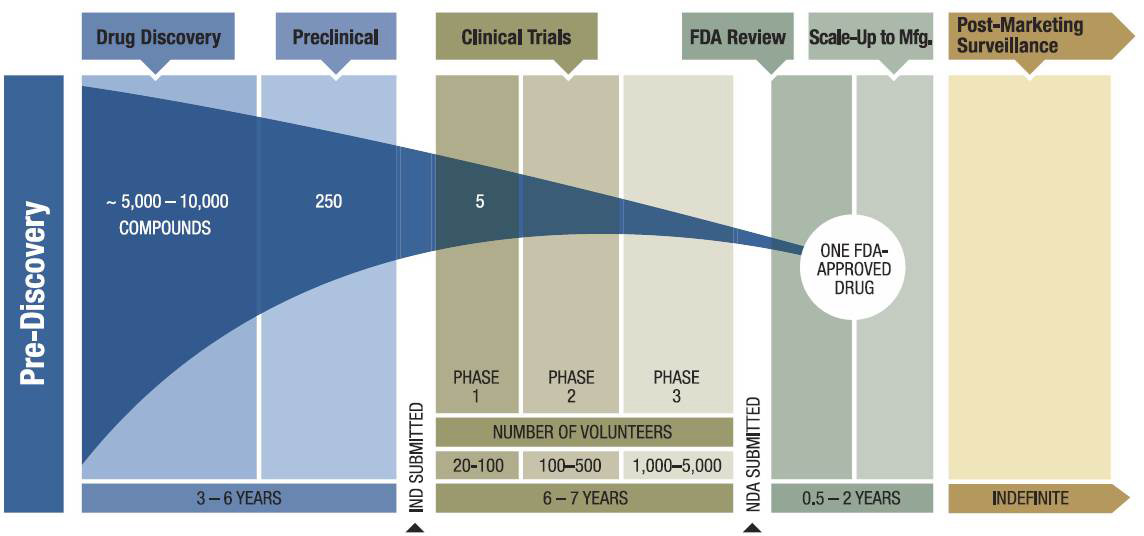
Source: American Association of Cancer Research 2011 Cancer Progress Report.
There are a number of companies developing AI platforms for application in drug discovery including IBM Watson and Google DeepMind Health. The approach many pharmaceutical companies are taking is to engage in partnerships with AI venture-funded companies offering different solutions to identify new drug targets, find new uses for existing drugs, or secure faster approval.
One notable vendor, Insilico Medicine, applies deep learning algorithms to drug discovery. Through its GENTRL platform, the company applies generative adversarial networks (GANs) and reinforcement learning algorithms to generate new molecular structures and to find the biological origin of a disease. GAN is a branch of AI that works when a generator produces real-looking images while the discriminator attempts to recognize which one is a fake to increase accuracy in generating images, videos, and text.
Atomwise offers Atomnet, a deep learning neural network platform for structure-based drug design and discovery. Atomnet’s deep learning algorithms analyze molecules and predict how they might act in the human body, including their potential efficacy as medication, their toxicity, and their side effects, thereby reducing the time researchers need to spend synthesizing and testing compounds.
Another notable vendor, BenevolentAI, has developed machine learning capabilities and algorithms to mine research literature, molecular data, and proprietary research databases. The company is focused on repurposing or resurrecting existing assets in which significant investment has already been made. The tool improves the selection of candidates and drug targets using AI analytical capabilities. BenevolentAI’s platform analyzes molecular data, study findings (both positive and negative), and unstructured data related to compound efficacy and combines it with a host of commercially relevant reimbursement and outcomes data that can inform strategic decision-making. Other notable vendors include BERG, TwoXAR, Owkin, Numerate, Phenomic AI, Exscientia, Qrativ, XtalPi, ReviveMed, Arpeggio Biosciences, Auransa, and Genomenon. We estimate the current annual market for AI in drug discovery to be~$1 billion.
Tapping into commercial insights during the early phases of clinical trials can lead to more informed decisions about clinical trial design and strategies. Similarly, insights gleaned from clinical trials can lead to improved decisions about regulatory and payer approvals, market access, reimbursement and formulary inclusion, and other areas that are critical to drive commercial success. Clinical trial data is a key corporate asset, providing evidence of efficacy and safety as well as a drug’s potential economic value to the market.
TABLE 17: How Saama leverages AI in key areas of clinical operations
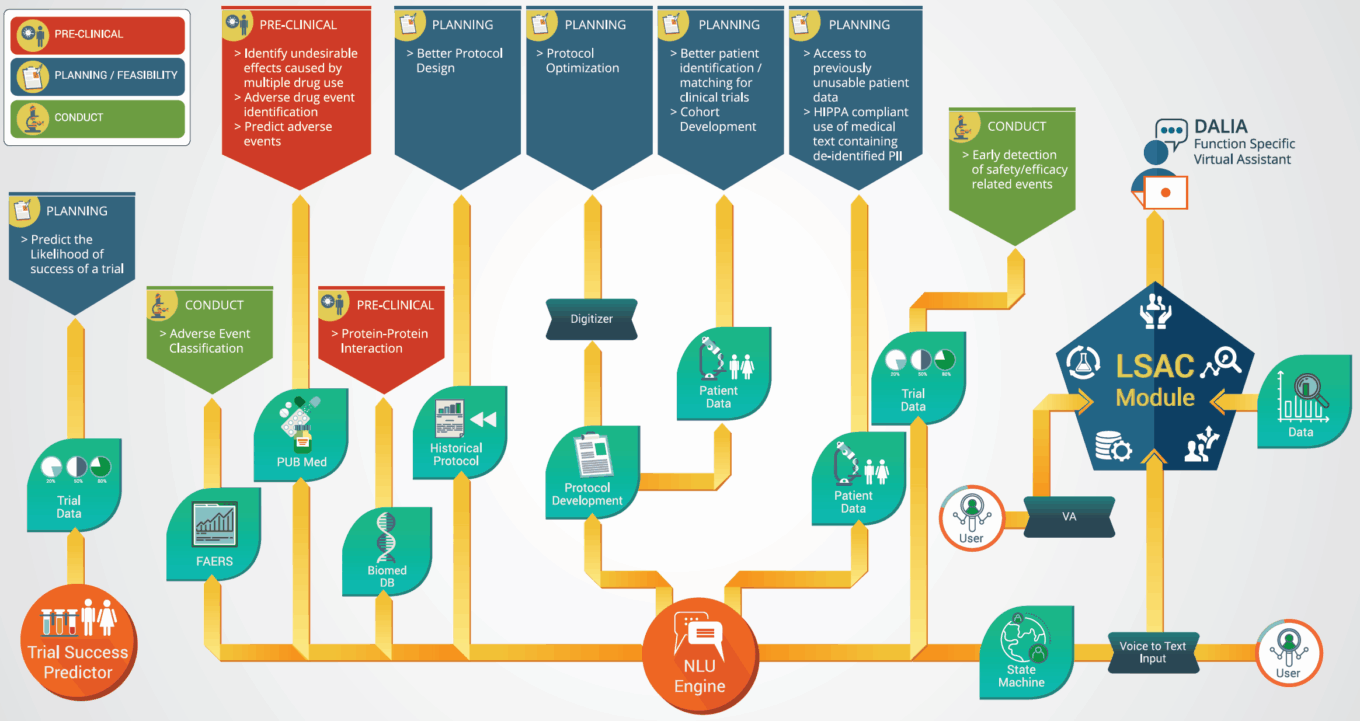
Source: Saama Technologies.
A pharmaceutical company typically has multiple clinical trials running simultaneously. Each trial generates a very large amount of data, which is recorded in electronic data capture (EDC) systems and clinical trial management systems (CTMS). With the introduction of RWD into various stages of the trial process, the volume and sources of data have increased tremendously, making it difficult to derive meaningful insights at an enterprise level. This is where AI can make a difference.
One notable vendor, Saama, offers a unified, AI-driven clinical data analytics platform that integrates, curates, and animates sources of structured and unstructured data and RWD to deliver actionable insights. Saama’s solution comes equipped with a virtual assistant, DaLIA (Deep Learning Intelligent Assistant), which is context-aware and hence reduces the cycle time for understanding information. DaLIA is designed to accelerate business outcomes, innovations in drug discovery, and market reach.
We believe an area within clinical trials that is ideal for AI is patient recruitment. Patient recruitment for clinical trials often leads to bottlenecks in the drug development process and ultimately to delays in launch dates for new drugs, particularly as trials become increasingly complex and specialized (i.e., rare diseases). Industry estimates suggest that 50% of clinical trials need to extend their recruitment period, resulting in a loss of up to $1.3 million per day for a given drug candidate. Additionally, 11% of trial sites fail to recruit any patients, while 37% under-recruit and fail to meet their enrollment targets. In the United States alone, we estimate $11 billion is spent each year on patient recruitment. This equates to 20% of pharma’s annual U.S. clinical development spend (phases I-IV).
By leveraging new patient-centric technologies, like that of Deep 6 AI, which offers software that uses NLP to analyze structured data such as ICD-10 codes and unstructured clinical data, including doctor’s notes, pathology reports, genomic data, discharge summaries, and operating notes, trial sponsors can improve the identification and enrollment of potential participants as well as make trials more accessible and convenient.
TABLE 18: Clinical trial recruitment costs
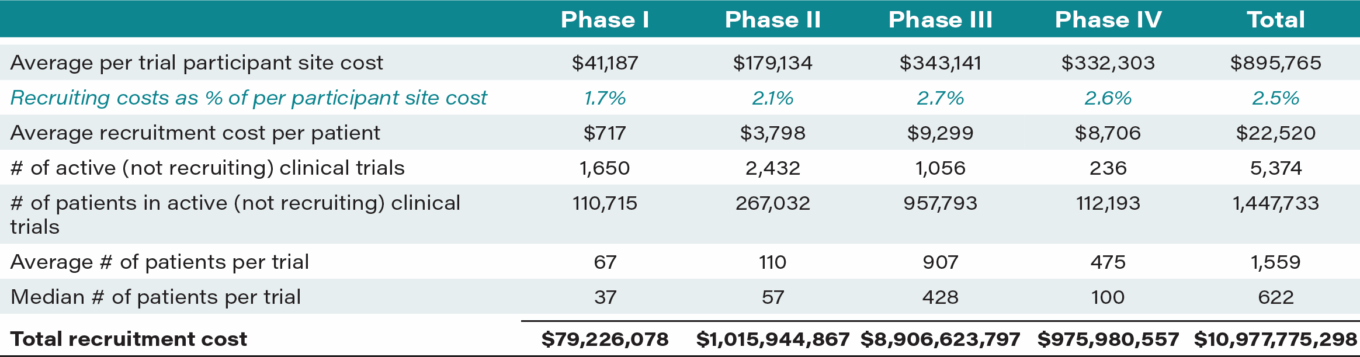
Source: ERG Study, TEConomy Partners Study, ClinicalTrials.gov, First Analysis estimates.
An interesting vendor working in the clinical trial enrollment space is VIDA, whose AI-powered image analysis software for lung diseases helps expedite the time-sensitive patient screening process. Another notable vendor in the lung space is Biodesix, with its AI-powered Diagnostic Cortex platform. Diagnostic Cortex’s AI platform incorporates multi-omic biomarker data, clinico-pathologic characteristics, and clinical outcome data to design diagnostic tests that identify which patients are likely to benefit from a particular drug or therapy. Other notable vendors with a focus on clinical trials include Tempus, AiCure, Datavant, Mendel.ai, TriNetX, and ArcheMedX.
TABLE 19: Biodesix’s diagnostic test design approach
Source: Biodesix.
Facilitating R&D is perhaps the most common use case for AI applications in the pharmaceutical industry, but AI has applications beyond R&D. A recent Accenture survey found that 90% of industry executives recognize AI as important in driving commercial innovation. As discussed above, commercialization is changing dramatically due to the inaccessibility of HCPs and the transition to outcome-based reimbursement models. At the same time, the industry is becoming more competitive and dynamic.
Apart from the traditional sales-force visits to HCPs, there are a myriad of other ways of engaging HCPs and patients, but in order to do so, commercial teams need access to deeper market insights to craft data-driven strategies and implement their efforts with speed and precision. Actionable insights help brand managers, field reps, and medical science liaisons (MSLs) improve decision-making and take smarter actions to personalize their engagements and achieve greater commercial success. AI combined with real-world data from pharmacies, payers, HCPs, labs, marketing campaigns, KOLs, and social media can replace traditional gut instinct with evidence that uncovers specific HCP and patient-level insights without compromising data privacy rules.
With real-time insights and suggestions to drive HCP engagement and sales-force execution, Veeva has built AI functionality into its CRM offering with the recent release of its Andi application. IQVIA has Ada, which generates recommendations for sales reps or marketing teams based on data stored horizontally across the enterprise through a combination of machine learning and natural language processing.
TABLE 20: LungPrint discovery
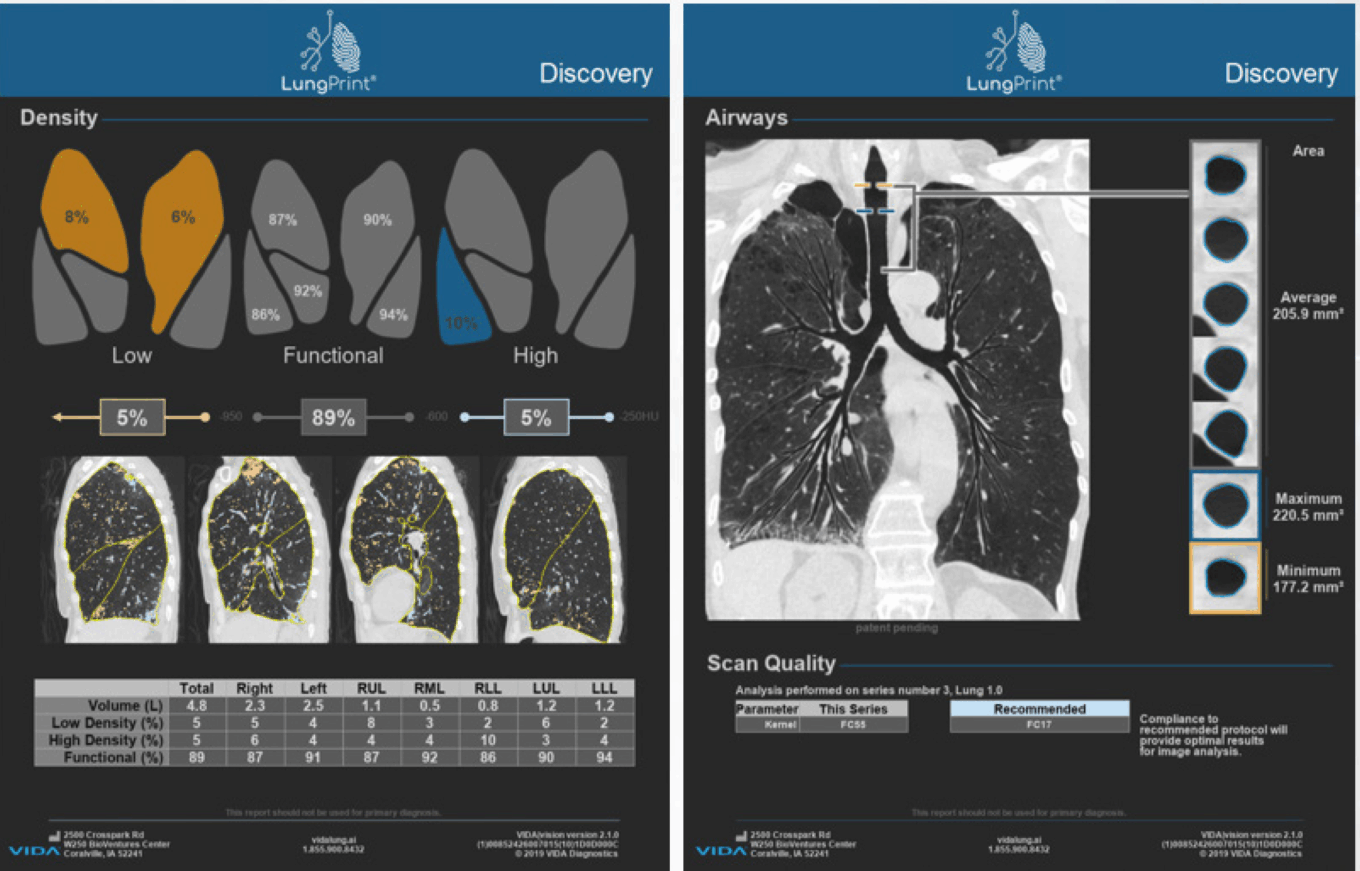
Source: VIDA.
Built on top of a platform composed of more than 17,000 clinical trials and a structured, standardized clinical data repository with over 4.8 million patients, Medidata (now owned by Dassault Systèmes) recently launched its own AI offering called Acorn, which is designed to provide actionable insights from R&D to commercialization.
Aktana’s Decision Support Engine provides pharma sales reps with next-best-action insights, such as whether or not to visit an HCP, send an email, send a letter, provide drug samples, or invite an HCP to a seminar or webinar. The machine learning model is platform agnostic and uses customer CRM data derived from Salesforce, IQVIA, Veeva, or Marketo. Other notable vendors in the space include Hexi.ai, Closerlook, Komodo Health, Syneos Health, Innoplexus, Anju Software, Axtria, Mu Sigma, Prescrib-er360, and Decision Resources Group (DRG).
TABLE 21: Aktana’s decision support engine
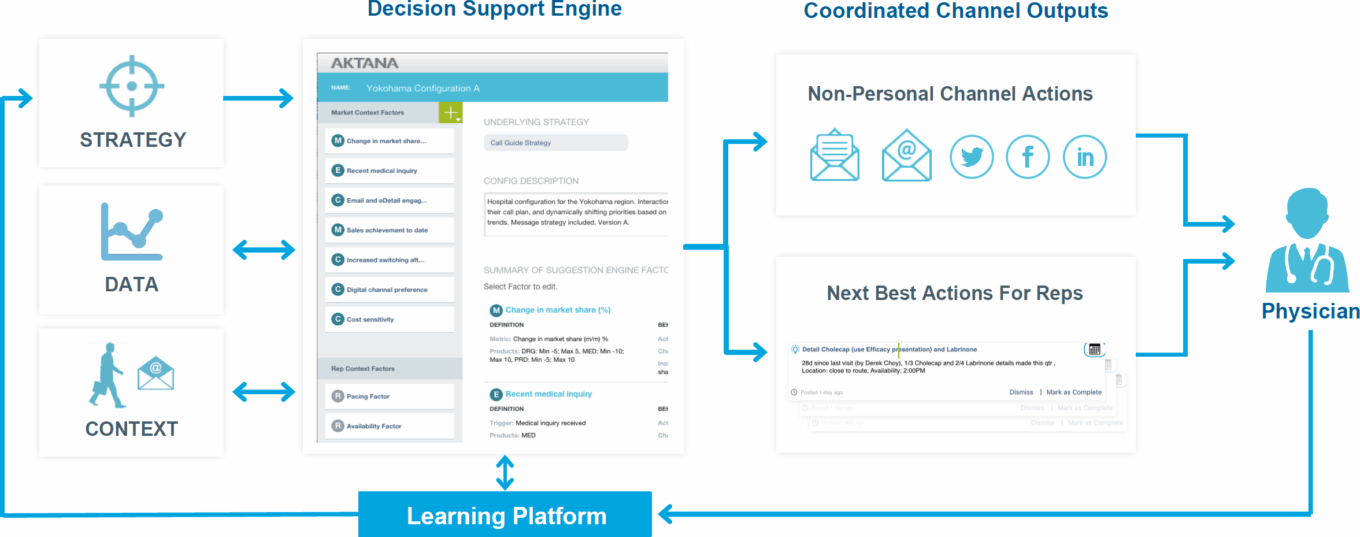
Source: Aktana.
The FDA requires companies to monitor approved drugs for as long as they stay on the market and requires them to submit periodic reports on safety and tolerability. Companies must also report any serious and unexpected adverse events that occur from the use of the drug to the FDA in an expedited manner. Furthermore, the FDA sometimes requires companies to conduct phase IV clinical trials, which evaluate the long-term safety or effects of drugs in specific patient subgroups. This area within pharma is known as pharmacovigilance (PV). PV is defined as the science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problem.
Outside of clinical trials, ~95% of treatment-related adverse events are estimated to be underreported by HCPs. Additionally, underreporting by patients in the FDA Adverse Event Reporting System (FAERS) continues to be an issue, with only 20–33% of the minimum number of expected serious events being reported. Because social media has been a go-to source for patients to seek advice and share experiences, social media listening via AI is an important tool to augment post-marketing safety surveillance, such as FAERS.
Reflecting the potential importance of social media data for PV, the FDA signed an agreement in 2015 with PatientsLikeMe to determine how patient-reported data could help generate insights into drug safety. In terms of PV software, Oracle, with its database and capture software Argus and its data, analytics, and workflow software under its Empirica brand, is the market leader.
There are other companies working to gain market share from Oracle, such as ArisGlobal, largely seen as Oracle’s most significant competitor, as well as smaller companies like AB Cube and MyMeds&Me. Two other notable vendors, Commonwealth Informatics (recently acquired by Genpact) and Advera Health Analytics, have also built analytical solutions into their workflow platforms.
TABLE 22: Pharmacovigilance reported in social media posts

Source: S. Chowdhury, C. Zhang, P. Yu, Multi-Task Pharmacovigilance Mining from Social Media Posts.
Emerging digital channels
The most common type of data that pharma uses to target HCPs for product promotions is prescription data, which physicians generate when they write prescriptions. Prescription data analytics can provide insights into product adoption, diagnosis and treatment, adherence, compliance, product switching, and physician specialty. While this data is useful, it lacks crucial engagement components for pharma that it needs to drive a long-term relationship with HCPs. As a result, pharma is turning to outside vendors to leverage this data in differentiated channels.
One such channel is augmented reality/virtual reality (AR/VR). AR/VR applications, such as games, both educate and entertain HCPs and are making inroads increasing engagement. According to the 2019 MM&M/Deloitte Healthcare Marketers Trend Report, 32% of pharma and biotech companies implement AR/VR in their HCP engagement strategy. Physicians can earn continuing medical education (CME) credits by completing games that help them make better treatment decisions, and games can help surgeons sharpen their skills. HCPs tend to be competitive by nature, as evidenced by a MedLive.com survey of 500 MDs that found that 82% of MDs are interested in competing against global peers in online health games that test their knowledge.
One innovative company in the space is Level Ex. Using high-end graphics standard to the video game industry, Level Ex has partnered with pharmaceutical and medical device companies to create mobile games in specialties ranging from cardiology to gastroenterology that allow HCPs to navigate complex diagnosis scenarios, administer treatments, and learn how patients respond. The company’s games, available for both iOS and Android, are proving to be popular, with over 500,000 HCP users and 3 million in-game medical cases played in 2018, and it recently launched its fourth smartphone-based game titled Cardio Ex, which is for practicing cardiologists. Level Ex isn’t the only company helping pharmaceutical and medical device companies engage with HCPs through AR/ VR. ImmersiveTouch, for example, offers 3D surgical simulation software designed to train nurses and doctors on new devices.
TABLE 23: Pulm Ex Game
Source: Level Ex.
Background on KOLs
Historically, the pharmaceutical industry has depended heavily on influential doctors to help validate and promote the use of its drugs. The key reason is that doctors trust other doctors, especially leaders in the field. The pharmaceutical industry calls these doctors key opinion leaders or thought leaders. Companies know that messages coming directly from pharmaceutical companies are likely to be viewed skeptically by physicians.
As a result, the concept of using KOLs as an “independent” source of information has significantly expanded since the mid to late 1990s. KOLs are typically physicians who have a recognized expertise in a specific field (e.g., oncology, endocrinology, urology, neurology). They are a key lever in pharma drug development, clinical trials, and commercialization, often serving as product champions and exerting their influence through research publications, presentations, webinars, contributions to editorial boards, guideline committees, and professional societies. According to a 2015 Healthcasts survey (n=399), 84% of physicians are likely to prescribe a new drug if it’s endorsed by a KOL.
That being said, the role of KOLs in promotional activities is controversial. The industry wants to associate with the top HCPs in their specialties, but questions and concerns regarding potential conflicts of interest continue to emerge, such as those regarding scientific sessions sponsored by a drug company and employing a KOL to speak on its behalf for fees in the thousands of dollars. Despite the concerns, pharma continues to see the value in KOLs and devotes 15-25% of its annual marketing budget to them.
Thanks to regulatory hurdles and the proliferation of specialty drugs, identifying and gaining access to KOLs has become a challenge for pharmaceutical companies. Some health-care systems, such as Kaiser Permanente and academic medical centers, prohibit or limit speaker fees, non-research consulting, and meals. Additionally, in 2018, New Jersey adopted gift restrictions, capping annual consulting and speaking payments to $10,000 and meals to $15.
Historically, teams of medical science liaisons would identify KOLs via surveys, clinician advisory boards, membership in professional societies, and literature searches. Besides building and maintaining relationships with KOLs, MSLs also act as the bridge between HCPs and the pharma company by serving as scientific peers and providing information and resources on the company’s drugs that are both commercial and in development. The emergence of data capture technology has enhanced the ability of MSLs to gain deeper insights into HCPs and has resulted in KOL mapping.
TABLE 24: Delving into the KOL’s world: valuable insights for pharma
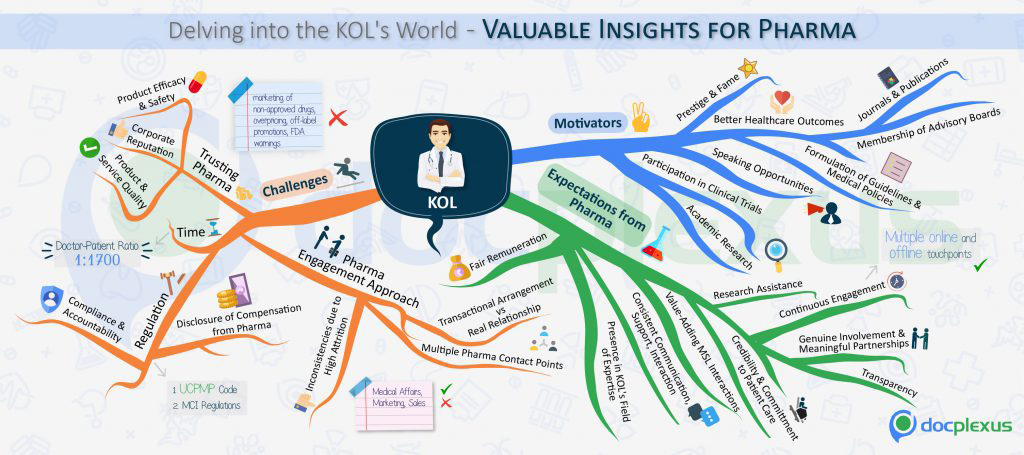
Source: Docplexus Online Services.
KOL mapping is a data- and analytics-driven approach to identifying KOLs at the local, regional, national, and global levels. Mapping identifies influential thought leaders who can create a buzz through peer discussion long before a product reaches the market. One such KOL AI-based solution, Fabric from rMark Bio, is a BI platform that uses machine learning to mine and analyze internal data sources, such as KPIs, CRM, and sales rep data, along with publicly available global health data (journal publication data, grants, clinical trials, and Sunshine Act data) to provide MSLs recommendations on KOLs that match the pharma company’s business strategy.
Two other notable vendors are Swoop and ACTO. Swoop’s IPM.ai subsidiary helps pharma companies discover and match HCPs based on a drug’s ideal target patient population. ACTO’s cloud software provides MSLs with the capability to search and share key product and clinical information with KOLs in real time.
Digital KOLs
Today, HCPs are more open to using the internet and social media to communicate with peers. This has resulted in the emergence of digital KOLs like Dr. Zubin Damania, known as ZDoggMD, whose medical-related parody music videos have attracted 272,000 followers on Instagram and 1.4 million followers on Facebook. In addition to ZDoggMD, a number of other physicians have built a following online, including podcast doctor Rob Orman and healthcare bloggers Dr. Scott Weingart and Kevin Pho. These doctors have a strong digital presence, and other clinicians enjoy reading their take on trends, research, and new products. It’s not that traditional KOLs have diminished in influence. They continue to play an important role in marketing and remain highly sought after.
But where KOLs were once viewed as an oligarchy, they’re now joined by a new breed of digital-savvy HCPs. As social media has evolved, medically focused professional communities have been established. These networks are often private and restricted for nonmembers and offer digital KOLs a platform to communicate instantly and update peers. According to a 2018 survey (n=815) from Decision Resources Group (DRG), 80% of U.S. physicians use social media with 56% sticking to physician-only communities such as Sermo, Doximity, and Skipta.
TABLE 25: HCP social media communities

Source: First Analysis.
Physicians prefer physician-only communities that offer a “walled garden” for collaborative learning and engagement. With the ability to listen and mine social media content to derive sentiment, conduct in-community surveys, and deliver targeted rich content, pharma can glean valuable insights into their own drugs’ performance and competitor activities, identify KOLs, uncover new potential indications for existing drugs, and enhance drug launch strategy. NLP makes it possible to carry out real-time social media listening into these communities to identify relevant conversations and insights. NLP enables analysis of language patterns on social media, and human interpretation reveals the significance of those patterns in context. Eularis is one company that offers NLP software that can analyze social media data to predict and locate future KOLs, reveal how a drug brand is being shared on social media, and report the audience reaction to new advertisements.
Patient centricity
The economic burden placed on patients as a result of rising costs has led to greater patient influence over healthcare delivery. For example, the number of workers on high-deductible plans has grown from 4% in 2006 to 28% in 2017. Additionally, the percentage of employer-sponsored plans requiring deductibles for pharmacy benefits has risen from 23% in 2012 to 52% in 2017.
The rise in healthcare consumerism, coupled with the democratization of data and information, has resulted in patients being more informed than ever before. They are researching conditions and potential treatments online (including a drug’s out-of-pocket cost, benefits, and side effects), sharing experiences via social media and online communities, discussing and selecting care regimens with their physician, and using new wearable technologies and mobile health (mHealth) applications to monitor and track their health. Every day, more patient experiences are being shared digitally, perhaps none more prevalent than patient reactions to the rising cost of prescription drugs.
Prescription drug pricing has become a large target thanks to recent and highly publicized price increases that have put the industry in the crosshairs of the public and politicians on both sides of the aisle. President Trump has stated that reducing drug prices is one of his highest priorities. The administration recently issued new regulations requiring manufacturers to list the price of their drugs in television ads. Congress has already passed the Know the Lowest Price Act and the Patient Right to Know Drug Prices Act, both of which promote transparency in prices. The Senate Special Committee on Aging recently conducted a series of hearings on drug pricing, and the Senate’s bipartisan Prescription Drug Pricing Reduction Act aims to reduce government spending on drugs by an estimated $100 billion over the next decade.
TABLE 26: Healthcare & pharma industry digital ad spending in the U.S., 2016-2020
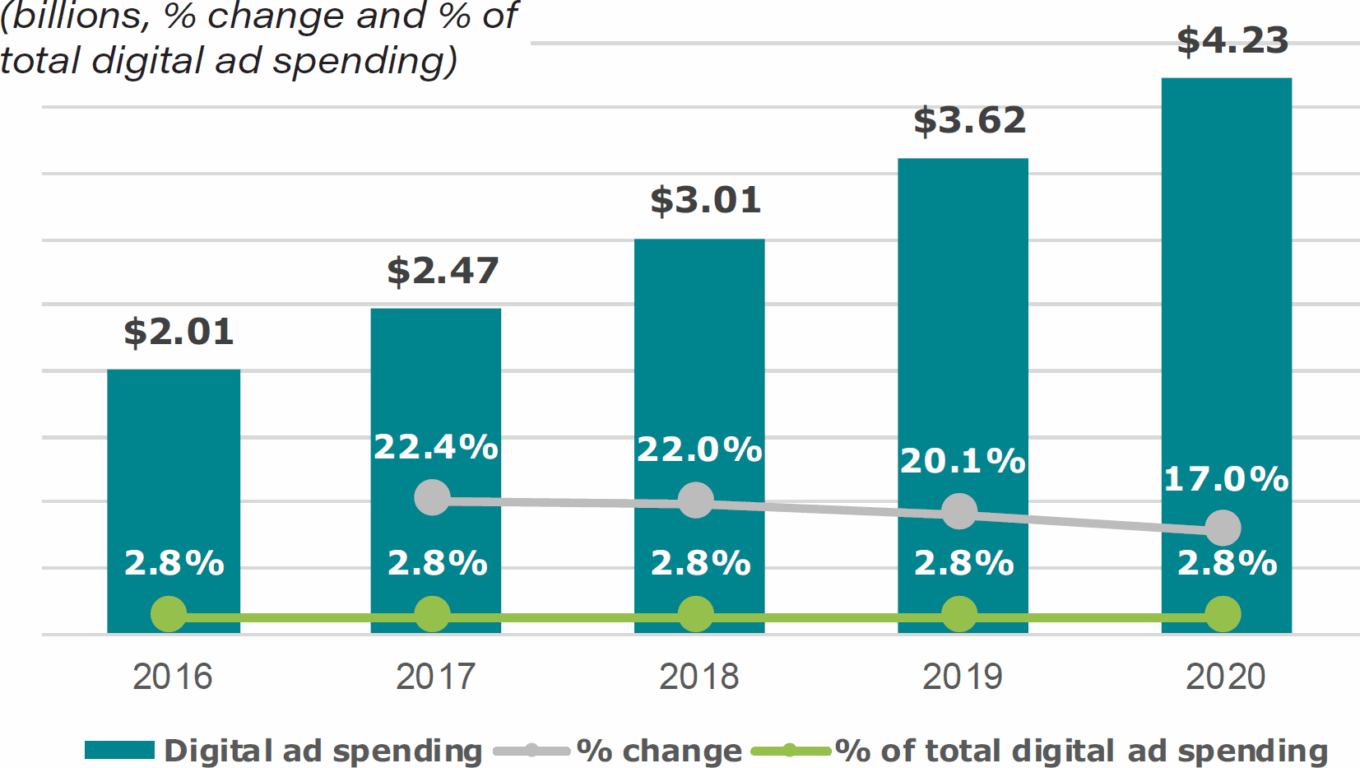
Source: eMarketer.
According to a recent Gallup poll, the pharmaceutical industry is now the most poorly regarded industry in the country, with 58% of those polled holding a negative view vs. 27% holding a positive view. Approximately 40% of patients don’t know any of the pharma companies behind their treatments, and only ~15% of patients feel like they have a relationship with the pharmaceutical companies that make their medications. As a result, we believe the pharmaceutical industry needs to expand its focus beyond the supply side of the equation and focus on the demand side by adopting more of a patient-centric approach.
Being patient-centric essentially means having a deeper understanding of the patient experience in terms of what patients value and need, their attitudes and behaviors, and what is most likely to improve treatment outcomes. The concept of patient centricity is not new. Over the last several years, there have been numerous patient engagement initiatives involving regulatory bodies, patient advocacy groups, HCPs and payers, and the pharma industry. That being said, patient centricity is a moving target as the industry works toward achieving the triple aim of improving patients’ experience of care, driving advances in population health outcomes, and reducing the per-capita cost of healthcare.
From a legal perspective, the Healthcare Insurance Portability and Accountability Act (HIPAA), the California Consumer Privacy Act (CCPA), the EU General Data Protection Regulation (GDPR), the FTC, and clinical trial institutional review boards (IRBs) provide a regulatory framework for how patient data can be used. Traditional healthcare data sources, such as prescription data, which capture a physician’s prescribing behavior, tend to fall short in tracking patient behavior and total care. Pharmaceutical companies also understand that their therapies work for some, but not all, patients. Instead of targeting every person with the same drug, the industry approach has shifted toward precision or personalized medicine, which essentially means targeting the right patient for the right drug at the right time. As a result, pharma companies are joining payers and clinicians in adopting new digital technologies that collect and analyze longitudinal patient-level data, often spanning different areas of the pharma value chain, from R&D to product launch to disease management.
For example, R&D processes in combination with commercial functions can organize and collect data that may be required in the commercialization and marketing of a drug while perhaps assisting in the design of clinical trials. The patient-level data is deidentified to meet HIPAA regulations and can be derived from physician visits, hospitalizations, clinical labs, long-term care therapy, outpatient care, drug therapy, and payers.
Another use case is real-time insights based on clinical lab tests. Although historically focused on hospitals and physicians, clinical labs are currently instituting more patient outreach initiatives thanks in large part to the growing popularity of direct-to-consumer genetic test offerings from companies like 23andMe, Ancestry, and Orig3n. Clinical laboratory tests provide objective information on the functioning of the human body so patients can be diagnosed, treated, or monitored precisely and as quickly as possible. The information furnished by these tests, which are performed on a patient’s blood, saliva, and/or tissues, provides the necessary data for physicians to make informed decisions.
Clinical laboratory tests influence more than 70% of medical decision-making and account for less than 5% of overall healthcare spending in the United States. The tests are conducted in two distinct settings: inside the hospital (includes hospital inpatient and outpatient testing) and outside the hospital (includes independent labs, physician-office labs, and hospital outreach [non-hospital patients] labs). More than 7 billion tests are performed in the United States each year.
One company we see making significant inroads in collecting and interpreting this data is Prognos, which has partnered with diagnostic testing labs to collect and analyze testing data on 200 million patients spanning 50 disease states, which it marries with reimbursement data from payers to assist pharmaceutical companies in identifying which patients could benefit from their drug, as well as who and where their HCPs are.
Direct-to-consumer
To offset their lack of access to physicians while ensuring consumers learn about their therapies, pharma companies have increasingly turned to direct-to-consumer (DTC) advertising. Currently, the United States and New Zealand are the only two countries that allow companies to market their medicines via DTC advertising. In the United States, pharma marketing is regulated by the FDA. FDA regulations require all prescription drug promotion to be truthful, to avoid misleading information, to be based on “substantial evidence or substantial clinical experience,” to provide a “fair balance” between the risks and benefits of the promoted drug, and to maintain consistency with labeling approved by the FDA. The FDA’s Office of Prescription Drug Promotion (OPDP) enforces these requirements. From 1997 through 2016, spending on medical marketing of drugs, disease awareness campaigns, health services, and laboratory testing increased from $17.7 billion to $29.9 billion. DTC prescription drug advertising increased from $1.3 billion (79,000 ads) to $6.4 billion (4.6 million ads [including 663,000 TV commercials]), with a shift toward advertising high-cost biologics and cancer immunotherapies.
While DTC is an effective tool to increase awareness, it’s not foolproof and doesn’t ensure a brand’s message is correctly received. In addition, the increased scrutiny of DTC TV advertising from both industry watchdogs and regulators could reduce its effectiveness, particularly for specialty drugs. In 2015, the American Medical Association (AMA) called for a ban on DTC advertising because of concerns that it drives up drug costs without adding benefit.
Currently, the main issue is whether or not drug pricing should be included in DTC ads. The price disclosure rule issued by the U.S. Department of Health and Human Services (HHS) in May 2019 would require prescription drug manufacturers to include a statement of “list price,” defined as 30-day-supply wholesale acquisition cost (WAC) in all DTC TV ads for prescription drugs and biologics. In practice, this could be challenging as pricing for treatments for certain diseases like cancer are complex and may include multiple parties and variables. Additionally, disclosure of the list price without additional context could be misleading, as most patients do not end up paying the full list price for drugs due to insurance, rebates, and coupons.
TABLE 27: Crossix approach to measuring marketing campaigns
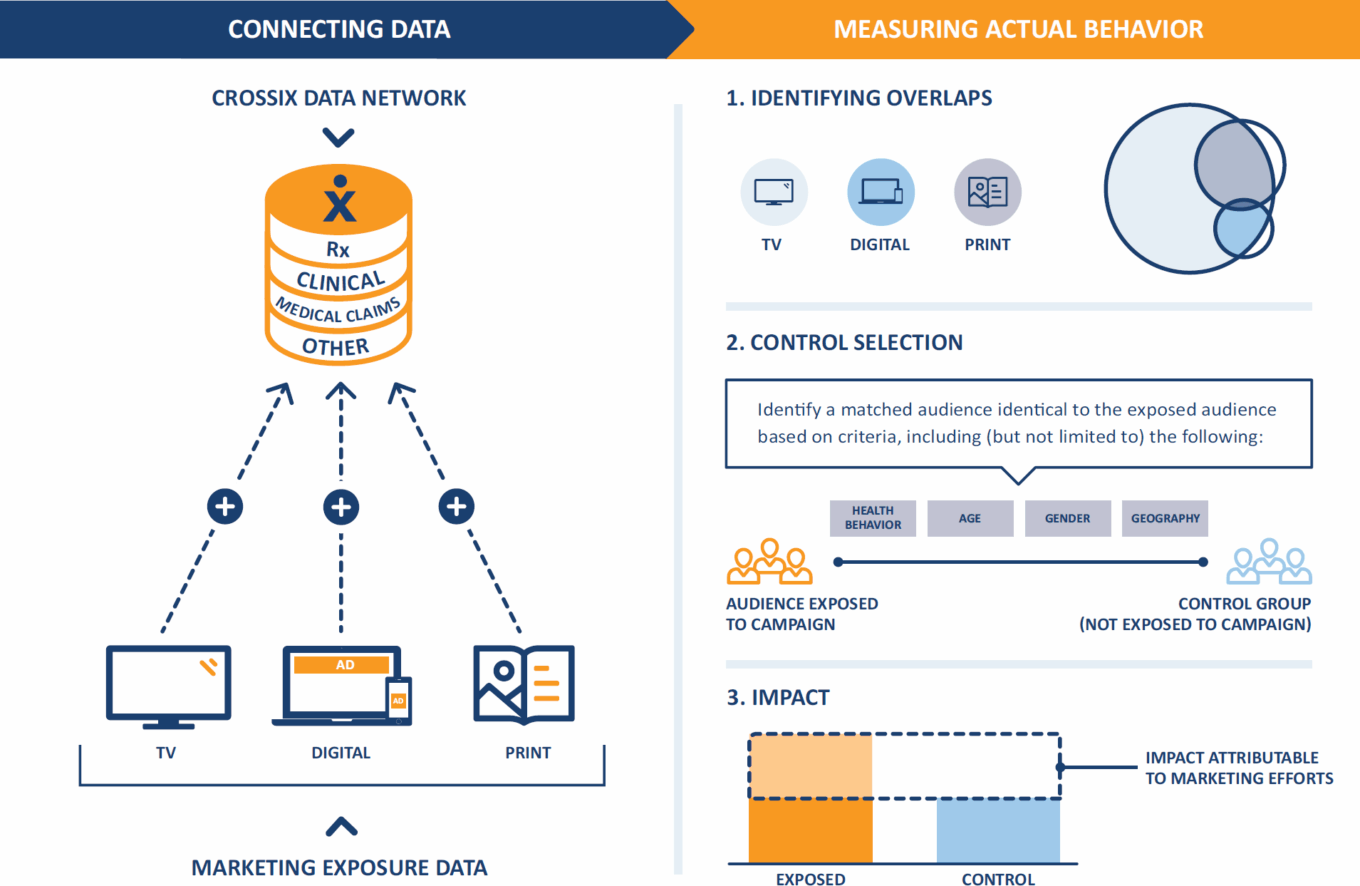
Source: Crossix.
According to PhRMA, on average, 40% of the list price of drugs is given as rebates or discounts to insurance companies, the government, pharmacy benefit managers, and other entities in the supply chain who often require large rebates in order for a drug to be covered. These rebates and discounts exceeded $166 billion in 2018 and are growing every year. In July 2019, a federal judge granted the plaintiffs’ (Merck, Eli Lilly, Amgen, and the Association of National Advertisers) motion to stay the final rule’s effectiveness, ruling that HHS lacked the authority from Congress to compel drug manufacturers to disclose list prices. In September 2019, HHS filed an appeal in D.C. Circuit Court challenging the judge’s ruling. In November 2019, senators from both parties unsuccessfully pushed for a vote on a bill that would require DTC drug ads to include list prices.
The bill, known as the Drug Price Transparency in Communications (DTC) Act, was introduced in May by Chuck Grassley (R-IA) and Dick Durbin (D-IL). If passed, the DTC Act would amend the Social Security Act to require that drug ads include “truthful and non-misleading pricing information.” The DTC Act would circumvent the aforementioned HHS rule. Given that the issue doesn’t appear to be going away anytime soon, pharma companies are turning to digital alternatives like point-of-care (POC) marketing to get their message out.
Point-of-care
According to a 2017 survey from the Medical Group Management Association (MGMA), patients waited ~20 minutes on average in the HCP’s waiting room before heading to the exam room. This waiting time offers pharma another opportunity to engage with patients through relevant content delivered either on digital screens or in the form of paper brochures with messages such as when and how to know if a treatment is working, ways to save on prescription costs, and medication adherence tips. In addition to helping pharma deliver its message, POC materials can foster the patient-HCP relationship beyond the appointment.
The biggest issue with POC has always been how to measure and validate its impact or ROI. This has been exacerbated by the 2017 scandal involving POC market leader Outcome Health. At the time, the Wall Street Journal reported that Outcome Health misled its pharmaceutical clients by charging them for ad placements on more video screens than the company had installed. Additionally, the company created documents that inaccurately verified that ads ran on certain doctors’ screens and manipulated third-party analysis showing the effectiveness of the ads.
Despite the scandal, POC remains an important and growing engagement channel for pharma due to it essentially being the “last mile” before a patient sees an HCP. To alleviate concerns about ROI, pharma companies and POC-focused companies like PatientPoint and Phreesia have partnered with third-party marketing data and analytics vendors like IQVIA, Symphony Health (owned by PRA Health Sciences), and Crossix to measure and validate campaign effectiveness using metrics such as prescription lift. According to the Point of Care Communications Council, industry spending on POC is $600 million a year and expected to grow 15% annually through 2022.
In addition to DTC and POC, pharma companies are engaging patients by deploying wearables and mobile devices, mobile apps, social media, games, and hub services to facilitate and map the patient journey. By mapping the patient journey, pharma companies are better equipped to identify potential obstacles that may impact a patient’s ability to receive a prescribed treatment, as well as identify the best time to reach out directly. For example, a pharma company may want to reach out to patients to remind them to pick up their prescriptions from the pharmacy or take their medication.
mHealth
The rapid adoption and use of smartphones and other mobile devices has been widespread. According to Pew Research, 96% of Americans (91% of seniors) have cellphones that enable text messaging, and 81% have smartphones that enable mobile applications. This presents a significant opportunity for life sciences companies to engage with patients and gather valuable RWD. Innovations in digital technology, such as web-connected wearable mHealth devices and applications, are putting health in the hands of the individual, whether for general health and wellness or for chronic disease management. Currently, there are more than 325,000 health apps worldwide, twice as many as there were in 2015 and five times the number in 2013.
Many of the most popular apps also connect to wearable biosensors and no longer rely purely on patient input data. Wearable biosensors collect information on a variety of health parameters and vital signs by reading or measuring energies from a person – pressure, temperature, light, etc. – and transmit that data via electric signals to be interpreted. Activity monitors measure consumer motion patterns (e.g., movement, rotation, and position) and translate them into measures of routine activity like sleep, steps, and exercise.
The concept of wearable biosensors is not new; in 1960, the first continuous ECG data was collected from patients. The industry breakthrough came in 2007 with the release of Fitbit (recently acquired by Google), which popularized the use of wearable devices to monitor personal health information. Since then, the use of wearables has been increasing throughout healthcare, including in clinical trials. A recent search of Clinicaltrials.gov shows that ~400 clinical trials currently incorporate some kind of wearable device.
The emergence of wearables provides clinical trial sponsors with the means to generate significant amounts of patient-generated RWD between site visits. Additionally, wearables offer the opportunity to collect data for novel endpoints and supplemental data that may improve the regulatory case or make the case for reimbursement more compelling. In addition to phase I trials, there is a growing demand for the use of wearables in phase IV studies.
As a result, there continue to be a number of collaborations between life science companies and traditional technology companies such as Apple, Verily (Google), and Amazon, as well as life science-focused companies that are developing platforms to integrate sensor data like Koneska Health, Validic, and Human API. For example, Apple has developed ResearchKit, which is open source software that allows researchers to create mobile apps supporting efficient data collection specialized to a therapeutic area. CareKit, another open source framework provided by Apple, allows for the development of apps to assist patients to manage their own healthcare more efficiently.
On top of these wearables and biosensors are digital biomarkers, which are patient-generated physiological and behavioral measures collected through connected digital tools (hardware and software) that can be used to explain diseases, influence fitness behavior, predict drug response, and/or anticipate health-related outcomes. For example, Omron’s HeartGuide, the first wearable blood pressure device, recently announced it will integrate with PhysIQ‘s pinpointIQ analytics platform to indicate potential cardiovascular risk.
Other companies developing digital biomarkers include Evidation Health, whose platform creates novel digital biomarkers and analyzes real-world behavioral data to quantify health outcomes, and ActiGraph, which provides medical-grade wearable activity and sleep monitoring solutions for the global research community. In addition, AliveCor, has developed an FDA-approved six-lead electrocardiogram (ECG) device called KardiaMobile that jackets a standard iPhone and is used to detect atrial fibrillation, bradycardia, tachycardia, or normal heart rhythm. The company’s new spokesperson, Olympic gold medalist swimmer Mark Spitz, has atrial fibrillation and uses KardiaMobile to monitor his heart.
Pharma companies are also using mHealth technology for lab testing in clinical research. The most notable is U.K.-based genetic sequencing platform provider Oxford Nanopore Technologies with its MinION, a portable nanopore device that sequences DNA/RNA in real time. The device, which runs from a USB flash drive, is being used in clinical research to develop novel diagnostic or monitoring tools, drugs that address specific biological pathways, and companion diagnostic tests designed to ensure that drugs are targeted at the correct subgroup of patients.
TABLE 28: The MinION
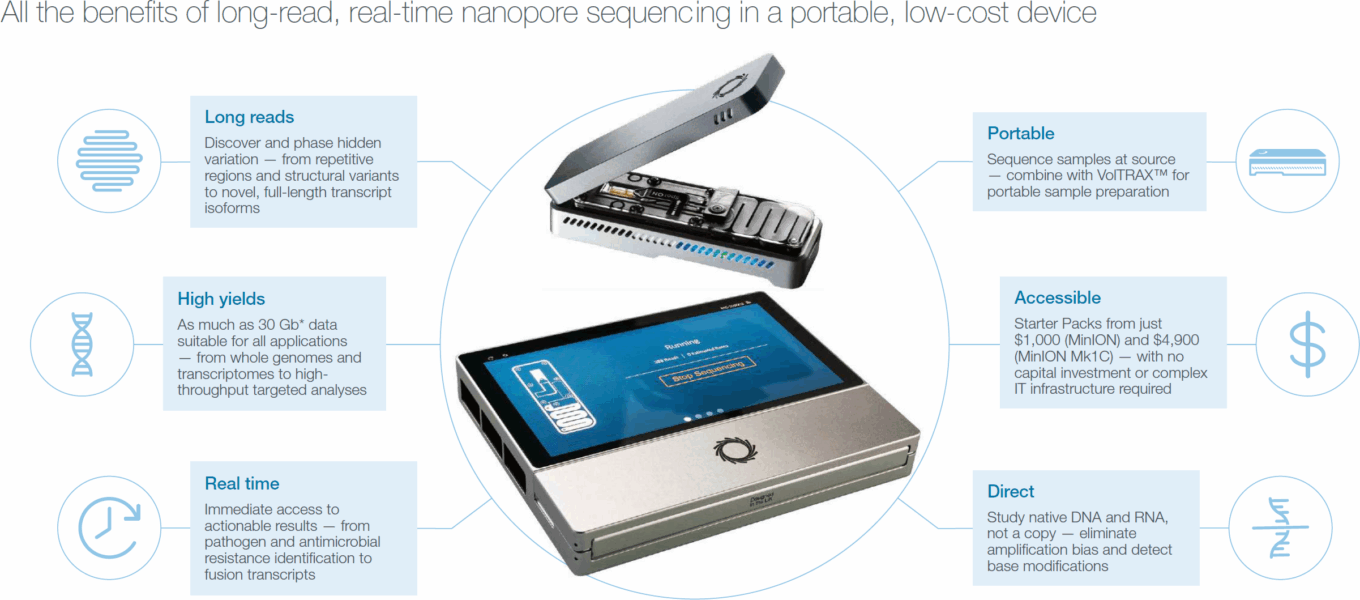
Source: Oxford Nanopore Technologies.
Digital therapeutics (DTx)
Building on the foundation that digital sensors and biomarkers provide as well as the need to demonstrate value in the eyes of payers, pharma companies are expressing interest in a new area called digital therapeutics (DTx). DTx software connects with wearables, biosensors, cellphones, tablets, or durable medical equipment. Although the software appears similar to consumer wellness apps, DTx solutions are focused on improving patient outcomes through diagnosis, treatment, and coordination of care.
The main difference between the various DTx solutions is the strength of their claims. For example, digital services DTx solutions typically aim to modify patient behavior in some way. Companies that offer these DTx solutions will often publish studies that show they help drive a clinical outcome, but they don’t claim a specific therapeutic benefit. A well-known DTx that falls under this tier is Omada Health’s digital behavior coaching platform that aims to help patients reduce their risk of diabetes and other chronic diseases.
The next tier of DTx solutions, adjunctive DTx, are used in conjunction with a drug treatment regimen to enhance effectiveness and adherence but stop short of claiming a therapeutic benefit. Adjunctive DTx solutions offer pharma a companion software tool that allows it to differentiate itself from the competition. Additionally, they can help extend product lifecycles, differentiate products in development, actively engage patients, and gather RWE.
Companies offering the third tier of DTx technology, digital drug replacements, seek to provide a clinical benefit through the digital technology itself and not through any other source. Because of this, they must perform randomized controlled trials and gain FDA approval prior to use. A number of pharmaceutical and medical device companies see the opportunities and benefits DTx can provide. As a result, several DTx companies have attracted investors and/or partners from the sector.
For example, Roche purchased a digital diabetes management platform in 2017, and in January 2019, Otsuka signed a deal with Click Therapeutics worth up to $300 million to develop and commercialize a digital therapeutic solution to treat major depressive disorder (MDD). In 2018, ResMed acquired Propeller Health for $225 million to leverage its sensor-enabled digital platform to serve patients across the chronic respiratory disease spectrum. In 2018, Novartis and its Sandoz subsidiary invested and partnered with Pear Therapeutics to develop and commercialize digital therapeutics for the treatment of psychiatric disorders. In November 2018, Sandoz and Pear launched their reSETapp, which is used to treat substance abuse disorder. reSET is the first software-only therapeutic cleared by the FDA.
Medication adherence & compliance
One area of the patient journey that deserves attention and that data applications are attempting to address is low medication adherence. Low medication adherence is a major problem driven by several factors, including complicated prescriptions, negative side effects, high costs, a patient’s physical or cognitive impairments, lack of transportation services to pharmacies, low health literacy, and patient behavior such as forgetfulness and procrastination.
According to the World Health Organization, medication adherence can have a more direct impact on patient outcomes than the specific treatment itself. Medication adherence can affect quality and length of life, health outcomes, and overall healthcare costs. According to industry reports, patients who don’t take their medications as prescribed can annually account for up to 50% of treatment failures, around 125,000 preventable deaths, up to 25% of hospitalizations in the United States, and ~$300 billion in preventable healthcare costs. Additionally, studies have shown 20-30% of medication prescriptions are never even filled and that 50% of medications for chronic diseases, such as high blood pressure, are not taken as directed. Medication non-adherence costs the pharmaceutical industry $640 billion in lost revenue each year worldwide and $250 billion in the United States alone.
TABLE 29: Why patients skip doses
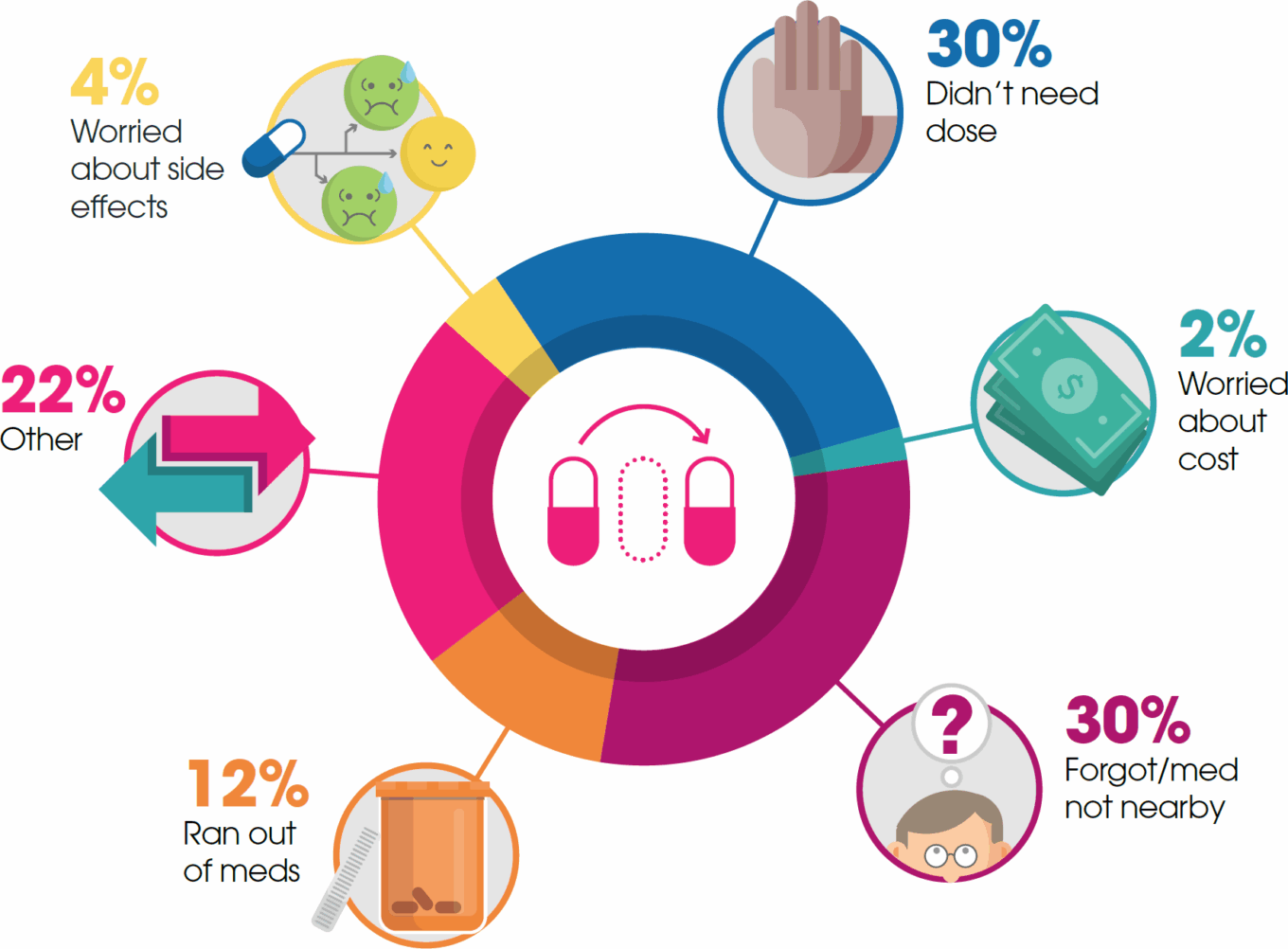
Source: Medisafe.
There is no one-size-fits-all approach to non-adherence, and a number of data-driven vendors have emerged to address it. Notable examples include AiCure, a mobile app that leverages a visual recognition platform that confirms when patients ingest their medication, AllazoHealth via its AllazoEngine, which combines behavioral sciences and AI to predict which patients are at-risk for non-adherence, trUStr, which utilizes an AI-powered chatbot to engage and motivate patients to improve adherence, and Medisafe, a cloud-based medication adherence and compliance platform with ~5 million registered users worldwide and over 2 billion doses managed that personalizes content such as educational videos, coupons, and motivational messages and provides pharma clients with real-time insights into patient behavior.
Another vendor, OptimizeRx, uses cloud-based software applications that are directly integrated into electronic health record (EHR) workflows to drive adherence and compliance by providing patients with a HIPAA-compliant mobile messaging platform that allows pharmaceutical companies to directly engage with patients. Features include information regarding cost-saving and co-pay programs, medication reminders, refill notices, and explanations of side effects.
TABLE 30: Automated patient engagement

Source: OptimizeRx.
Another solution trying to tackle the medication adherence and compliance problem is gamification. Gamification in patient care involves extracting the elements of gameplay that patients find compelling and using them to drive adherence to therapy and positive behavior outside of a game context. The goal of gamification in the pharmaceutical industry is to turn routine tasks associated with medication into something that brings a moment of joy to patients – treating them as active participants in their health rather than passive patients.
One company we see leading the way in patient gamification is HealthPrize, whose platform uses gamification, behavioral economics, and loyalty marketing to improve patients’ medication adherence. HealthPrize’s web and mobile app-based patient engagement software gamifies drug adherence and the refilling of prescriptions in a way that motivates patients both extrinsically (i.e., a tangible, physical reward) and intrinsically (i.e., intangible, self-gratifying). Instant rewards create a positive experience, reinforcing drug adherence with targeted content at every dose and refill. Game-derived elements include a monthly leaderboard competition and weekly drawing. For example, when patients adhere to their doses for three weeks, they can earn points on the platform that can then be traded in for real-world prizes like gift cards, health and fitness-related merchandise, or charitable donations. According to the company, the HealthPrize platform increases average adherence 52% over a drug’s baseline rate.
Patient communities & social media
In our previous whitepaper, eClinical Solutions: Streamlining the introduction of new drugs and medical technology, we highlighted the importance of patient-reported outcomes (PROs) in clinical trials and how they are being integrated electronically to drive more accurate and cost-effective measurements of patient well-being. Electronic PROs (ePROs) usually come in the form of digital questionnaires. They are widely used for tracking urinary and vasomotor symptoms, vomiting in infectious disease trials, and pain, nausea, and tiredness in cancer patients. ePROs are easier for patients to complete than traditional questionnaires, improve the quality of data, and increase patient engagement.
Outside of clinical trials, ePROs are collected from a variety of sources, including patient registries, wearable devices, and social media, both to drive research and for commercialization. The integration of ePROs into prospective data capture is, however, resource intensive, and maintaining patient engagement can be a challenge, particularly for patients who suffer from rare diseases and their caregivers. Rare disease patients often require the coordination and collaboration of several different HCPs, extensive treatment plans, and continuous monitoring throughout their medical journey. As a result, the data generated tends to be overwhelming for patients and their caregivers to manage. That being said, this data can be pivotal in helping understand in real time how a patient feels and functions.
One innovative company that is making inroads in helping patients manage their health journey is Backpack Health. Backpack Health has created a mobile health platform for individuals and families to share and manage their medical journey. Recently, the company announced users can automatically integrate their EMRs (with their consent) into their personal backpack accounts. This new functionality allows users to connect and update health records of their choice. Additionally, via surveys and natural history data, Backpack’s platform enables medical foundations, pharma companies, and advocacy groups to collect meaningful data as well as build and support branded communities based on the needs of their members/patients.
Much as it is for physicians, social media has quickly become a go-to place for patients to seek out and share information, either in specific patient communities such as Patients-LikeMe (recently acquired by UnitedHealth), Health Union, Inspire, WEGO Health, and Liquid Grids or on broader social media sites such as Twitter, YouTube, Instagram, and Facebook. According to a recent survey from Inspire, 88% of patients turn to social media for support, while 41% of social media users have asked for or received advice related to health. As a result, having relevant content is important for patient engagement. One company meeting this demand is Health Union, which, through its 25 condition-specific online health communities, publishes original content on a daily basis that cultivates patient relationships with the goal of helping chronic disease sufferers find the information and/or connections they seek.
In addition to relationship building, social media offers pharma an unfiltered view of patients who often describe experiences differently than clinicians would. This, in turn, has generated a potentially rich but analytically messy source of RWD. The ability to harness this data has been assisted in recent years by the application of advanced analytics. Approaches such as NLP, along with machine learning, address the complexities of data extracted from social media, such as multiplicity of terms, duplicate posts, misspellings, and abbreviations. For example, Liquid Grids offers pharma clients a healthcare-focused social media intelligence platform designed to aggregate and identify healthcare dialogue across the web using advanced NLP. The Liquid Grids Social Health Intelligence platform enables pharma clients to follow patient conversations about a particular disease, condition, treatment, or procedure while providing insights into patient personas.
Hub services
The proliferation of specialty drugs over the last 25 years has created unique challenges for pharmaceutical manufacturers looking to obtain dispensing, adherence, and switching data. These drugs require more complicated administration and also cost significantly more than traditional drugs. Only a handful of specialty drugs were available in the mid-90s. By 2009, specialty drugs accounted for 26% of net U.S. medicines spending, and in 2018, specialty spending share hit 50%. Additionally, 39 specialty therapies were approved by the FDA in 2018, and it is projected that from 2019-2023, 65% of newly launched medications worldwide will be considered specialty. This includes a wave of new targeted immunotherapies and gene and cell therapies that are expected to come to market.
TABLE 31: Real net per capita medicine spending and growth by product type (U.S. dollars)
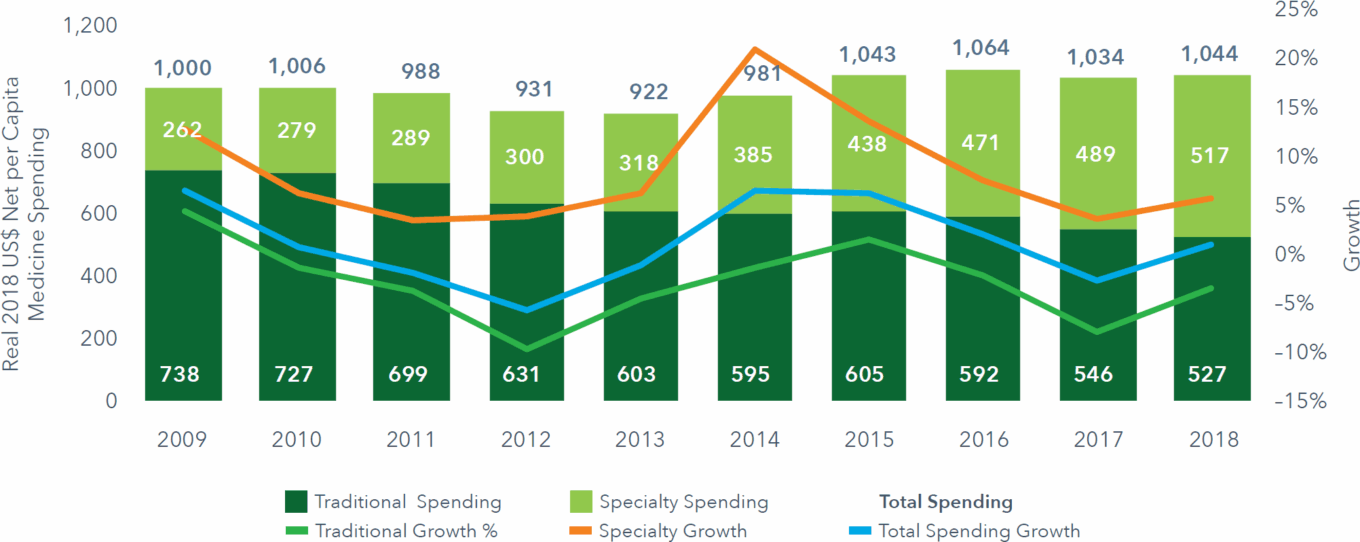
Source: IQVIA.
While there are no established criteria for what makes a drug a ‘specialty’ medicine, most stakeholders look for a combination of characteristics. Factors may include the drug’s clinical use to treat chronic, rare, or complex diseases (multiple sclerosis, Hepatitis C, oncology, etc.); a requirement for special handling or administration (injection, infusion, cold storage, etc.); availability via exclusive, restricted, or limited distribution; special patient monitoring for efficacy or safety; or high cost (i.e., greater than $6,000 per year). Stakeholders agree that cost is an important factor, and for some, it is the only factor that determines specialty designation. Thanks to the proliferation of specialty drugs, hub service providers have emerged to help streamline the specialty drug prescription process.
Hub service providers, also known as “wraparound service” providers, help get patients on therapy and manage their journey once on therapy. In the early 2000s, hubs helped patients and HCPs obtain permission to use specialty drugs and with reimbursement. They are now tech-enabled service platforms that include data collection and reporting, prior authorization and appeals programs, patient assistance programs, quick start and bridge programs, risk evaluation and mitigation strategies (REMS), patient education, and e-prescription routing.
TABLE 32: Hub services offerings
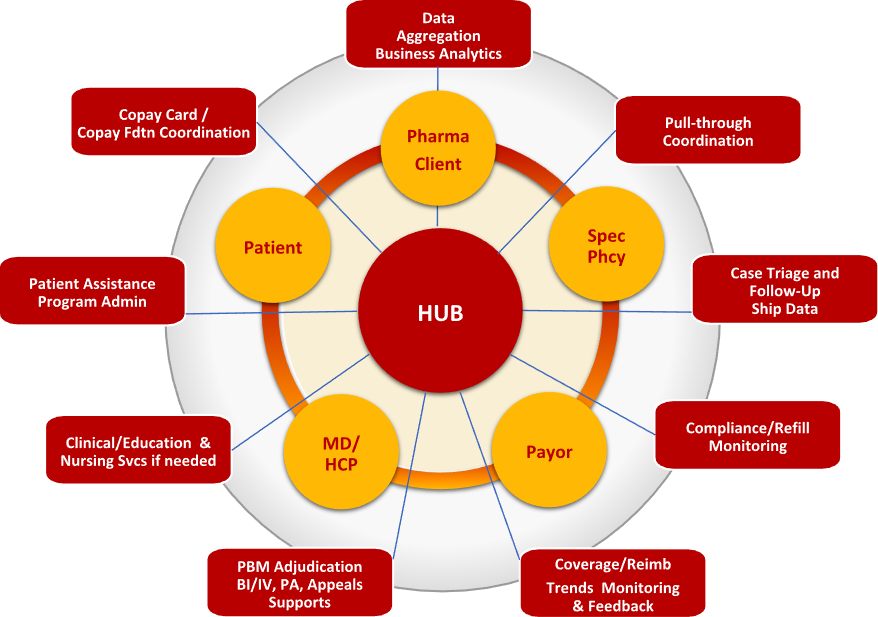
Source: CareMetx.
While some pharmaceutical companies create and manage their own hub programs, most contract with third-party hub providers as they recognize the risk of potential HIPAA, Anti-Kickback Statute, and False Claims Act violations. According to a 2018 Helio Health survey on the mix of insourced and outsourced services, hubs provide a wide and growing range of services but are most frequently used to manage patient onboarding, benefit verification, and prior authorization. Offering a direct connection with patients, hubs may take on a greater role in delivering patient education, particularly around the cost of therapy and available assistance programs. Often, a patient’s first in-depth conversation about the cost of a therapy happens on a phone call with the hub. Insourcing is highest for patient education, patient assistance programs, and copay assistance. The trend is for activities involving financial support, such as copay programs, to be managed by an outsourced provider.
Although there are pros and cons to insourcing and outsourcing hub functions, the ~$5 billion annual market for outsourced hub service providers is expected to grow in lockstep with the growth in specialty drugs and continue to be an essential component in commercialization. Hubs are full of valuable data for pharma because they can provide a holistic view of the patient journey that involves key interactions between patients, providers, and payers. Additionally, data leveraged from hub-based patient interactions can provide pharma with a better understanding of treatment trends and potential hurdles to medication adherence. As a result, hubs have focused more on software platforms that emphasize data collection and analytics. Another driver is the current trend toward value-based contracting. Hub providers are evaluated on how well they manage patients, so the software platform is a critical component to the business. Notable proprietary software platforms include bAsembia1, Cardinal Health’s Connect-Source, TrialCard’s QuickPath, CareMetx’s CareMetx Connect, and CoPilot’s Salesdoor.
TABLE 33: The U.S. pharmacy distribution and reimbursement system for patient-administered, outpatient brand-name drugs
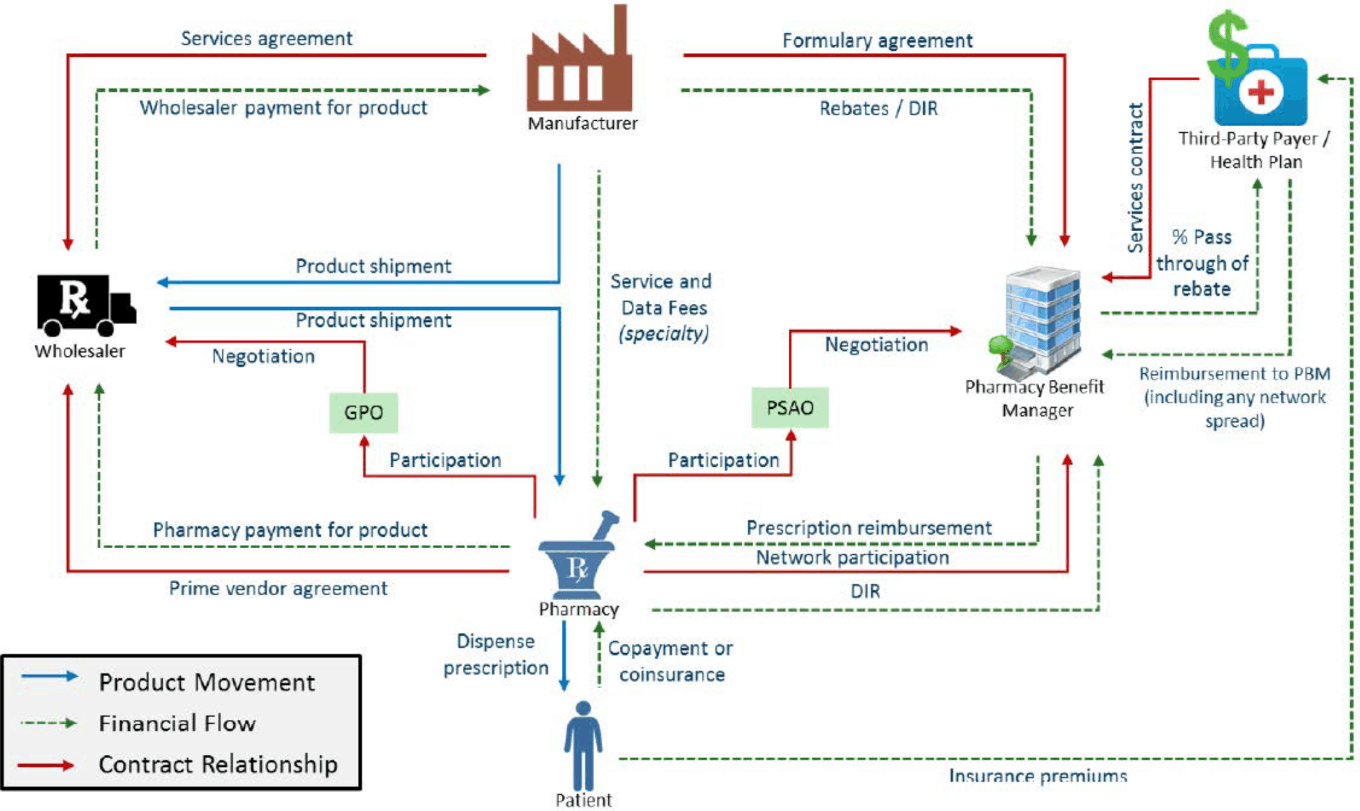
Source: Fein, Adam J., The 2019 Economic Report on U.S. Pharmacies and Pharmacy Benefit Managers, Drug Channels Institute, 2019.
Other key areas within hub services where digital technology has been adopted are prior authorization (PA) and benefit verification (BV). All hub providers are positioned to handle BVs and PAs, which are critical services for overworked, time-constrained physicians’ offices. Some hub providers have developed their own database of necessary forms and contacts with insurers; others rely on outside vendors like Surescripts to do the process electronically.
The leader in the space is CoverMyMeds (now owned by McKesson), which has linkages to 700,000+ HCPs, 94% of payers, 96% of pharmacies, 500+ EHR systems, and 200+ pharma brands. Traditionally, PA was a paper-based, time-consuming process and was accomplished through multiple faxes and phone calls between providers, pharmacists, and payers, all while keeping the patient waiting. To solve this problem, CoverMyMeds developed an automated process by creating online PA forms to be filled out, then securely and electronically transmitted and tracked within the clinical workflow.
Another innovative hub services provider leveraging technology is ConnectiveRx. Its new BrandHub offering is a web-based portal with features that include electronic benefit verification (eBV), electronic prior authorization (ePA), in-office product tracking for HCPs, specialty pharmacy fulfillment tracking, EMR integration with HCP portals, and tracked eOrdering that enables HCPs to initiate therapy quickly. Additionally, BrandHub provides pharma companies with the ability to add all of their brands to a single prescriber portal.
The growth of specialty drugs has also given rise to specialty pharmacy providers. Specialty pharmacy providers offer services that mirror those of traditional hubs, such as benefits verification and prior authorization support. Two specialty pharmacy technology vendors we see bringing differentiated solutions to the marketplace are IntegriChain and KloudScript.
Engaging payers
With rising healthcare costs, reimbursement is becoming more difficult to obtain as both commercial and government payors are demanding more evidence of a drug’s potential cost-effectiveness in the real world. This is reflected in the emergence of outcomes-based payments models. Given the complexities surrounding reimbursement, pricing and contracting are critical drivers in the launch-planning process of a new drug or the expanded indication of an already commercialized drug. This is especially true for innovative drugs such as biosimilars and injectables. In addition to payors, regulators such as the FDA are increasingly requiring drug sponsors to demonstrate the economic value of new treatments prior to approval.
The FDA may also require implementation of a REMS when it determines it is necessary to ensure, through the accumulation of additional evidence, that the medicine’s benefits outweigh its risks over time. A REMS can be required before or after FDA approval and can apply to one drug or a class of drugs. For example, a REMS may outline specific safety procedures for HCPs before dispensing a drug, such as patient education on the warning signs of infection.
In 2016, Congress passed the 21st Century Cures Act, which places more emphasis on the use of RWE to support regulatory decision-making. In 2017, the FDA issued draft guidance concerning RWD and RWE, “Use of Real-World Evidence to Support Regulatory Decision-Making for Medical Devices.” The objective of the guidance is to clarify how the agency evaluates RWD to determine if it can be used to generate the type of RWE the FDA needs to make decisions about medical devices. In December 2018, the FDA released its Framework for FDA’s Real-World Evidence Program, which “created a framework for evaluating the potential use of real-world evidence (RWE) to help support the approval of a new indication for a drug already approved under section 505(c) of the FD&C Act or to help support or satisfy drug post-approval study requirements.”
The changing regulatory and reimbursement landscape has led to the growth and use of RWE studies to address blind spots that randomized controlled trials might miss, such as potential benefits and adverse events. Health economics and outcomes research (HEOR) and RWE are important pieces of the puzzle because of the pivotal role they play in evidence development, treatment access, reimbursement coverage, and overall commercialization.
TABLE 34: HEOR evidence and product lifecycle
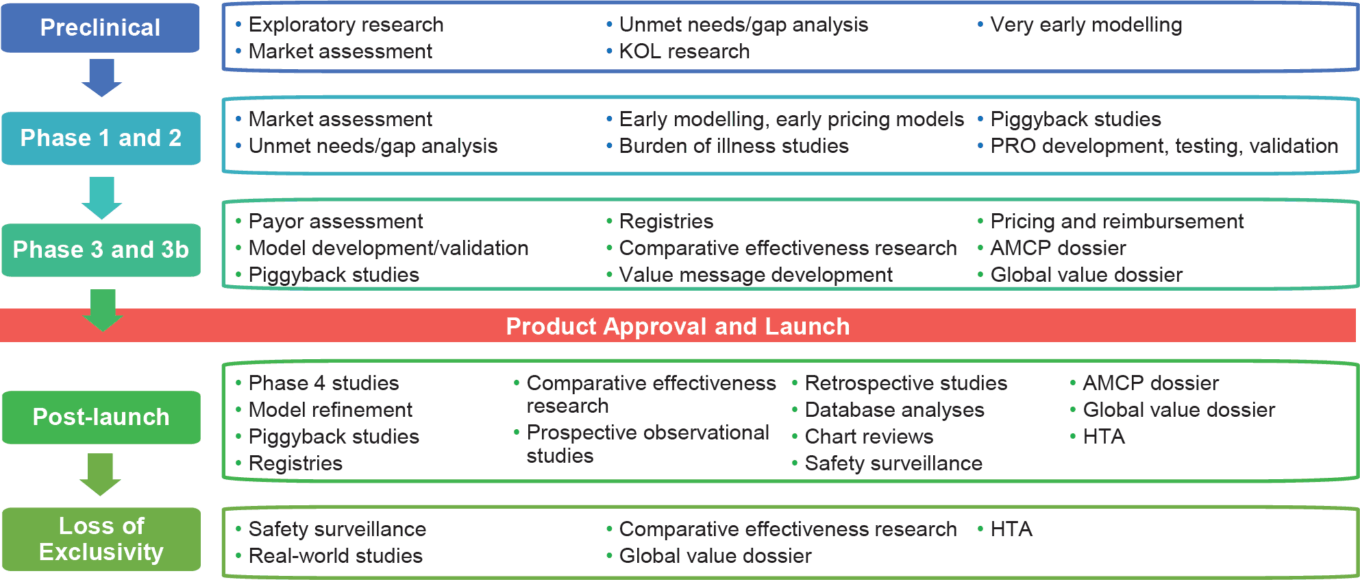
Source: Pharmerit.
Outcomes projects on market access, value evidence generation, and commercial strategy are costly. According to IBM, projects can cost $1.1 million on average and can range from $100,000 to $6 million. Additionally, data scientists and HEOR specialists are in limited supply, which has led to a significant use of outsourcing to trusted third-party partners.
According to a 2018 survey jointly conducted by HealthEconomics.com and Scientist.com, greater than 71% of companies outsource more than half their HEOR and RWE projects. There are primarily two types of partnerships in the industry: contract research organizations (CROs) collaborating with data and analytics service providers to provide greater RWE and outcome analytics and pharma companies collaborating with providers for predictive analytics and data solutions.
In the case of CROs, companies like IQVIA, Syneos Health, Parexel, Covance, ICON, and PPD are trying to improve their value proposition to pharma by offering a one-stop shop for clinical research and commercial services such as business intelligence, forecasting, market research, HEOR, RWE, market access, sales-force effectiveness, and marketing.
In the case of data analytics providers, we see vendors like GNS Healthcare making inroads in the space. GNS’s AI-based REFS (reverse engineering, forward simulation) platform leverages RWD to provide insights into drug performance during both clinical trials and post launch to accelerate development and to define a product strategy and value proposition earlier in the clinical trial phase, demonstrate market differentiation during a drug’s growth phase, expand indications, and provide evidence for lines of therapy.
An up-and-coming vendor, Analytical Wizards, offers IZE, an AI platform of customizable modules for marketing, sales, market access, HEOR, clinical development, and R&D teams. The IZE HEOR Evidence Generator module provides market access teams with early outcome indicators for an individual drug or treatment combination across therapeutic areas. Other notable vendors include MMIT, Cota, Medimix (recently acquired by Evidera, a business unit of PPD), OM1, Syapse, Flatiron Health (owned by Roche), TARGET Pharma Solutions, inVibe Labs, and Inovalon.
Featured company profiles
CRM
QPharma, founded in 1994, is a provider of system validation and compliance consulting services. QPharma evolved from several precursor organizations founded by Patrick P. Den Boer, who remains the company’s president and CEO. The company enjoyed success validating pharmaceutical sample management systems before developing its own system in 1999. In 2006, QPharma opened its own accredited sample fulfillment center. In 2016, the company rebranded its flagship suite of commercial and compliance software tools – which by then included modules for key opinion leader engagement, product launch management, field sales force training, and physician payments transparency reporting – as Titanium. Today, Titanium is a fully cloud-based, SaaS platform recognized by the industry as state-of-the-art. Most recently, QPharma introduced Ti CRM, a full-featured customer relationship management tool for the field sales force. The company is headquartered in Morristown, N.J.
StayinFront provides a complete, PDMA-compliant solution that automates the execution, management, and monitoring of sales and marketing activities. The solution allows sales and management teams to target, prepare, and execute business plans, while actionable analytics guide and support data-driven decisions. StayinFront makes it easier for primary care, specialty care, managed care, institutional sales, and medical sciences liaisons teams to manage their territories, while enabling sales managers to coach, measure, and respond to teams for better results. Since its inception in 2000, this Fairfield, N.J.-based company has deployed its SaaS technology in over 50 countries and in over 25 languages.
Synergistix, founded in 1997 and headquartered in Sunrise, Fla., is a provider of CRM solutions and other services for life sciences companies. Its Customer Analysis and Targeting System (CATS) Software Suite combines field-based data capture, CRM, management reporting, and sample accountability services to deliver a PDMA-compliant solution for automating, managing, and monitoring sales force activity.
TikaMobile, based in New York City, is a provider of cloud-based business intelligence and CRM software for the life sciences industry. The company’s product suite streamlines the sales and marketing activities for launching and growing drugs and medical devices. Applications include targeting and customer segmentation, call planning, CRM, customer business review, forecasting, and case tracking. Tika has offices in the United States, Europe, and India.
Veeva Systems, founded in 2007 and headquartered in Pleasanton, Calif., is a cloud-based software provider for the life sciences industry. The company started out primarily as a customer relationship management (CRM) software provider built on top of the Salesforce platform. It now has two pillars of its core business, Veeva Commercial Cloud and Veeva Vault. Commercial Cloud is an interoperable solution that brings together customer data, multichannel interactions, and compliant content to help life sciences companies achieve commercial excellence. Veeva Vault is a cloud-based content management solution with built-in collaboration features designed specifically for the life sciences industry. It consists of multiple modules that independently perform separate functions. These modules power clinical trials, regulatory submissions, product safety programs, and quality assurance. Prior to its IPO in 2013 (NYSE: VEEV), the company raised $7M from Lead Edge Capital and Emergence Capital Partners.
MDM/reference data
HealthLink Dimensions, based in Atlanta, provides a suite of compliance-grade data solutions and services that help pharmaceutical, biotech, and medical device marketers better identify their target audience and effectively reach healthcare professionals across multiple channels. Marketers are able to extend their reach to healthcare professionals by leveraging the company’s Healthcare Provider Database, which includes more than 2.7 million healthcare providers.
MedPro Systems, headquartered in Mt. Arlington, N.J., provides healthcare license solutions for the pharmaceutical and medical device industries based upon a powerful combination of licensing data and information technology. The company’s MedProID database contains 22 million healthcare practitioner records and 800,000 healthcare organization records. The company has been in business for 19+ years and has over 500 active life sciences customers, including 22 of the Top 25 pharmaceutical manufacturers.
Reltio, headquartered in Redwood City, Calif., is a provider of data-driven applications designed for modern data management through a Platform as a Service (PaaS). The company’s data-driven application delivers enterprise applications with reliable master data management (MDM), big data insights, and intelligent recommended actions, enabling companies to access and collaborate on reliable, relevant data for better planning, customer engagement, and risk management. ~70% of the company’s clients are in the life sciences industry. In 2019, the company was named to the Inc. Magazine 5000 list of the nation’s fastest-growing companies. It posted 2018 revenue of $37.8 million and three-year growth of 316%. The company was founded in 2011.
RxS, based in Manalapan, N.J., provides end-to-end, multi-channel sample management solutions for pharmaceutical and life science organizations. RxS aims to remove the complexity and administrative burden of managing the regulated environment for clients. Between its two core services, LinkedRx and TeleTargetRx, clients are provided with a holistic, integrated platform to manage sales force automation, sample channels, and process disparity. This unified system approach allows RxS to work hand in hand with organizations across all of their regulatory and compliance concerns and streamline the necessary administrative tasks that have long burdened industry leaders.
Real world data
Life Image delivers real-world data capabilities to healthcare providers, life sciences, and artificial intelligence platforms globally, leveraging a broad network of hospitals, medical devices, and solution providers. Its Real World Imaging offering provides large-scale, heterogeneous, de-identified imaging sets that are linkable to other longitudinal data. Founded in 2008 and headquartered in Newton, Mass., Life Image’s digital platform is connected to 10,000 facilities with more than 150,000 providers in the United States and 58,000 global clinics. The company has raised ~$95 million to date and counts Echo Health Ventures, Cambia Health Solutions, Galen Partners, and Zaffre Investments among its investors.
NeoGenomics, headquartered in Fort Myers, Fla., and founded in 2001, is a publicly traded independent medical laboratory company specializing in genetic and molecular testing for cancer. The company provides a range of genetic testing services to two primary types of customers, clinical and biopharmaceutical and research. Clinical customers include community-based pathology practices, oncology groups, hospitals, and academic centers. Biopharma and research customers include pharmaceutical companies to whom NeoGenomics provides testing to support their studies and clinical trials.
Optum, headquartered in Eden Prairie, Minn., and founded in 2011 as part of the UnitedHealth Group family of businesses, is a provider of technology-enabled healthcare services. Through its three primary business units – OptumRx, OptumInsight, and OptumHealth – Optum leverages its core competencies in data analytics, pharmacy care services, population health management, healthcare delivery, healthcare operations, and advisory services to work with a diverse set of clients, including payers, healthcare providers, employers, governments, life sciences companies, and consumers. Since its founding, Optum has grown to serve more than 67,000 pharmacies, 127 million individuals, and 100,000 physician practices and other healthcare facilities, as well as 127 million individual consumers.
Truven Health Analytics is an IBM Watson Health company that offers healthcare data and analytics services. It provides information, analytic tools, benchmarks, research, and services to hospitals, government agencies, employers, health plans, clinicians, pharmaceutical companies, biotech, and medical device companies. Truven provides an array of products and services to optimize performance, improve member health, ensure regulatory compliance, increase employee engagement, and reduce healthcare costs. Truven Health Analytics began as part of Thompson Corporation before becoming a standalone company in 2012 and being acquired by IBM Watson for $2.6 billion in 2016.
AI in drug discovery
Arpeggio Biosciences, founded in 2017 and based in Boulder, Colo., offers a drug assessment platform that combines machine learning and RNA sequencing to identify new druggable targets and molecular markers of disease affected by a drug. The platform enables medical scientists to identify new drug targets and improve the success rate when customers advance new drugs into the clinic. The company has raised $2.1 million in venture capital from FundersClub, Khosla Ventures, and Y Combinator.
Atomwise, founded in 2012 and based in San Francisco, develops artificial intelligence systems using powerful deep learning algorithms and supercomputers for drug discovery. The company was formerly known as Chematria Inc. and changed its name to Atomwise in 2015. Atomwise has raised over $50 million from leading venture capital firms, including Baidu Ventures, Dolby Family Ventures, Tencent Holdings, Monsanto Growth Ventures, Vickers Venture Partners, OS Fund, Kosla Ventures, and Y Combinator.
Auransa, founded in 2014 and based in Palo Alto, Calif., is an AI-driven biotechnology company developing precision medicines. The company’s SMarTR Engine, which leverages machine learning, advanced analytics, and mathematics in an AI framework, generates insights from molecular data for a deep understanding of disease biology and patient subtypes. This information, when paired with the company’s breadth of in-house drug discovery and development expertise, drives the identification of novel compounds designed to address significant unmet medical needs for clinically meaningful disease subtypes. Investors include Lux Capital Management, 11.2 Capital, AME Cloud Ventures, Data Collective, and Medivation.
BenevolentAI, founded in 2013, is a developer of artificial intelligence and computational medicine technologies intended to change the way drugs are designed, developed, tested, and brought to market. The company’s computational and experimental platform for drug discovery seeks to improve patient lives by applying technology designed to generate better data decision-making and in doing so lower drug development costs, decrease failure rates, and increase the speed at which medicines are generated. The Benevolent Platform, used by over 200 BenevolentAI scientists and technologists, ingests molecular, clinical, pharmacological data, and scientific literature to derive contextual relationships across all relevant biomedical entities, leading to the proposal of novel and optimal drug targets. The platform supports scientists in the design and optimization of the right drug molecule for a unique patient population. BenevolentAI is headquartered in London with a research facility in Cambridge (U.K.) and offices in New York. BenevolentAI has active R&D drug programs in disease areas such as ALS, Parkinson’s, Ulcerative Colitis, and Sarcopenia. The company has raised over $360 million since inception and has an estimated post-money valuation of $1 billion. Notable investors include Temasek Holdings, Woodford Investment Management, Lansdowne Partners, Lundbeck, and Upsher Smith Laboratories Inc.
BERG, founded in 2006 and based in Framingham, Mass., is a clinical-stage, artificial intelligence-powered biotech company leveraging its proprietary platform, Interrogative Biology, to map disease and revolutionize treatments across oncology, neurology, and rare diseases. By taking a “back to biology” approach, BERG is able to identify critical biomarkers that can accelerate the discovery and development of treatments aimed at the most promising therapeutic targets and pathways. BERG has leveraged both Interrogative Biology and traditional R&D methods to develop a robust pipeline of first-in-class product candidates and diagnostics that advance bold innovations to improve patient lives. The company is backed by Carl Berg, its co-founder, chairman, and principal investor.
Exscientia, founded in 2012 and based in Oxford, U.K., is applying AI and big data processing to accelerate drug discovery and development in complex disease areas. Exscientia’s AI-driven systems actively learn from experimental results. They identify and assimilate multiple subtle and complex compounds and formulations, balancing potency, selectivity, and pharmacokinetic criteria. This leads to new opportunities for the treatment of complex diseases where the target mechanisms are often unknown. The company has received $26 million in funding from investors that include Celgene, GT Healthcare Capital Partners, Evotec, and Frontier IP Group.
Genomenon is a developer and provider of genomic data interpretation software that improves disease diagnosis and treatment. The company’s flagship product, the Mastermind Genomic Search Engine, is used by hundreds of genetic labs worldwide to accelerate diagnosis, increase diagnostic yield, and assure repeatability in reporting genetic testing results. Genomenon licenses its Mastermind Curated Genomic Datasets to pharmaceutical and biopharma companies to inform precision medicine development, deliver genomic biomarkers for clinical trial target selection, and support CDx regulatory submissions with empirical evidence. The company was founded in 2014 and is based in Ann Arbor, Mich. Genomenon has raised ~$5 million in seed funding from a base of investors that includes Invest Detroit, IrishAngels Ventures, Michigan Biomedical Venture Fund, Invest Michigan, Three Leaf Ventures, Invest Detroit Ventures, and Danmar Capital.
Insilico Medicine is an artificial intelligence company headquartered in Hong Kong, with R&D and management resources in six countries sourced through hackathons and competitions. The company utilizes innovative AI solutions for drug discovery and aging research, as well as deep learning technology for drug repurposing for aging and age-related diseases, allowing for the identification of molecules that may be effective against a variety of cancers, as well as metabolic, cardiovascular, and central nervous system diseases. Since 2015, Insilico Medicine has developed a broad range of generative adversarial networks (GANs) and reinforcement learning approaches to identify novel protein targets, generate novel molecular structures with specified properties, and generate synthetic data. Insilico has raised $61 million since inception and counts Baidu Ventures, F-Prime Capital Partners, Qiming Venture Partners, Deep Knowledge Ventures, Juvenescence, Pavilion Capital, and Bold Capital Partners among its base of investors.
Numerate, headquartered in San Bruno, Calif., is a developer of a drug engineering platform designed to transform small molecule drug discovery. Its novel AI-based software platform is designed to overcome small molecule chemical design challenges to increase speed and success while reducing cost. The company was founded in 2007 and has raised $19 million since inception. Notable investors include Lilly Ventures, Lanza Tech Ventures, and Atlas Venture.
Owkin, founded in 2016 and headquartered in New York City, is an artificial intelligence company that has developed software that turns real-world patient data into predictive models, enhancing the research capabilities of physicians and pharmaceutical companies as well as patient treatment, drug discovery, and development. Owkin has raised $31 million since inception from investors that include F-Prime Capital Partners, Eight Roads, Cathay Innovation, and GV.
Phenomic AI is a Toronto-based company whose AI-based platform is designed to discover and optimize therapeutics directly against phenotypic assays, enabling pharmaceutical companies to discover drugs in an easier way. The company was founded in 2017 and has raised ~$2 million since inception. Investors include Cantos Ventures, Garage Technology Ventures, Hemi Ventures, Radical Ventures, Hike Ventures, and Inovia Capital.
Qrativ is a joint venture formed by the Mayo Clinic and nference to identify treatments for rare diseases using a drug purposing approach. The company has developed a proprietary drug purposing platform, Darwin.ai, that leverages proprietary data from the Mayo Clinic (EHRs, prescription & diagnosis data, patient-treatment omics) and the nference knowledge synthesis AI platform (including data extracted from PubMed and clinical trials). Starting in the early stages of drug development, Qrativ partners can leverage Darwin.ai to search for all possible uses of a drug candidate, identifying potential rare disease indications and finding subsets of patients who may respond favorably to a given candidate. The company was founded in 2017 and is based in Cambridge, Mass. It has received ~$8 million in venture capital funding from Matrix Capital Management, Matrix Partners, and Mayo Clinic Ventures.
ReviveMed is an artificial intelligence-driven drug discovery platform provider. Based in Cambridge, Mass., ReviveMed’s platform is designed to overcome the difficulties of identifying a large set of metabolites for each patient. The AI platform combines the data from small molecules with other molecular data and translates it into novel therapeutic insights for drug discovery. Currently, ReviveMed is collaborating with pharmaceutical companies and pursuing internal drug discovery, with its initial focus on metabolic diseases, including non-alcoholic fatty liver disease. The company has raised ~$1.5 million in seed capital from TechU, Rivas Capital, Good Growth Capital, and Team Builder Ventures, among others.
TwoXAR, founded in 2014 and based in Mountain View, Calif, is an artificial intelligence-driven drug discovery company. It leverages its computational platform to identify promising drug candidates, validate and de-risk them through preclinical studies, and progress candidates to the clinic through industry partnerships. The company has raised $14 million since inception from an investor base that includes OS Fund, SoftBank Ventures Asia, Andreessen Horowitz, Stex25, CLI Ventures, and StartX.
XtalPi is a Cambridge, Mass.-based pharmaceutical technology company founded in 2014 by a group of quantum physicists at MIT that uses artificial intelligence to accelerate the development of new drugs. Through its tightly inter-woven quantum mechanics, artificial intelligence, and high-performance cloud computing algorithms, the company’s Intelligent Digital Drug Discovery and Development (ID4) platform enables pharmaceutical companies to increase their efficiency, accuracy, and success rate at critical stages of drug R&D. XtalPi has received $66 million in funding from a base of investors including Zhen Fund, Frees Fund, Tencent Holdings, Sequoia Capital China, Hanhai Investment, Alphabet, Yael Capital, and SIG.
AI in clinical trials
ArcheMedX provides operational intelligence and workforce readiness solutions for life science and healthcare organizations. Ready, the company’s digital platform for clinical trials, enables sponsors and CROs to architect decisions that accelerate site initiation and preparation, enhance operational efficiency, and reduce the risk of underperforming trials. The platform analyzes unique behavioral data to identify leading indicators of site readiness, provide actionable alerts to managers, and automatically re-engage staff and site personnel around critical information. ArcheMedX was founded in 2013 and is based in Charlottesville, Va. The company has raised $2.53 million to date from investors, including Sanofi, Regeneron Pharmaceuticals, Amplifier Ventures, and the Center of Innovative Technology.
Biodesix, founded in 2006 and based in Boulder, Colo., is a lung cancer diagnostic company that addresses the continuum of patient care from early diagnosis of lung nodules through late-stage cancer. The company develops diagnostic tests that address important clinical questions by combining simple blood draws and multi-omics with artificial intelligence. The company’s Lung Reflex strategy integrates its GeneStrat (genomic) and VeriStrat (proteomic) tests to support non-small cell lung cancer decisions with results in 72 hours. The company’s newest test, the BDX-XL2 nodule test, evaluates the risk of malignancy, enabling physicians to triage patients to the most appropriate course of action. The company has raised $176 million in capital since inception. Notable investors include SVB Leerink, Westfield Capital Management, and Crabtree Ventures.
Datavant aggregates and analyzes biomedical data for use in clinical trial decision-making. The company employs artificial intelligence to improve the clinical trial process and to improve the design and interpretation of clinical trials, helping pharma companies manage privacy, security, and compliance and enable safe data sharing. Based in San Francisco, Datavant was founded in 2017 after being spun out from Roivant Sciences. The company has raised $41 million in venture capital since inception. Investors include Founders Fund, Roivant Sciences, and SoftBank Group.
Deep 6 AI, founded in 2015 and based in Pasadena, Calif., is an AI-based eClinical solutions company focused on clinical trial recruitment. The company’s proprietary software uses AI to analyze structured and unstructured healthcare data in real time to find and match patients for clinical trials. The Deep 6 AI software accelerates the time it takes to run trials, cutting drug development costs significantly and getting life-saving cures to people faster. It allows sponsors and CROs to select better trial subjects and helps with trial feasibility design, site selection, and real-time analysis of study results. The company recently raised $17 million in Series A funding led by Point72 Ventures.
Mendel.ai is a developer of AI-based software that solves problems at each stage of the clinical trial process, from feasibility to recruitment to post-trial pharmacovigilance. The company’s software uses deep learning technology to sift through unstructured data in medical literature and data in patient health records to suggest evidence-based treatment options. It updates the results whenever a new matching trial or treatment emerges. Mendel.ai is currently focused on oncology and cancer patient populations. The company, founded in 2016 and based in San Francisco, has raised ~$2 million in seed capital to date.
Saama, founded in 1997 and based in Campbell, Calif., is an advanced clinical data and analytics company. Saama’s unified, AI-driven clinical data analytics platform seamlessly integrates, curates, and animates unlimited sources of structured, unstructured, and real-world data to deliver actionable insights across therapeutic areas. The platform gives real-time visibility into clinical data, enabling drug sponsors to file new drug applications (NDAs) more efficiently and cost effectively. Saama has raised $76 million in venture funding from a base of investors that includes Carrick Capital Partners and Perceptive Advisors.
Tempus, founded in 2015 and based in Chicago, is a data analytics company aimed at advancing precision medicine through artificial intelligence. The company’s large library of molecular and clinical data, combined with an operating system that makes the data accessible and useful, provides genomic sequencing services. It empowers users to make real-time and data-driven decisions to deliver personalized patient care and facilitate the discovery, development, and delivery of optimized therapeutic options for patients through distinctive solution sets. The company has raised ~$520 million in venture funding from a base of investors that includes Revolution Group, Baillie Gifford, Novo Holdings, T. Rowe Price, New Enterprise Associates, Lightbank, and Franklin Templeton Investments.
TriNetX is a global health research network that optimizes clinical trial design and recruitment processes. The company’s network combines EMR data, including demographics, diagnoses, procedures, medications, labs, genomic, and deep oncology data, with data derived from clinical documentation, including discharge summaries, radiology reports, and pathology reports. TriNetX enables healthcare organizations, biopharmaceutical companies, and contract research organizations (CROs) to collaborate, enhance trial design, accelerate recruitment, and bring new therapies to market. The company, founded in 2013 and based in Cambridge, Mass., has raised $94 million in capital to date. Investors include Merck Global Health Innovation Fund, Deerfield Management, Mitsui & Co., MPM Capital, F2 Ventures, and Itochu Technology Ventures.
VIDA, founded in 2003 and based in Coralville, Iowa, offers AI-powered solutions to provide greater precision and personalization in the treatment of emphysema, asthma, interstitial lung disease (ILD), and lung cancer. The company’s flagship offering, LungPrint, is a fully automated AI-powered analysis of an inspiratory chest CT scan that provides a quick flag of lung density abnormalities that may be indicative of emphysema or ILD. LungPrint is part of the VIDA|vision Software Suite and available for clinical use in the United States, European Union, Canada, and Australia. The company has raised $11 million in venture funding since inception. Notable investors include UnityPoint Health Ventures, Chartline Capital Partners, Next Level Ventures, and Rittenhouse Ventures.
AI in commercialization
Atkana, founded in 2008 and headquartered in San Francisco, helps life science sales and marketing teams transform brand strategy into better performance. Through the use of its AI-powered Decision Support Engine, pharmaceutical companies are able to provide their sales and marketing representatives with relevant information and suggested actions on how to engage with target healthcare professionals and healthcare organizations. The company delivers its data-fueled suggestions and insights within a sales professional’s existing CRM workflow, serving as a critical ally in data leverage and better decision making. Nearly half of the world’s top 20 pharmaceutical companies rely on Aktana to drive more insightful marketing and sales programs. Aktana has raised $69 million in capital to date with notable investors including Leerink Transformation Partners, Safeguard Scientifics, HLM Venture Partners, 1315 Capital, and CRV.
Anju Software, an Abry Partners portfolio company based in Tempe, Ariz., provides data and application software to life sciences companies, contract research organizations, and medical device manufacturers. Through its 2018 acquisition of Zephyr Health, Anju also offers insights-as-a-service. The company’s Zephyr platform supports marketers, medical affairs, and sales and analytics teams by merging and analyzing public and private data sets into a unified view. The company was founded in 2015.
Axtria, headquartered in Berkeley Heights, N.J., is a data analytics company that combines industry knowledge, analytics, and technology to help clients make better data-driven decisions. The company’s data analytics and software platforms support sales, marketing, and risk management operations in life sciences. Founded in 2009, the company has raised ~$50 million to date.
Closerlook, founded in 1987 and based in Chicago, combines biopharma brand marketing, digital marketing, and marketing analytics solutions to help pharmaceutical companies promote new therapies. It also offers strategic, creative, and account services, including marketing strategy development, user experience design, and content development.
Decision Resources Group (DRG), headquartered in Burlington, Mass., is a provider of syndicated research, consulting services, web-enabled research, and predictive analytics via proprietary database software to the pharmaceutical, biotech, medical device, managed care, and global healthcare industry. In 2012, the company was acquired by India-based Piramal Enterprises for $635 million (EV/sales of ~4x). The company was founded in 1990.
Hexi.ai, founded in 2018 and based in Chicago, uses AI to identify and map healthcare leaders, including providers, payers, health system executives, and KOLs. The data collected includes buying intentions, relationships, and decision-making power. Hexi.ai pharma customers use the data to accelerate their commercialization strategies. Currently, Hexi.ai has over 1.3 million healthcare leaders in its database, which continues to grow by ~10,000 people each week.
Innoplexus leverages its artificial intelligence and blockchain technology-based platform to support all stages of drug development from pipeline to market. The company identifies and extracts structured and unstructured life science data by scanning the web and merging this data with enterprise and third-party data in an ongoing, real-time process that allows life sciences organizations to generate actionable insights across the pre-clinical, clinical, regulatory, and commercial stages of drug development using data analytics. Using Innoplexus’ technology, commercial, marketing, digital innovation, marketing intelligence, market access, and sales teams can generate the best go-to-market strategy. Founded in 2011 and headquartered in Eschborn, Germany, Innoplexus has raised ~$20 million to date. Investors include NVIDIA GPU Ventures, FinLab EOS, Atai Life Sciences, and HCS Beteiligungsgesellschaft.
Komodo Health, founded in 2014 and based in San Francisco, provides an AI platform that cross-links patient journey data with other medical and healthcare data to extract new insights. It tailors its insights, clinical alerts, and predictive analytics to specific patient cohorts, which it delivers through its end-user products and enterprise partnerships. Its Healthcare Map links over 150 payer datasets, providing real-time and longitudinal data on the complete healthcare journeys of more than 300 million patients in the United States, combining over 50 billion clinical encounters with 15 million new encounters every day. The company has raised $64 million to date. Investors include IA Ventures, Felicis Ventures, and Silicon Valley Bank.
P360 Solutions (Prescriber360), based in Piscataway, N.J., is built on Microsoft’s cloud platform and powers pharmaceutical organizations with advanced, scalable, and ROI-built commercial strategy and technology solutions. Delivering a 360 view through the pharma, prescriber, and patient ecosystem, P360 designs and deploys capabilities that drive efficiencies and returns on sales operations, data management, clinical trials, patient centricity, and IoT innovation. The company was founded in 2015.
SHYFT Analytics is a cloud data company for commercial and real-world data analytics with products designed specifically for the pharmaceutical, biotech, and medical device industry. SHYFT’s portfolio of solutions – which includes Lumen (insights), Quantum (real-world evidence), Nova (patient navigator), and Strata (data storage and integration) – provides clients with on-demand clinical and commercial insights on a unified cloud platform from data that has been aggregated from sources such as providers, payers, prescriptions, claims, and electronic medical records (EMRs). The company was founded in 2004 and is based in Boston. SHYFT raised $39 million prior to being acquired by Medidata in 2018 for $195 million.
Pharmacovigilance
Advera Health Analytics, based in Santa Rosa, Calif., offers a SaaS-based pharmacovigilance platform. The company’s flagship offering, Evidex, aggregates and curates large disparate datasets and deploys proprietary analytics to provide insight on the safety of marketed and pipeline drugs. Evidex aims to mitigate risk by improving the transparency and actionability of drug safety data. The company was founded in 2010 and has raised ~$3 million in private funding to date.
ArisGlobal, based in Miami, offers a drug development platform, LifeSphere, that integrates cognitive computing technologies to automate the core functions of the product lifecycle. Designed with deep expertise and a long-term perspective that spans more than 30 years, the LifeSphere platform delivers actionable insights, boosts efficiency, ensures compliance, and lowers total cost of ownership through a multi-tenant SaaS model. Nordic Capital acquired ArisGlobal through a $700 million LBO in 2019. ArisGlobal was founded in 1987.
MyMeds&Me, based in London, offers a web-based platform that provides adverse event and product quality capture services. The platform simplifies the process of data capture directly from patients and HCPs. Pharma companies use the coded data for rapid evaluation and prompt regulatory reporting. The company, founded in 2011, has raised ~$3 million since inception from Albion Capital.
Augmented reality/virtual reality
Level Ex, headquartered in Chicago, creates mobile, augmented reality, and virtual reality games for physicians. The company uses state-of-the-art video game technology and cognitive neuroscience to capture the challenges of practicing medicine – helping physicians keep up to speed on rare and challenging cases, new medical devices, and drug therapies. The company’s medical video games, played by 500,000 healthcare professionals and leveraged by top 20 pharmaceutical and medical device companies and medical societies, offer continuing medical education (CME) credits and are available free on the Apple App Store and Google Play. Level Ex was founded in 2015. Investors include 4490 Ventures, OSF Ventures, and JAZZ Venture Partners.
ImmersiveTouch, based in Chicago, is a developer of virtual and augmented reality medical software designed to train nurses and new doctors through virtual reality simulators. The company’s software converts radiology scans into 3D simulations, helping surgeons study, assess, and plan their surgeries. The company started under a United States grant initiative and was developed as a National Institutes of Health training tool. The company raised $1.5 million of convertible debt financing from undisclosed angel investors in May 2019.
Key opinion leaders
ACTO, headquartered in Toronto, offers a cloud-based commercialization platform for life sciences companies. The platform helps field reps and medical science liaisons provide physicians and key opinion leaders with the right information so they can champion the right treatment for better patient outcomes. Founded in 2014, ACTO has raised ~$2 million in private funding to date from an investor base that includes MaRS Investment Accelerator Fund, Vermillion, Extreme Venture Partners, Good News Ventures, Innovation Grade Ventures, Cogito Ventures, and Panache Ventures.
rMark Bio, based in Chicago, offers a proprietary deep learning BI platform that analyzes global healthcare data in conjunction with internal clinical research data to optimize partnership and engagement strategies across the lifecycle of a product for pharmaceutical companies, academic institutions, and medical device companies. rMark Bio’s platform aims to reduce the time and cost of data analysis while increasing opportunities to align real-time business strategies between R&D, product, commercialization, and external teams. Founded in 2015, rMark Bio has raised ~$3 million to date from a base of investors that includes Lincoln Park Capital, Math Venture Partners, SaaS Ventures, M25, Stout Street Capital, and Heartland Angels.
Swoop, based in Cambridge, Mass., solves healthcare problems through machine learning and artificial intelligence. Swoop enables better health outcomes by helping the biopharma industry understand, find, and engage its ideal stakeholder populations. Its wholly owned subsidiary, IPM.ai, provides clients with a unique mix of healthcare subject matter expertise, big data capability, and patient privacy protection. Founded in 2011, the company has raised $16 million to date from an investor base that includes Archer Venture Capital, U.S. Venture Partners, and General Catalyst.
Clinical lab testing
Orig3n, based in Boston, provides consumers direct, affordable access to information about their lifestyle-related genes, empowering them to make educated, proactive, and personalized lifestyle choices that may impact their well-being. Since its founding in 2014, Orig3n has built a suite of direct-to-consumer genetic tests. The company has raised $86 million in venture capital funding since inception. Investors include Hatteras Venture Partners, Alexandria Venture Investments, LabCorp, Spectrum Health Ventures, Vectr Ventures, 180 Degree Capital, Mountain Group Partners, KTB Ventures, DEFTA Partners, MMIC Investment, and Haitong International Securities Group.
Prognos, based in New York City, is a healthcare AI company focused on predicting diseases to drive earlier decisions in collaboration with payers, life sciences companies, and diagnostics companies. The Prognos Registry is the largest source of clinical diagnostics information in 50 disease areas, with nearly 24 billion medical records for 200 million patients. Prognos has 1,000 extensive proprietary and learning clinical algorithms to enable earlier patient identification for enhanced treatment decision-making, risk management, and quality improvement. Investors include Safeguard Scientifics Inc., Merck Global Health Innovation Fund, Cigna, GIS Strategic Ventures, Hikma Ventures, Hermed Capital, and Maywic Select Investments. The company was founded in 2010.
Point of care
Crossix, based in New York City, empowers healthcare companies with the ability to plan, measure, and optimize their marketing and commercialization strategies. Crossix connects Rx, OTC, CPG, clinical, medical claims, hospital, consumer, and media data with best-in-class privacy safeguards far exceeding HIPAA requirements. Driven by its distributed data network technology and a comprehensive set of health and non-health data of more than 250 million U.S. consumers, Crossix analytics enables pharmaceutical companies to focus their efforts on targeting the ideal patients and healthcare providers for their suite of therapies. Founded in 2004, Crossix was recently acquired by Veeva for $430 million.
PatientPoint, based in Cincinnati, connects pharmaceutical and health and wellness brands to their most valuable patients and physicians at critical decision-making moments. Harnessing the power of digital and mobile technology, the company’s omnichannel platform of patient engagement solutions includes digital waiting room screens in physician offices and hospitals and interactive touchscreens in exam rooms. The company develops the majority of its content internally, tailoring education to specific specialties to ensure that it meets the unique needs of patients and providers in the point-of-care environment. Founded in 1987, the company has raised $171 million to date from an investor base that includes Searchlight Capital Partners and Silver Point.
Phreesia, headquartered in New York City, provides an integrated platform that offers an array of solutions for care organizations, such as patient intake, appointment scheduling, revenue cycle management, clinical support, and analytics reporting. The company also offers pharma-targeted, direct digital marketing access in point-of-care settings and counts 13 of the top 20 global pharma companies as clients. Phreesia, founded in 2005, raised $119 million before going public in 2019 (NYSE: PHR).
mHealth
AliveCor, based in Mountain View, Calif., is a developer of mobile electrocardiogram (ECG) technology designed to improve stroke prevention through early atrial fibrillation detection. The company’s device enables patients to monitor their heart health via their smartphones and provides physicians with an additional heart health assessment tool, helping them detect atrial fibrillation and normal sinus rhythm in an ECG. The company has raised $92 million since inception from an investor base that includes WP Global Partners, Qualcomm Ventures, Oklahoma Life Science Fund, the Mayo Clinic, and Khosla Ventures. The company was founded in 2010.
Evidation Health, based in San Mateo, Calif., is a tech-enabled solutions and services provider that helps healthcare companies quantify and realize value in the digital era of medicine. Evidation developed its real-world evidence study platform to accelerate and enhance outcomes research through direct-to-patient “siteless trials.” The platform enables the passive collection and deep analysis of continuous behavior data alongside traditional healthcare information, empowering individuals and companies to understand and influence the everyday behaviors that create better health outcomes. Evidation works across the healthcare ecosystem with top pharmaceutical companies, payers, providers, and digital healthcare companies. Founded in 2014, Evidation has raised $61 million in capital from SV Health Investors, Rethink, B Capital Group, GE Ventures, Revelation Partners, and Sanofi Ventures.
Human API, based in San Mateo, Calif., offers a platform that allows customers to integrate consumer data from across the healthcare ecosystem into their products and solutions. The company’s real-time health data network connects, normalizes, and structures data from 85% of acute care and critical access hospitals, along with pharmacies, labs, and 300+ wearable devices and fitness apps. The company empowers corporate wellness, insurance, health plans, digital therapeutics, and clinical research organizations to build consumer-centric health apps and services. The company has raised $17 million since inception from an investor base that includes BlueRun Ventures, Andreessen Horowitz, SpringRock Ventures, and SciFi VC. Human API was founded in 2013.
Koneksa Health, based in New York City, enables the biopharmaceutical industry to develop new medicines with fewer patients. The company’s SaaS solution integrates and analyzes data from a wide variety of wearables and mobile devices from different manufacturers. By unlocking the potential of real-world data from remote, wearable, and other digital technologies, Koneksa speeds up the time required to understand how a drug is working and helps to develop real-world evidence for how medicines can impact the daily lives of patients. The company was founded in 2015 and has raised ~$1 million in seed capital to date from Waterline Ventures and Merck’s Global Health Innovation Fund.
Oxford Nanopore Technologies (ONT), based in the United Kingdom, was founded in 2005 as a spin-out from the University of Oxford by Hagan Bayley, Gordon Sanghera, and Spike Willcocks. ONT is the first company to commercially launch a nanopore-based DNA sequencer. In a nutshell, nanopores are small holes through which DNA can travel, generating an electronic signal that is used to sequence the individual bases. ONT has raised $709 million in private equity and venture funding to commercialize nanopore-based real-time DNA sequencing systems, which include MinION, PromethION, and GridION, all of which are adaptable for the analysis of DNA, RNA, proteins, small molecules, and other types of molecules.
PhysIQ, based in Naperville, Ill., and founded in 2013, enables proactive care delivery models through pinpointIQ, its highly scalable cloud-based platform for personalized physiology analytics. The company’s FDA 510(k)-cleared data analytics platform is designed to process multiple vital signs from wearable sensors to create a personalized dynamic baseline for each individual. By mapping vital sign relationships, PhysIQ’s analytics detect subtle deviations that may be a precursor to disease exacerbation or changes in health. With applications in both healthcare and clinical trial support, the company provides insight for providers, health systems, payers, and pharmaceutical and medical device companies.
Validic, based in Durham, N.C., offers a healthcare platform designed to simplify personal health data. The company’s platform captures data from clinical and remote-monitoring devices, sensors, fitness equipment, and patient-wellness applications, enabling users to access, manage, and utilize device-generated data. Validic’s clients reach over 223 million people in 52 countries and include health systems and providers, payers, wellness companies, pharma, health IT companies, as well as app and device manufacturers. Validic connects to over 400 in-home clinical and mobile health devices and applications. The company has raised ~$20 million since inception from an investor base that includes Greycroft, SJF Ventures, Kaiser Permanente Ventures, and NTT Domoco Ventures. The company was founded in 2010.
Digital therapeutics (DTx)
Omada, based in San Francisco, is a digital care program provider that empowers people to achieve their health goals through sustainable lifestyle changes. Working primarily through health plans, employers, and integrated health systems, the company delivers personalized interventions for individuals at risk for, or dealing with, type 2 diabetes and hypertension, as well as anxiety and depression. Combining data-powered human coaching, connected devices, proprietary technology platform, and curriculum tailored to an individual’s specific conditions and circumstances, Omada has enrolled more than 250,000 participants to date. Omada partners include Cigna, Kaiser Permanente, Health Care Services Corp. (HCSC), and Blue Cross and Blue Shield Minnesota. Founded in 2011, the company has raised ~$200 million since inception from an investor base that includes US Venture Partners, Andreessen Horowitz, Norwest Venture Partners, Kaiser Permanente Ventures, Humana Ventures, and Providence Ventures.
Pear Therapeutics, based in Boston, discovers, develops, and delivers clinically-validated software-based therapeutics to provide better patient outcomes, smarter engagement and tracking tools for clinicians, and cost-effective solutions for payers. Pear’s lead product, reSET, treats Substance Use Disorder, and was the first prescription digital therapeutic to receive marketing authorization from the FDA to treat disease. Since its founding in 2013, Pear has raised $139 million from an investor base that includes Temasek Holdings, Jazz Venture Partners, 5AM Ventures, Arboretum Ventures, and Novartis.
Medication adherence & compliance
AiCure, founded in 2010 and based in New York City, offers scalable medication adherence and intelligent medical assistant (IMA) solutions that leverage a visual recognition platform to monitor patient progress on mobile devices. The company’s solution confirms medication ingestion in clinical trials and high-risk populations. Its software identifies the patient and medication and uses various features to detect medication diversion and identify duplicate enrollment. It captures evidence of medication ingestion, detects non-adherence to allow for immediate intervention, and provides longitudinal tracking of adherence patterns. The company has raised $69 million in capital since inception. Notable investors include Asahi Kasei, New Leaf Venture Partners, Palisades Growth Capital, Baird Capital, Pritzker Group Venture Capital, SpringRock Ventures, and Tribeca Venture Partners.
AllazoHealth, headquartered in New York City, is an artificial intelligence/machine learning company that focuses on driving the behavioral change of patients through personalized and proactive intervention. The company’s mission is to be the leader in precision engagement and predictive analytics, revolutionizing the way enterprise healthcare clients engage and improve the health of their patient population. The company was founded in 2011 and joined the New York Digital Health Accelerator and Wharton Venture Initiation Program, receiving an undisclosed amount of funding in 2014.
HealthPrize Technologies, based in Norwalk, Conn., provides a Software-as-a-Service, HIPAA-compliant platform and services to create unique digital experiences that drive patient engagement, education, and increased medication adherence for healthcare companies and pharmaceutical brands. The company utilizes a proprietary gamification approach along with behavioral economics and loyalty marketing to ensure patients adhere to their medications. Notable clients include Pfizer, Walgreens, Gilead, and Novo Nordisk. The company was founded in 2009 by industry veteran Tom Kottler and neurosurgeon Katrina Firlik. The company has raised ~$15 million since inception.
Medisafe, based in Boston, offers a digital health platform for medication management. Data-powered technology delivers personalized interventions based on behavioral insights, keeping patients engaged in, and adherent to, their treatments. Medisafe leverages real-world data to design scalable programs around therapeutic needs while personalizing engagement into patients’ daily lives. Since its launch in 2012, Medisafe has cultivated a user base of 4.5 million across iOS and Android smartphones and tablets, recording over 2 billion medication doses taken correctly. The company has raised $21.5 million since inception from an investor base that includes Lool Ventures, Qualcomm Ventures, Pitango Venture Capital, 7wire Ventures, Octopus Ventures, and M Ventures. The company was founded in 2012.
OptimizeRx, based in Rochester, Mich., is a publicly traded (NASDAQ: OPRX) digital health technology company, connecting life sciences, payers, providers, and patients using a point-of-care communications platform and disease-specific digital health frameworks. The cloud-based solution supports patient adherence to medications by providing real-time access to financial assistance, prior authorization, education, and clinical information. The company was founded in 2006.
Patient communities & social media
Backpack Health, based in Dover, Mass., is a health technology company that builds digital tools to help people manage their health journey – including wellness, illnesses, injuries, and chronic health conditions. The Backpack Health mission is to make it easy for everyone to access, own, and control their health information to attain better health for themselves, their loved ones, and their communities. Backpack Health provides a platform for organizations to engage patients, collect up-to-date data, and build communities around the globe. Founded in 2016, the company has raised $8 million since inception.
Health Union, based in Philadelphia, encourages social interactions that evolve into valuable online health conversations, helping people with chronic conditions find the information, connections, and validation they seek. The company creates condition-specific online communities – publishing original, daily content and continuously cultivating social conversation – to support, educate, and connect millions of people with challenging, chronic health concerns.
Liquid Grids, based in San Diego, has developed a purpose-driven, automated platform in social media specifically targeted for application in the healthcare industry. The technology aggregates and classifies online health-related dialogue into subject disease discussions, applying complex analytics that allow users to gather meaningful insight into the who, what, and when of health conversations within virtual communities. The company uses this data to connect brands in the healthcare industry to target audiences. Liquid Grids was founded in 2010.
Hub services
ConnectiveRx, based in Whippany, N.J., is a technology-enabled services company that works strategically with biopharmaceutical manufacturers to help commercialize and maximize the benefits of specialty and branded medications. The company’s services include patient and provider messaging, the design and operation of co-pay, voucher, and patient assistance programs, and hub services. It also owns the iconic Physicians’ Desk Reference, now known as the Prescribers’ Digital Reference. The company was founded in 2005 and has raised $300 million of debt and equity since inception from an investor base that includes Genstar Capital and Foresite Capital Management.
CoPilot, based in New Hyde Park, N.Y., was founded in 2010 for the purpose of providing innovative, patient-centric hub and market-access software and services to manufacturers. Through partnerships with those manufacturers, the company designs customizable solutions that improve productivity and reduce administrative costs while improving pull-through and clinical outcomes. The company’s proprietary CRM solution, Salesdoor, is designed specifically for the specialty pharmacy, pharmaceutical, and healthcare industries.
IntegriChain, based in Philadelphia, is an analytics company that helps innovative pharmaceutical and biotechnology manufacturers identify and remove barriers to patient therapy initiation and adherence. The company’s ICyte Platform and solutions focus on what happens after the patient and provider commit to therapy. More than 140 life science manufacturers rely on insights delivered from IntegriChain’s data, analytics, and managed services to drive strategic decision-making and ongoing business management across their payers, specialty pharmacies, patient services, and distribution channel partners. The company raised $7 million in private funding prior to being acquired by Accel-KKR through an LBO in 2016 for an undisclosed amount. The company was founded in 2005.
KloudScript, based in Oakbrook Terrace, Ill., provides technology and support services for specialty medications. KloudScript partners with community pharmacies, prescribers, pharmaceutical manufacturers, and payors to make specialty medications available locally for patients through a simplified care experience from their pharmacist. Through its technology platform, KETU, and support services, KloudScript helps patients receive specialty pharmacy care in their community and helps community pharmacies participate in the fastest-growing segment of the pharmacy market.
Health economics/outcomes research, real world evidence
Analytical Wizards, based in Edison, N.J., is a data science and analytics firm that delivers actionable insights from data through its AI/ML-powered technology, domain expertise, and high-touch relationship model. Analytical Wizards has a growing global presence with offices in New Jersey, Boston, California, and Bengaluru, India. The company raised $3 million of Series A venture funding from Sopris Capital Associates in October 2019.
GNS Healthcare, based in Cambridge, Mass., is a healthcare analytics company focused on advancing and applying industrial-scale data analytics to empower key healthcare stakeholders to solve complex care, treatment, and cost challenges. Founded in 2000, the company has raised $56 million since inception from an investor base that includes Cigna Ventures, Celgene, Alexandria Venture Investments, Amgen Ventures, and Echo Health Ventures.
Inovalon, based in Bowie, Md., is a publicly traded (NASDAQ: INOV) provider of cloud-based analytics and platforms for healthcare providers and pharmaceutical firms. Inovalon supports thousands of clients, including 24 of the top 25 health plans in the United States, 22 of the top 25 global pharma companies, and 19 of the top 25 healthcare provider systems in the United States. Inovalon’s technology platforms and analytics are informed by data pertaining to more than 980,000 physicians, 546,000 clinical facilities, 287 million Americans, and 48 billion medical events. The company was founded in 1998.
inVibe Labs, based in Costa Mesa, Calif., aims to redefine the way life science companies listen to their customers by capturing real voices in conversation-like interviews without reliance on live moderators, allowing participants to share their stories uninterrupted and translate them into actionable insights. inVibe’s structured methodology, combined with sociolinguistic analysis and AI-powered acoustic metrics, provides unique emotion and customer insights that are used by major pharmaceutical companies, marketing agencies, and healthcare startups. The company was founded in 2014.
IQVIA (NYSE: IQV), headquartered in Durham, N.C., and formed through the 2016 merger of IMS Health and Quintiles, is a leading provider of information, technology solutions and contract research services focused on using data and science to help healthcare clients find better solutions for their patients. IQVIA offers a range of solutions that harness advances in healthcare information, technology, analytics, and human ingenuity. It enables companies to rethink approaches to clinical development and commercialization, innovate with confidence, and accelerate meaningful healthcare outcomes. IQVIA’s approach to human data science is powered by IQVIA CORE, driving unique actionable insights at the intersection of big data, advanced technology, and analytics.
OM1, based in Boston, is a healthcare technology company focused on providing outcomes measurement, comparison, and prediction of treatment outcomes. OM1’s real-world evidence platform, clinical registries, and AI technologies enable clients to accelerate research, measure and benchmark health outcomes, and personalize patient care. Founded in 2015, the company has raised $91 million since inception from a base of investors that includes Scale Venture Partners, General Catalyst, 7wire Ventures, and Polaris Partners.
Scientist.com, based in San Diego, is a software company that is driving a new approach to scientific research. The company runs a B2B online marketplace where scientific researchers can connect with technical experts and purchase innovative tools, technologies, and outsourced services. Founded in 2007, the company has raised $38 million since inception from an investor base that includes Leerink Transformation Partners, 5AM Ventures, Bootstrap Venture Partners, and Heritage Provider Network.
TARGET PharmaSolutions, based in Durham, N.C., is a clinical data company that designs and sponsors real-world evidence (RWE) communities of large disease-specific patient cohorts to generate RWE and provide regulatory-grade data to partners to improve clinical, medical, and commercial outcomes throughout the drug development and commercialization stages. TARGET currently has six disease communities, including TARGET-NASH (nonalcoholic steatohepatitis), TARGET-PBC (primary biliary cholangitis), TARGET-HCC (hepatocellular carcinoma), TARGET-IBD (inflammatory bowel disease) TARGET-DERM (immune-mediated inflammatory skin conditions), and TARGET-HBV (chronic hepatitis B). The company has partnerships with nine leading pharma companies and a network of 200+ community and academic sites. Founded in 2015, the company was acquired by Norwest Venture Partners in May 2018.
List of acronyms
ACA — Affordable Care Act
ACO — Accountable care organization
AI — Artificial intelligence
AMA — American Medical Association
AR/VR — Augmented reality/virtual reality
BI — Business intelligence
BV — Benefit verification
CCPA — California Consumer Privacy Act
CME — Continuing medical education
CMS — Centers for Medicare & Medicaid Services
CRM — Customer relationship management
CRO — Contract research organization
CTMS — Clinical trial management systems
DEA — Drug Enforcement Administration
DTC — Direct-to-consumer
DTC Act — Drug Price Transparency in Communications Act
DTx — Digital therapeutics
eBV — Electronic benefit verification
ECG — Electrocardiogram
EDC — Electronic data capture
EHR — Electronic health record
EMR — Electronic medical record
ePA — Electronic prior authorization
ePRO — Electronic patient-reported outcome
FAERS — FDA Adverse Event Reporting System
FDA — U.S. Food and Drug Administration
GAN — Generative adversarial networks
GDPR — General Data Protection Regulation
GPO — Group purchasing organization
HCO — Healthcare organization
HCP — Healthcare provider
HEOR — Health economics and outcomes research
HHS — Department of Health and Human Services
HIPAA — Healthcare Insurance Portability & Accountability Act
IDN — Integrated delivery network
ILD — Interstitial lung disease
IMA — Intelligent medical assistant
IRB — Institutional review board
KOL — Key opinion leader
MDD — Major depressive disorder
MDM — Master data management
ML — Machine learning
MSL — Medical science liaison
NDA — New drug application
NLP — Natural language processing
NPI — National Provider Identifier
OPDP — FDA Office of Prescription Drug Promotion
PA — Prior authorization
PaaS — Platform as a Service
PBM — Pharmacy benefits manager
PDMA — Prescription Drug Marketing Act
PhRMA — Pharmaceutical Research and Manufacturers of America
POC — Point of care
PRO — Patient-reported outcome
PV — Pharmacovigilance
RCT — Randomized controlled trial
REMS — Risk evaluation and mitigation strategies
RWD — Real-world data
RWE — Real-world evidence
SaaS — Software-as-a-Service
SCHIP — State Children’s Health Insurance Program
SUPPORT Act — Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act
TDDD — Terminal Distributor of Dangerous Drugs
WAC — Wholesale acquisition cost

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
