Quarterly insights: Pharma IT
How COVID-19 is reshaping clinical trials and commercialization

The COVID-19 world of social distancing, shelter-in-place orders, travel limitations, and geographical lockdowns has intensified the already substantial challenges of bringing a drug through clinical trials and commercializing it, given these processes traditionally have involved much face-to-face interaction among patients, healthcare professionals, and pharma company personnel.
However, the cloud of COVID-19 has a silver lining – it is accelerating adoption of clinical trial software and technology-based commercialization solutions that overcome many of the COVID-19 obstacles as well as increase the speed and efficiency of clinical trials and commercialization efforts. Recent regulatory relief in response to COVID-19’s impact on clinical trials has only added to this momentum.
We discuss the key relevant enabling technologies for clinical trials, such as telehealth in-home patient visits, direct-to-patient drug dispensing, remote data capture, and remote monitoring to maintain oversight of trial sites and highlight some of the innovative companies bringing them to market.
We also highlight solutions that address COVID-19’s challenges to the commercialization process. These include specialized virtual meeting solution providers, a remote physician engagement device, and an AI-driven recommendation engine for identifying key opinion leaders.
TABLE OF CONTENTS
Includes discussion of three public and 14 private companies
- COVID-19 a major obstacle to clinical trials
- COVID-19 driven regulatory relief means increased adoption of remote technology for clinical trials
- COVID-19 also hinders drug commercialization
- Impact well beyond the pandemic
- Commercialization and eClincial indices regaining ground lost during COVID-19
- Pharma services 2Q20 M&A activity to date consistent with previous two quarters
- Clinical trial software a prevalent theme in 2Q20 private placements to date
COVID-19 a major obstacle to clinical trials
As society deliberates over the short- and long-term impacts of COVID-19, the pharmaceutical industry is adjusting to a new world, a world driven by social distancing, shelter-in-place orders, travel limitations and geographical lockdowns. While these measures are mitigating the spread of the virus, they have created obstacles and challenges to clinical research.
Since the beginning of March about 100 companies, including Pfizer, Bristol-Myers Squibb, and Eli Lilly, have reported delays or disruptions of clinical trials. The industry’s shift in focus to developing vaccines and treatments in response to COVID-19 along with restricted travel for patients, trial sites closures, and the redeployment of research personnel to treat COVID-19 patients have disrupted clinical trials for diseases other than COVID-19.
The pharmaceutical industry has traditionally relied heavily on on-site monitoring, including frequent source data verification (SDV), to help ensure clinical trial processes, procedures, and records satisfy requirements for protocols, standard operating procedures, good clinical practices and regulatory compliance. Even under normal circumstances, achieving 100% SDV for trial sites can be a laborious and costly task. COVID-19 has only added to the complexity. On its first-quarter earnings conference call, industry bellwether IQVIA noted 80% of its clinical trial sites were inaccessible to clinical research associates for on-site visits, while competitor Syneos Health estimated 85% of its sites were inaccessible.
Keeping clinical trials going and on track wherever feasible and safe during COVID-19 is critical to bringing much-needed treatments to patients. At the same time, COVID-19 makes continuing trials under the traditional model while maintaining patient safety a significant challenge. Many patients enrolled in trials may be immunocompromised, putting them at particular risk if exposed to COVID-19. Further, some patients who are receiving treatments in trials may also require medical assessment, which may be difficult to obtain when healthcare facilities and personnel are focused on dealing with COVID-19. Other challenges include patient anxiety and reluctance to visit trial sites and elevated infection risk between patients and trial staff.
COVID-19 driven regulatory relief means increased adoption of remote technology for clinical trials
All this led to the March release of guidance from both the National Institutes of Health (NIH) and the U.S. Food and Drug Administration (FDA) regarding clinical trial conduct during the COVID-19 pandemic. Both guidance documents emphasize patient safety when it comes to trial recruitment, continued participation, and drug administration. Additionally, the FDA’s guidance provides some flexibility around trial conduct.
While trial sponsors still must report protocol changes, changes now can be implemented without institutional review board approval and before filing amendments with the FDA. Such changes include using alternative patient assessment methods such as telehealth in-home patient visits, direct-to-patient (DTP) drug dispensing, remote data capture, and remote monitoring to maintain oversight of trial sites. While delays and disruptions have been commonplace, many sponsors and contract research organizations (CROs) have been able to avoid outright cancellations thanks to increased adoption and implementation of such alternatives that use technology to enable decentralized trials.
We note many of these solutions have been in use for a number of years but have faced slow adoption due to the heavy regulation around developing and approving new medicines, which has made sponsors wary of changing processes. COVID-19 has rewritten the playbook. For example, IQVIA noted it has not had a single COVID-19-related cancellation thanks to implementing telehealth visits and remote monitoring solutions in about 50% of its current trials. This level of deployment is about five times the level prior to COVID-19. Beyond IQVIA, we see companies like Saama, Clue Points, and Thoughtsphere, which provide analytics-driven risk mitigation capabilities for trial monitoring, as prime beneficiaries of this change both now and in the months and years ahead.
The rapid adoption of cloud-based solutions, wearable biosensors, mobile phones, tablets, and telehealth has led to the emergence of DTP trial solutions, and COVID-19 increased the impetus to adopt them as evidenced by the recent partnerships between DTP provider Medable and Covance and DTP provider Science 37 and PPD. In addition to Medable and Science 37, other DTP solution providers like Hawthorne Effect, Virtrial, Empiramed, Thread and Transparency Life Sciences also provide for trial continuity by offering trial participants the ability to participate from their homes.
For example, Hawthorne Effect’s tech-enabled services platform empowers a global network of more than 1,800 certified healthcare professionals, called HEROs, to provide decentralized and remote trial services via in-home patient visits. HEROs collect the needed patient-level data and integrate it directly with the company’s HIPAA-secure cloud-based platform. According to CEO Jodi Akin, the end result is timely and comprehensive data collection.
One notable vendor addressing site capacity challenges, patient safety, and supply chain disruption is 4G Clinical1, which provides randomization and trial supply management (RTSM) software. In April, 4G launched DTP functionality in its Prancer RTSM, which enables sponsors to ship drugs directly from the site, depot, or central pharmacy to the patient’s home. According to CEO David Kelleher, 4G is experiencing strong demand from pharma clients for DTP functionality on both new and ongoing trials.
Looking ahead, at some point in the future when COVID-19 is either cured or no longer a significant threat to public health and trial sites are all opened, we think many of the DTP solutions adopted by pharma out of necessity today will continue to be employed in a hybrid fashion as we expect the current crisis to highlight to pharma sponsors DTP’s utility and feasibility.
COVID-19 also hinders drug commercialization
Commercialization has been hampered by COVID-19 as both elective procedures and in-person meetings between sales reps and healthcare professionals ground to a near halt. Despite the recent opening of some geographical regions and the resumption of elective procedures, access to physicians remains unusually difficult.
According to several sales reps we spoke with, in-person visits with healthcare professionals are still limited or non-existent, and many speculate they may never return to the pre-pandemic level. Even before the outbreak of COVID-19, pharma reps’ in-person visits were trending downward. As we noted in our Going Beyond the Pill: Digital solutions driving pharma commercialization and improving patient outcomes, the percentage of physicians classified as “rep-accessible” dropped from 80% in 2008 to 44% in 2016.
TABLE 1: Accessible prescribers since 2008
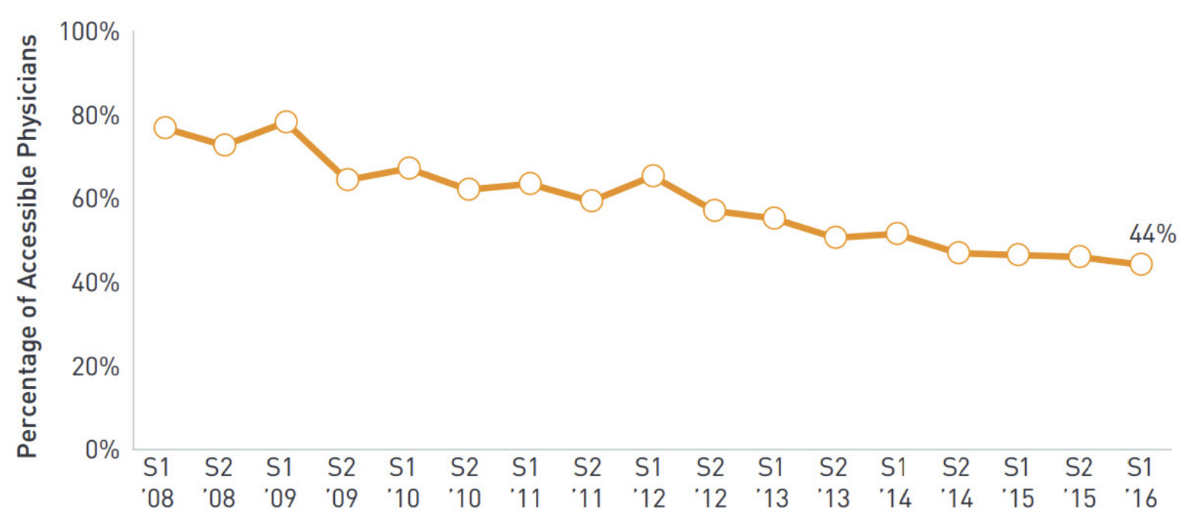
Source: ZS Associates AccessMonitor.
But the fact that doctors are spending less time with sales reps does not mean they don’t need or aren’t interested in the information the reps would like to present to them. In this context, the pandemic has accelerated the transformation of the sales rep’s role to that of a curator of content and virtual communication strategies.
According to industry leader Veeva, virtual meetings in the United States between pharma personnel and doctors increased by a factor of more than 30 from February to April, and approved email communications more than doubled. Virtual engagement services providers Meeting Protocol Worldwide and OptimizeRx are experiencing this trend as well.
OptimizeRx recently launched TelaRep, its latest electronic health record enabled digital health tool designed to support physicians who are treating patients with specialty drug therapies. According to the company’s chief commercial officer, Steve Silvestro, TelaRep helps fill an important communication gap between healthcare professionals and pharma medical science liaisons and reps when it comes to treating patients with complex disease by enabling video chat functionality inside the existing electronic health record workflow.
One innovative vendor offering a virtual sales rep solution right to the physician’s desk is P360, which recently launched Swittons, an end-to-end remote physician engagement device built on Microsoft Azure cloud that is designed to bridge the communication gap between pharma sales reps and physicians. Each button on the device can be preprogrammed with a specific action such as real-time communication with a sales rep or medical science liaison, sample refill, and access to important medical information. According to P360’s vice president of commercial excellence, Kimberly Brue, Swittons enables communication with remote healthcare professionals who may have been previously untargeted, provides additional coverage for resource-constrained sales teams, and helps boost brand awareness of new and existing drugs.
One last area of commercialization activity hindered by COVID-19 is healthcare conferences. With major in-person conferences likely to be among the last activities to resume in the wake of COVID-19, virtual conferences and webinars are replacing them, at least in the near term. According to rMark Bio CEO Jason Smith, this trend has forced pharma companies to rethink their key opinion leader (KOL) engagement strategies. The pharmaceutical industry has depended heavily on influential doctors to help validate and promote use of its drugs: doctors trust other doctors, especially thought leaders in their fields.
Smith noted rMark is seeing an uptick in inbound interest for the company’s artificial intelligence-based Fabric solution, which mines and analyzes internal pharma data sources, such as key performance indicators, CRM data, and sales rep data, along with publicly available global health data, to provide recommendations on KOLs that match a pharma company’s business strategy.
Impact well beyond the pandemic
Much like COVID-19 has accelerated adoption of remote workplace collaboration tools like Microsoft Teams, Slack, and Zoom and daily given millions of companies and workers the opportunity to experience firsthand how these tools can improve productivity well after the pandemic, we see COVID-19 accelerating adoption of digital solutions for clinical trials and commercialization and giving pharma sponsors firsthand experience with the efficiencies and other benefits they bring. We think this pandemic-driven field test will have a lasting impact and cause pharma to rethink development, sales, and marketing strategies, resulting in a further shift in spending and resources toward digital solutions.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
