Quarterly insights: Pharma IT
Technology is key in medical affairs’ rise as pharma’s third pillar

The pharmaceutical industry’s shift away from dependence on sales reps for commercialization, the broad evolution of healthcare in recent decades, and the effects of the pandemic on healthcare practices have set the stage for pharma companies’ medical affairs (MA) function to join commercialization and research and development as a key pillar of the industry. MA addresses the need to inform, sitting at a pivotal intersection in pharma and serving as an internal bridge between research and development and commercialization as well as a bridge between pharma companies and their external stakeholders.
Technology has been a key factor behind MA’s emergence as pharma’s third pillar. Digital tools have enabled MA to derive and communicate meaningful insights and evidence from the huge and complex mass of scientific data generated by the healthcare system. These tools continue to evolve, further empowering MA and making it even more effective as one of the three pillars.
We discuss three areas where innovative technology companies are delivering powerful new tools for MA personnel: finding key opinion leaders, engaging and collaborating virtually, and generating insights and evidence. For each, we highlight some of the technology providers that offer compelling solutions.
TABLE OF CONTENTS
- A brief history of medical affairs
- Technology among several factors making MA the third pillar
- Finding key opinion leaders
- Virtual engagement and collaboration
- Insights and evidence generation
- Powerful technologies strengthen pharma’s third pillar
- eClinical index lead over S&P 500 narrows; commercialization index turns negative
- Pharma IT M&A pace remains strong
- Pharma IT private placement pace accelerates from Q3 low
A brief history of medical affairs
Medical affairs got its start as a key pharma function in 1967 at Upjohn (now part of Viatris, VTRS). The increasing complexity of drugs led the company to rethink how it engaged and communicated with healthcare providers. It launched a technically sophisticated team of field personnel to work with sales representatives to improve the image of the company in the eyes of both clinical researchers and key opinion leaders. These medical science liaisons (MSLs), as they are known, employed a face-to-face strategy rooted in education to better understand the needs of Upjohn’s customers and leverage the insights gleaned to enhance the company’s product development strategy.
With time and broad adoption throughout the industry, the model evolved into the medical affairs team – a team of scientifically and medically trained personnel who are experts in communicating complex therapeutic scientific and medical information to a variety of stakeholders.
TABLE 1: Medical science liaisons at the center of information flows
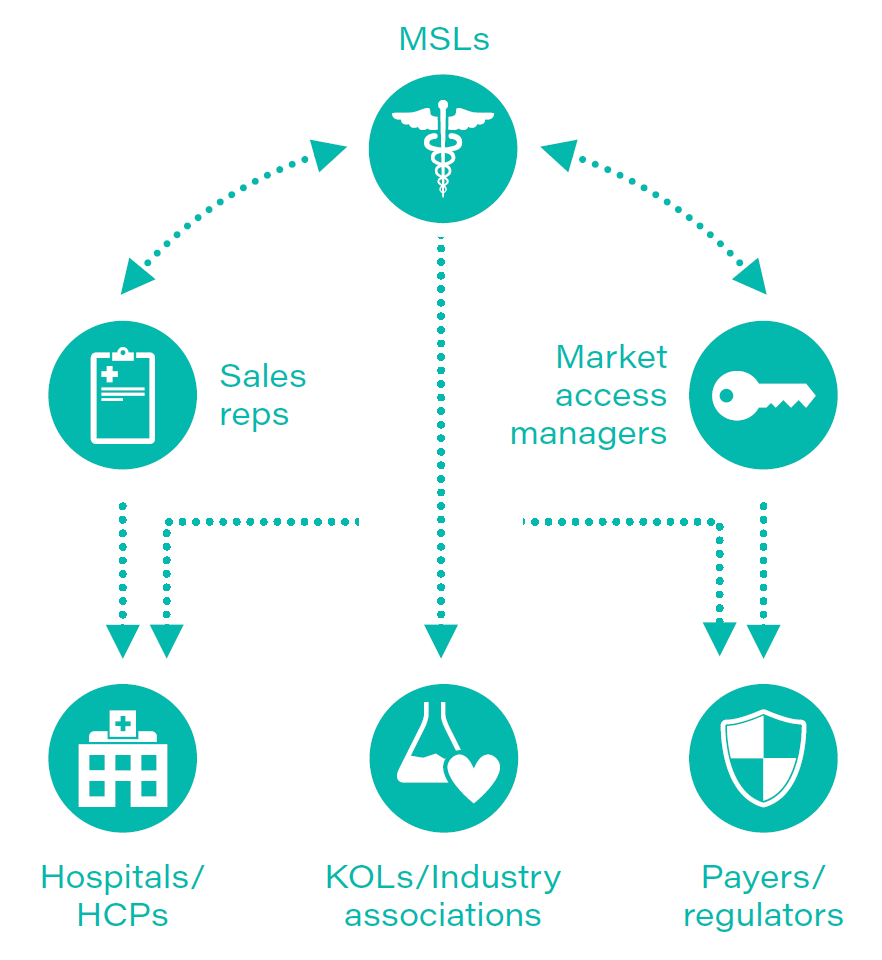
Source: First Analysis.
Technology has been a key factor behind MA’s emergence as pharma’s third pillar. Digital tools have enabled MA to derive and communicate meaningful insights and evidence from the huge and complex mass of scientific data generated by the healthcare system. These tools continue to evolve, further empowering MA and making it even more effective as one of the three pillars.
We discuss three areas where innovative technology companies are delivering powerful new tools for MA personnel: finding key opinion leaders, engaging and collaborating virtually, and generating insights and evidence. For each, we highlight some of the technology providers that offer compelling solutions.
Technology among several factors making MA the third pillar
Beginning in the 1960s, pharmaceutical companies relied heavily on teams of sales representatives for promotional activities when launching a new drug. Commercialization, the second pillar of pharma after research and development, depended largely on the number of sales reps, their selling skills, and their healthcare provider relationships and frequency of interactions. Sales reps met directly with healthcare providers to deliver information and free drug samples, aiming to convince providers to write prescriptions and eventually become advocates for their drugs.
While sales representatives still play a vital role in the industry, they no longer dominate the landscape. The healthcare community’s shift to focus on improving clinical outcomes and making decisions grounded in value-based evidence has made sales reps less relevant as a source for information and engagement. This is particularly true among younger physicians. They want to feel informed, not influenced, and are therefore turning to more scientific sources of information such as peer-reviewed journals, manufacturers’ websites, and webinars and conferences led by key opinion leaders (KOLs). The pandemic has only accelerated this shift as protocols and regulations controlling access to healthcare facilities have restricted sales reps’ access to providers.
These changes set the stage for MA, centered on teams of medical science liaisons, to emerge as pharma’s third pillar. MA addresses the need to inform, sitting at a pivotal intersection in pharma and serving as an internal bridge between research and development and commercialization as well as a bridge between pharma companies and their external stakeholders, including healthcare providers, payers and patients.
Medical science liaisons facilitate expanded access pro-grams for patients to obtain new drugs prior to approval, investigator-initiated research, real-world evidence generation, health eco-nomics outcomes research, scientific publications, medical education, and collaboration with patient advocacy groups, among many other activities. The industry-wide push for developing personalized medicines has made MA even more important given their highly complex nature that requires deeper scientific insights and greater stakeholder collaboration in therapies’ life cycles.
But technology is another key factor behind MA’s emergence as pharma’s third pillar. Digital tools have been key to enabling MA to derive and communicate meaningful insights and evidence from the huge and complex mass of scientific data generated by the healthcare system. These tools continue to evolve, further empowering MA and making it even more effective as one of pharma’s three pillars. We highlight three areas where innovative technology companies are delivering powerful new tools for MA personnel.
Finding key opinion leaders
An important role for MA is identifying and building relationships with key opinion leaders. KOLs are typically physicians who have recognized expertise in a specific field, such as oncology, endocrinology, urology and neurology. By publishing research, presenting at conferences and webinars, and contributing to advisory boards, guideline committees and professional societies, KOLs serve as knowledge resources and advisors for fellow healthcare providers. Their opinions and expertise lend credibility to pharmaceutical marketing campaigns, and the role of KOLs can be critical in a drug’s life cycle. According to a 2015 Healthcasts survey of 399 physicians, 84% were likely to prescribe a new drug if it was endorsed by a KOL.
TABLE 2: The foundations and pillars of medical affairs
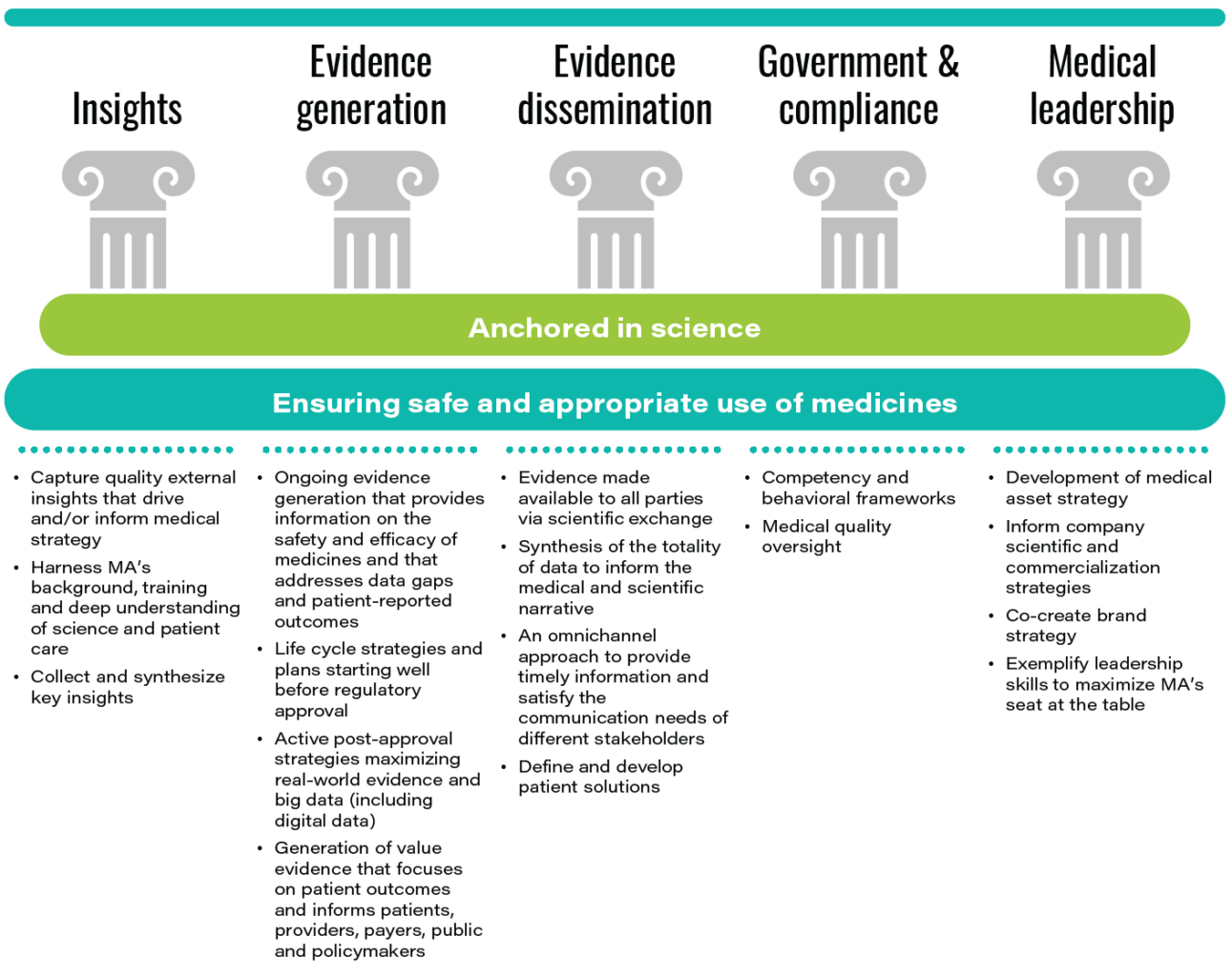
Source: Medical Affairs Professional Society.
Historically, the process of identifying KOLs involved teams of medical science liaisons subjectively mining siloed data in the hope of finding the right healthcare provider with the right expertise. The emergence of data capture and analytics technology has enhanced liaisons’ ability to gain deeper insights into providers’ points of view through KOL mapping. KOL mapping is a data- and analytics-driven approach to identifying KOLs on a geographic level. Mapping identifies influential thought leaders who can create buzz among their peers.
For example, Ontosight Influence from Innoplexus uses artificial intelligence to identify and rank KOLs based on information drawn from various sources such as publications, medical congresses, clinical trials, medical societies, health technology assessments, regulatory bodies, and advocacy groups and then provides liaisons with recommendations on KOLs that match their business strategies. Founder and CEO Dr. Gunjan Bhardwaj noted “Innoplexus’ goal is to decentralize science.”
Another notable vendor is H1, dubbed the LinkedIn for the healthcare industry. H1 provides pharma and life science customers with a platform called HCP Universe. With more than 10 million healthcare provider profiles, HCP Universe uses machine learning to construct profiles of the most influential doctors in each disease area based on credentials, special-ties, publications, clinical trials, conference presentations, social media posts, patient visits and medical claims data. Moreover, the platform provides insights into the patient groups a provider is likely to treat and the provider’s potential interest in participating in clinical trials.
H1 HCP Universe platform
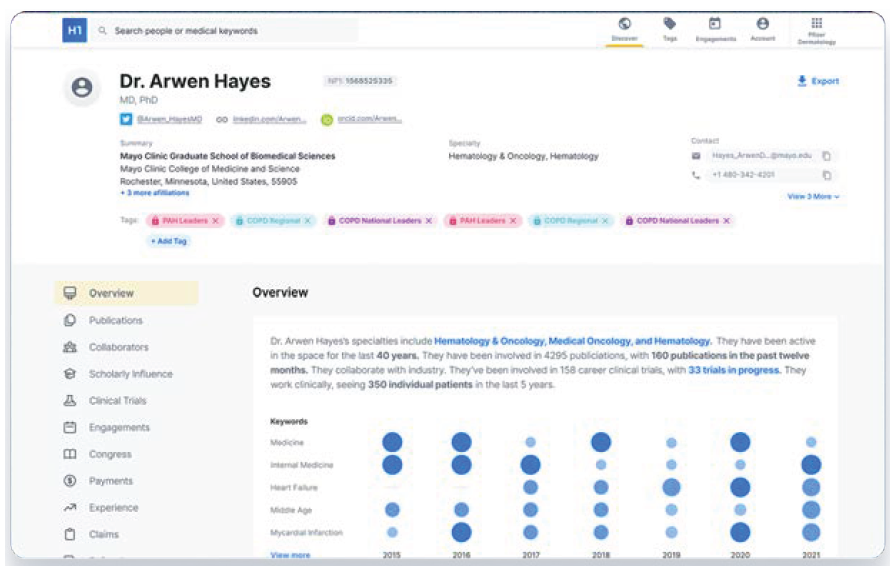
Source: H1.
Virtual engagement and collaboration
Prior to 2020, MA teams viewed virtual engagement with a high degree of skepticism, as face-to-face interactions were still the compliant gold standard. For example, in multi-center clinical trials, communication between coordinating centers and trial sites is essential. A commonly used engagement tool is in-person investigator meetings, hosted and paid for by drug sponsors, where site personnel (including providers) can train on protocols, receive trial updates, learn and share best practices, and troubleshoot problems.
These in-person meetings tend to be extremely costly: Sponsors can expect to pay $250,000 to $1 million for a typical Phase III in-person investigator meeting. While the pharma industry debated whether these face-to-face meetings warranted their high cost, the industry’s aversion to any change that puts compliance and overall trial success at risk always seemed to trump the arguments against them.
The pandemic, however, has dramatically shifted the debate. Pharma has rethought its overall model for engaging with stakeholders and particularly with healthcare providers, who now find themselves working longer hours with more restrictions around travel. With limited options for in-person interactions, Zoom and Microsoft Teams have met some of pharma’s immediate communication needs. But these tools lack the specific functionality needed by MA teams to support complex, collaborative, on-going work. One notable vendor bridging the gap is ArcheMedX.
Ready engagement platform by ArcheMedX

Source: ArcheMedX.
ArcheMedX uses an educational engagement model as the backbone of its Ready engagement platform. Ready can augment or replace in-person investigator meetings by transforming study documents into on-demand interactive virtual learning experiences. Additionally, the Ready platform uses behavioral science to develop insights trial sponsors or clinical research organizations can use to identify which trial personnel are trained and ready to execute studies and which may need supplemental training. The platform can then automatically re-engage with those in need of supplemental training.
Within3 provides a virtual engagement solution called Connect that combines video call features with asynchronous sessions. Within3’s compliant asynchronous format enables healthcare providers to log in and participate any time regardless of location or scheduling constraints. Within3 CTO Jason Smith explained that “Connect gives life sciences organizations the power to foster active meaningful discussions and share critical information among a global group of participants in a flexible and inclusive format.”
Another vendor focused on MA teams’ engagement needs is Alucio. “Designed for MSLs by MSLs,” the company’s Beacon platform supports both virtual and in-person meetings with healthcare providers and is designed for real-time content management and compliant scientific exchange. The solution also captures and stores relevant meeting data such as platform usage, meeting activity, content viewing and content sharing for reporting and analysis.
Alucio’s Beacon platform
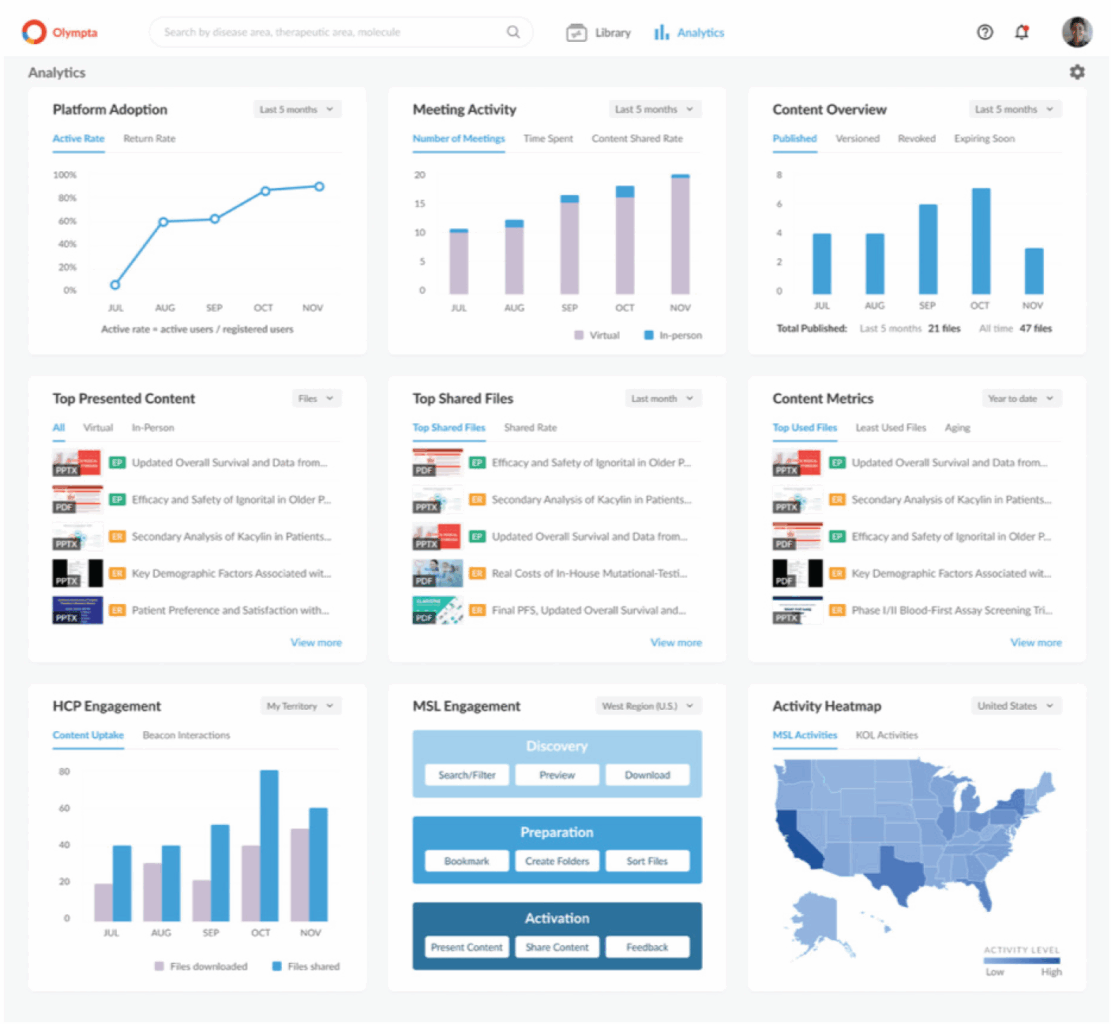
Source: Alucio.
Insights and evidence generation
The healthcare community’s increased focus on cost and outcomes has made generating insights and evidence to demonstrate therapeutic value a critical objective for MA. The pharma industry is swimming in data, such as data from randomized controlled trials, post-marketing safety studies, health economic outcomes data, scientific and medical literature, patient organizations, and data from KOLs and advisory boards; however, deriving meaningful insights and evidence from this data is challenging.
For example, while medical and scientific literature contains data on unmet medical needs, competitive intelligence, current standards of care, epidemiology and treatment pathways, searching journals can be an arduous and time-consuming task, especially given the number of industry publications is estimated to be doubling every three years. As the custodians and scientific voice of validated non-promotional information, MA teams are eager to adopt technology that can overcome the challenge of capturing, managing and interpreting complex data from multiple sources.
One notable provider of such technology is Sorcero via its Language Intelligence platform. Sorcero’s platform uses deep-learning algorithms and disease-focused ontologies to organize and analyze medical and scientific publications as well as internally generated literature. MA teams can use the platform to pull content from multiple sources, including Pubmed, Europe PMC, and LitCovid, into one centralized location to quickly uncover valuable insights. According to co-founder and Chief Commercial Officer Richard Graves, “The platform’s unified view not only provides MA teams the ability to track trends but also to beef up their own literature submissions.”
Sorcero’s Intelligent Literature Monitoring
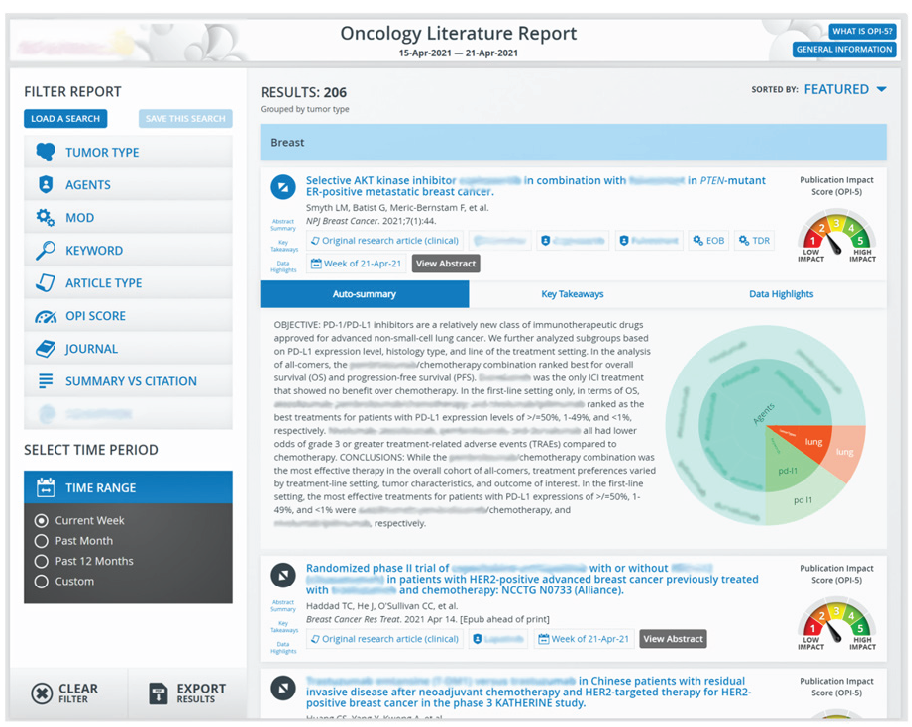
Source: Sorcero.
The changing pharma regulatory and reimbursement landscape has led to the growth and use of real-world evidence (RWE). RWE is generated by analyzing or synthesizing data collected from sources outside randomized controlled trials, such as electronic medical records and disease registries, to address issues that might go unnoticed in a trial, such as potential benefits and adverse events. RWE continues to grow in importance not only because of its utility in deter-mining safe and effective uses of drugs but also because it provides evidence needed to support treatment access and reimbursement coverage.
Lynx.MD offers a differentiated approach to RWE. The company’s platform focuses on security and privacy by providing pharma companies access to de-identified patient and outcomes data through a secure, regulation-compliant private cloud controlled by healthcare providers. Unlike other solutions that seek to collect complete original data and extract de-identified data, the Lynx solution leaves the complete original data at healthcare providers’ sites and only removes de-identified data. This alleviates concerns about data leaks and inappropriate use while providing pharma with evidence of how drugs perform in the real world.
MedaSystems also offers unique solutions for leveraging RWE. The company’s platform focuses on expanded access programs, also known as early access or compassionate use programs. For patients suffering from serious illnesses who are unable to enroll in clinical trials and have no viable treatment options available, these programs provide physicians and patients access to pre-approval, investigational drugs outside clinical trials.
MedaSystems expanded access platform
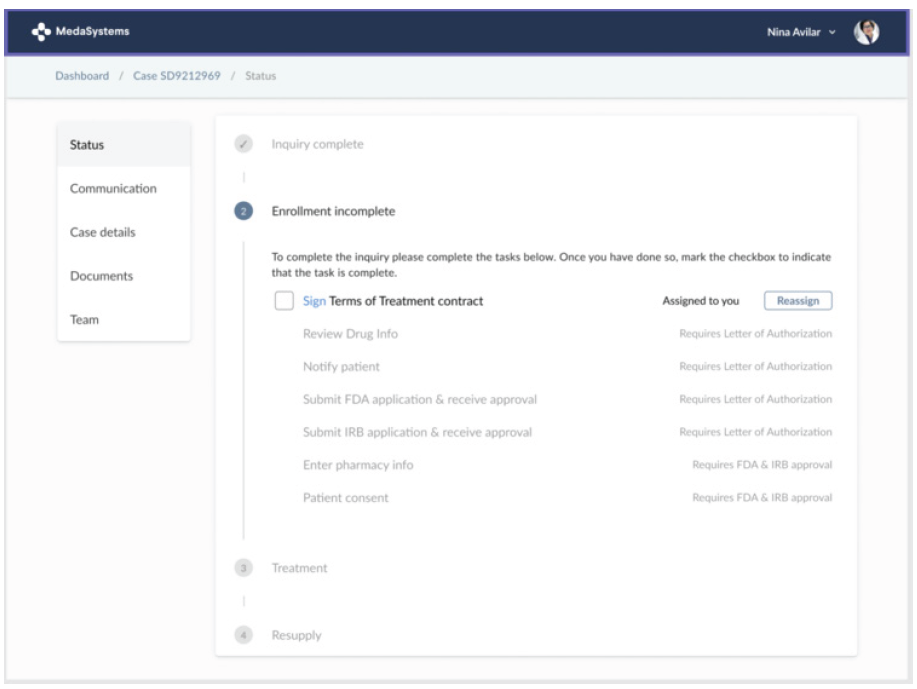
Source: MedaSystems.
As a result of the 21st Century Cures Act, all companies with investigational drugs in Phase II or III clinical trials must have an expanded access policy on their website. Expanded access programs come under the purview of MA and provide pharma companies with several benefits when it comes to generating RWE, such as additional safety and efficacy data and an early understanding of healthcare provider and patient experience prior to approval and launch.
MedaSystems CEO Fiona Smythe explained that “thousands of patients and physicians each year seek permission for expanded access, but pharma companies are ill-equipped to handle the growing number of requests. They’re using a hodgepodge of tools, including spreadsheets, to manage their programs and not receiving meaningful data in return.” To address this need, the MedaSystems platform links patients, providers and MA teams on a cloud-based solution that streamlines the early access process and collects and tracks RWE to understand how the therapies perform in patients.
Powerful technologies strengthen pharma’s third pillar
We think the combination of MA’s emergence as pharma’s third pillar with the emergence of powerful digital tools for leveraging the immense, complex data generated by the healthcare community has created a virtuous cycle of value creation. As technology enables MA to be more effective in helping pharma bring life-changing therapies to market, pharma industry efficiency overall increases, all to the benefit of patients and providers and the pharma companies and technology providers themselves.
Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
