Quarterly insights: Pharma IT
What the second Trump term could mean for pharma IT

We explore the potential impact of Trump’s second term on three aspects of the pharma sector and what the changes might mean for pharma IT.
First, we expect pharmaceutical benefit managers (PBMs) to face continued scrutiny from Trump’s incoming FTC chairman, Andrew Ferguson, as part of Trump’s initiative to reduce drug prices. Potential impacts on pharma IT companies include increased opportunities for companies providing solutions for health economic outcomes research, real-world evidence and patient reported outcomes.
Second, we expect the Trump administration to continue its push for expedited drug approvals and funding and tax incentives for research and development, while increasing use of artificial intelligence in drug discovery and development and emphasizing post-market surveillance to monitor patient outcomes. Potential impacts on pharma IT companies include increased opportunities for those focusing on real-world evidence and pharmacovigilance data as well as those focused on streamlining processes for collecting data and submitting it to the FDA.
We expect the administration’s focus on bolstering domestic manufacturing and reducing U.S. reliance on foreign supply chains to lead to renewed interest in onshoring drug development and manufacturing by both pharma companies and contract development and manufacturing organizations. Potential impacts on pharma IT companies center around opportunities in supply chain logistics.
TABLE OF CONTENTS
- Looking beyond vaccines: Three key areas to focus on
- Renewed focus on drug pricing
- Accelerated drug approvals and regulatory reforms
- Emphasis on domestic manufacturing
- Change likely to create new opportunities
- Pharma IT indexes down year-over-year
- Pharma IT M&A: Notable transactions include Greenphire and BuildClinical
- Pharma IT private placements: Notable transactions include Aqemia and Manas AI
Looking beyond vaccines: Three key areas to focus on
Donald Trump’s return to the presidency has generated a mix of optimism and uncertainty in the pharmaceutical industry. While much of the public discussion has focused on Trump’s selection of Robert F. Kennedy Jr. as secretary of the Department of Health and Human Services and his potential impact on vaccines, in this report we focus on other areas including drug pricing, regulatory reforms and manufacturing, and trade policies to assess the potential impact of Trump’s second term on the pharma sector and what the changes might mean for pharma IT.
Renewed focus on drug pricing
A primary concern for the pharmaceutical sector is the administration’s commitment to lowering drug prices and increasing price transparency. During his first term, Trump proposed several policies aimed at reducing the cost of prescription drugs. These included efforts to negotiate drug prices for Medicare, reform the pharmacy benefit manager (PBM) system, and implement international pricing reforms. Trump’s “America First” healthcare plan included provisions aimed at lowering drug costs by enabling Medicare to negotiate directly with pharmaceutical companies, an effort to make the system more transparent and competitive.
This last initiative (the biggest pharmaceutical regulation change in several years) came to fruition under the Biden administration’s Inflation Reduction Act (IRA) of 2022, which granted Medicare authorization to negotiate prices for its costliest prescription drugs. Prices for the first ten Medicare-negotiated drugs were released last August and are expected to go into effect in 2026. Although it’s unclear whether the Trump administration supports this change as it was laid out in the IRA, the Centers for Medicare and Medicaid Services recently announced plans to proceed with negotiating Medicare prices for the next 15 drugs selected under the IRA, listed in Table 1.
Table 1: Next 15 drugs slated to be negotiated by CMS (data from November 2023 to October 2024)
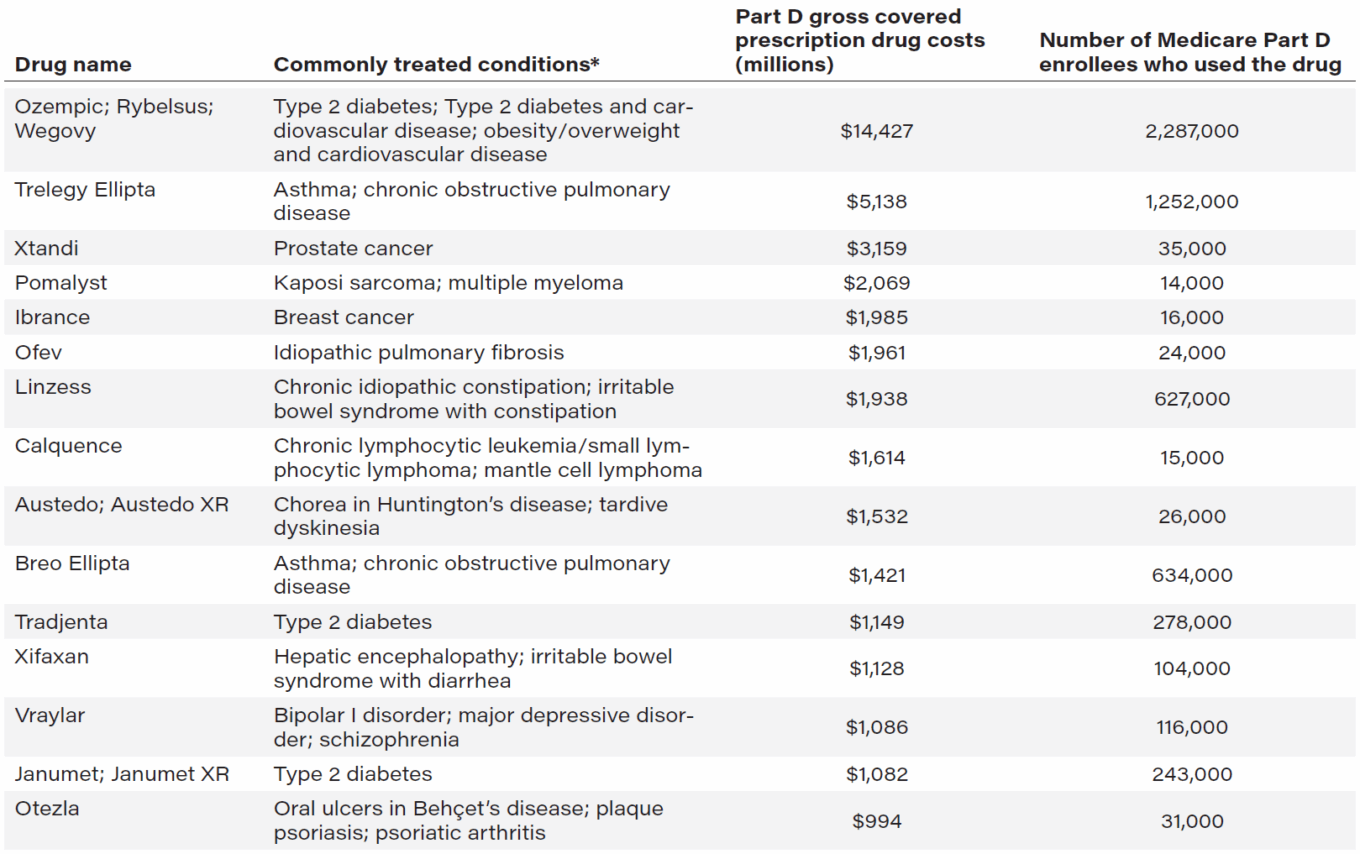
Source: Medicare Drug Price Negotiation Program: Selected Drugs for Initial Price Applicability Year 2027, January 2025. Centers for
Medicare & Medicaid Services.
Notes: *The commonly treated conditions are limited to conditions for which prescription drug coverage is currently available
under the Medicare Part D program.
Pharma companies hope Trump will focus more on cracking down on PBMs to lower drug prices than on manufacturers themselves. Trump has been a very vocal critic of PBMs and has accused them of being a key contributor to higher drug prices. In December 2024, Trump stated, “We’re going to knock out the middleman. We’re going to get drug costs down at levels that nobody has ever seen before.”
As a reminder, PBMs are intermediaries in the healthcare system that play an important role in managing prescription drug benefits for health insurers, employers and government programs. Their primary function is to negotiate drug prices with pharmaceutical manufacturers, establish formularies (lists of covered medications), and determine reimbursement rates for pharmacies. PBMs leverage their purchasing power to negotiate discounts and rebates from drug manufacturers, aiming to lower costs for insurers and patients. They also oversee drug utilization by implementing cost-control measures such as prior authorization, step therapy and tiered pricing. However, PBMs have faced criticism for a lack of transparency, as the savings they negotiate do not always directly translate into lower prices for consumers.
The “Big Three” PBMs – CVS Caremark, Express Scripts and OptumRx – process roughly 80% of U.S. drug claims. In July 2024, The Federal Trade Commission issued an interim report titled “Pharmacy Benefit Managers: The Powerful Middlemen Inflating Drug Costs and Squeezing Main Street Pharmacies.” In the report, the FTC said PBMs “wield enormous power and influence over patients’ access to drugs and the prices they pay. This can have dire consequences for Americans, with nearly three in ten surveyed Americans reporting rationing or even skipping doses of their prescribed medicines due to high costs.”
PBM landscape
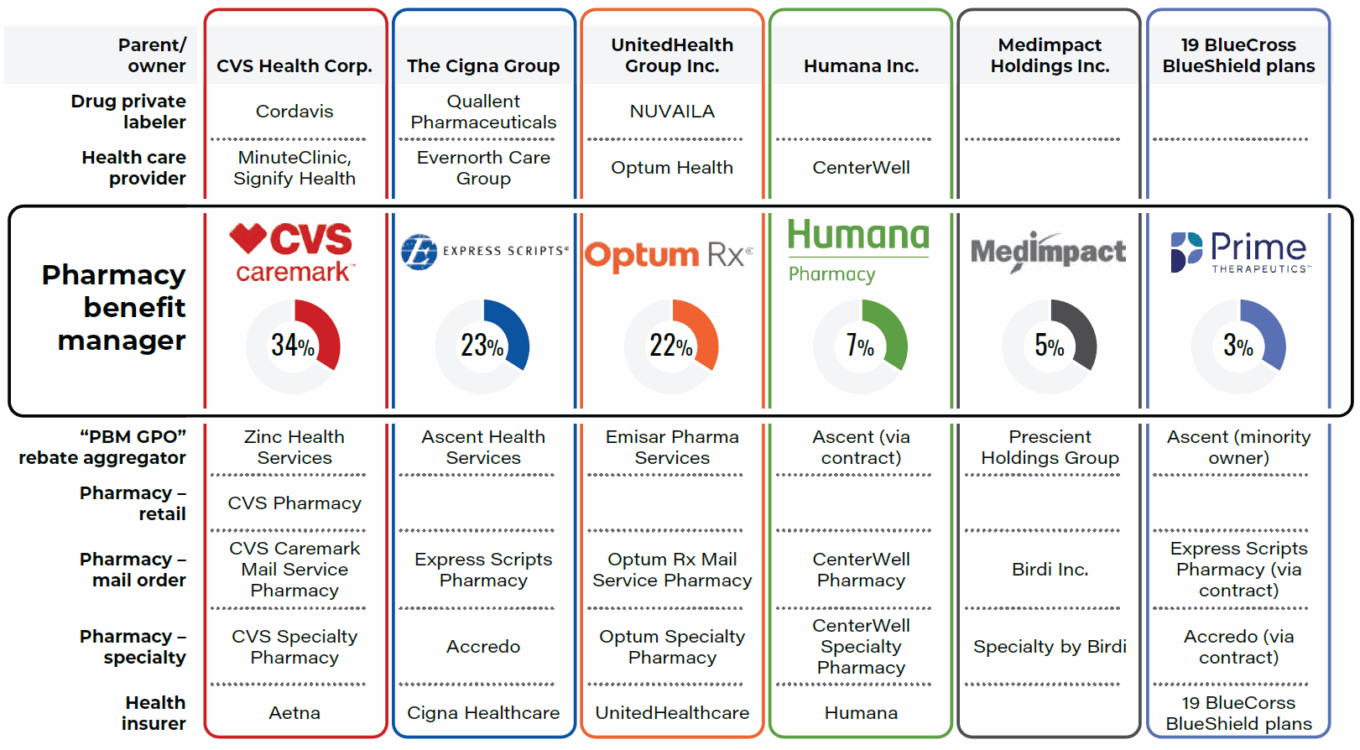
Source: Federal Trade Commission.
In September 2024, the FTC sued the Big Three, accusing them of engaging in anticompetitive and unfair rebating practices that have artificially inflated the list prices of insulin drugs, impaired patients’ access to lower-list-price products, and shifted the cost of high insulin list prices to vulnerable patients. In January 2025, the FTC published a second interim staff report on the prescription drug middleman industry, which focused on PBMs’ influence on specialty generic drugs, including significant price markups by PBMs on drugs for cancer, human immunodeficiency virus, and a variety of other conditions. The report said the Big Three marked up numerous specialty generic drugs dispensed at their affiliated pharmacies by hundreds or thousands of percent.
Such significant markups allowed the Big Three and their affiliated specialty pharmacies to generate more than $7.3 billion in revenue beyond the drugs’ price from the wholesaler to the provider from 2017-2022. We expect PBMs to face continued scrutiny from Trump’s incoming FTC chairman, Andrew Ferguson. Prior to his appointment, Ferguson served as an FTC commissioner and voiced support for the FTC’s reports on PBMs. One pharma executive at a top-ten pharma company we spoke with sees PBM reform as low-hanging fruit in Trump’s initiative to reduce drug prices.
We believe any PBM reform will result in greater transparency in drug pricing and ultimately compel pharmaceutical companies to generate more evidence to demonstrate value to payers in the form of health economic outcomes research (HEOR), real-world evidence (RWE) and patient reported outcomes (PRO). We believe pharma IT companies with solutions focused in those areas stand to benefit from PBM reform.
Accelerated drug approvals and regulatory reforms
One of the significant impacts of the first Trump administration was changes in regulatory oversight. Under the leadership of Scott Gottlieb, the FDA made several moves to streamline drug approval processes. The Trump administration aimed to reduce bureaucratic hurdles and expedite the introduction of new therapies. This focus on regulatory reform led to initiatives like the Drug Competition Action Plan, designed to encourage competition in the generic drug market and expedite generic drug approvals.
Additionally, Trump’s approach to regulatory reforms also focused on the concept of right-to-try legislation. This gave terminally ill patients access to experimental treatments not yet approved by the FDA. The emphasis on patient access to unapproved treatments created an environment where pharmaceutical companies were encouraged to speed up innovation to capture a more immediate market.
All in, the Trump administration’s emphasis on streamlining regulatory processes during its first term resulted in more novel drug approvals. We expect the new Trump administration to continue its push for expedited drug approvals alongside funding and tax incentives for research and development, particularly for treatments addressing rare diseases like cancer and neurological disorders. We expect these changes to be accompanied by increased emphasis on post-market surveillance to monitor patient outcomes. This will potentially enhance the FDA’s use of real-world evidence and pharmacovigilance data to approve drugs faster.
We also think the Trump administration could drive increased investments in drug discovery and development that leverages artificial intelligence (AI) as the administration looks to position the United States as a leader in digital health technology. In early January, the FDA issued draft guidance on the use of AI to support regulatory decisions about drugs’ and biological products’ safety, effectiveness and quality. This was the first guidance the agency had issued on using AI for developing drugs and biological products. Pharma companies adopting AI-driven platforms may benefit from federal support or incentives in the form of research and development tax credits.
We think the administration’s efforts to accelerate drug approvals may hit a roadblock if there isn’t more clarity on staffing after the recent layoffs at the FDA. The FDA employs more than 18,000 people, and if Trump’s nominee to lead the agency, Dr. Marty Makary, follows through with more agency job cuts, the reduced headcount could extend review times for drug approvals.
Along with real-world evidence (RWE) and pharmacovigilance, we think pharma IT companies with AI solutions that can drive greater efficiencies in trial data collection and help simplify data submission to the FDA stand to benefit from changes under the second Trump administration.
Emphasis on domestic manufacturing
According to the United Nations Comtrade database on international trade, pharmaceuticals have become the United States’ second-largest manufactured import, having totaled $178 billion in 2023 as U.S. spending on healthcare and drug prices increased and as leading multinational pharmaceutical companies continued to offshore drug manufacturing.
As Table 2 indicates, the U.S. pharmaceutical sector depends extensively on global supply chains for both finished medications and active pharmaceutical ingredients (APIs). The COVID-19 pandemic exposed the United States’ heavy dependence on overseas manufacturing for critical drugs and ingredients. According to a 2023 Washington University study titled “U.S. Generic Pharmaceutical Industry Economic Instability,” 83% of the top 100 generic medicines prescribed in the U.S. have no U.S.-based API source. Over 90% of the most frequently prescribed antivirals and antibiotics have no U.S. API manufacturing source.
TABLE 2: U.S. imports of pharmaceutical products (dollars in billions)
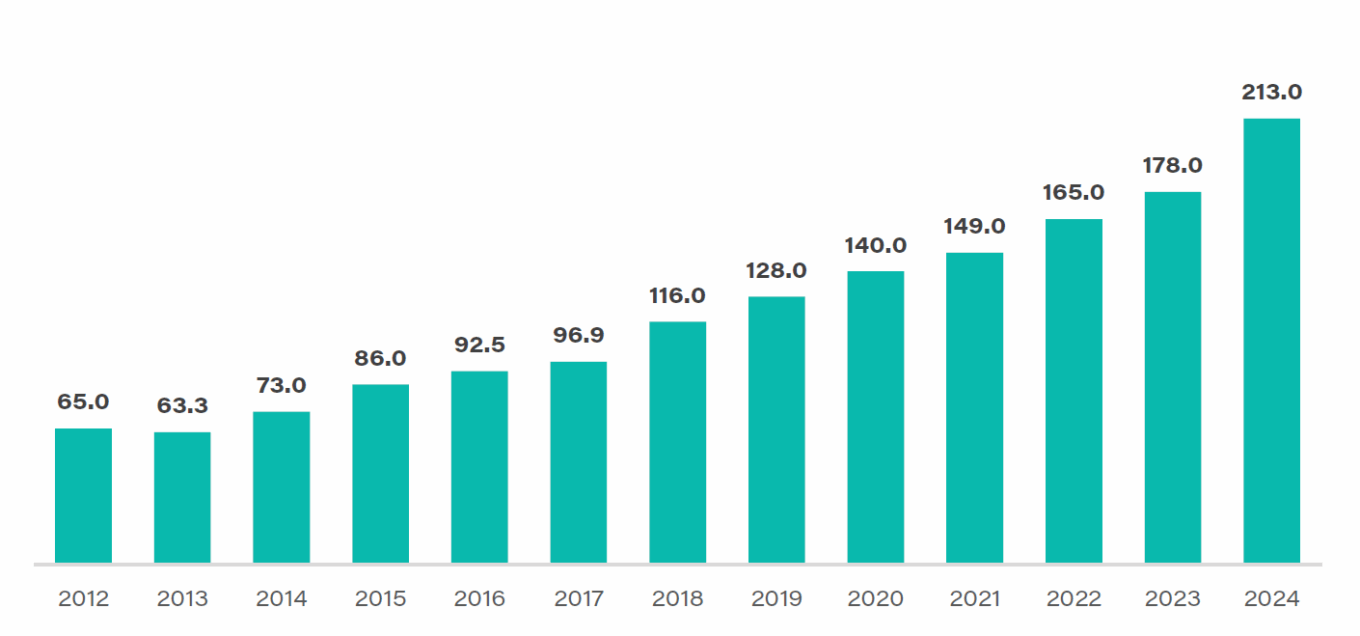
Source: United Nations Comtrade database on international trade.
Trump has been a vocal supporter of imposing tariffs to bolster domestic manufacturing and reduce U.S. reliance on foreign supply chains. On Feb. 1, Trump announced new tariffs on imports from Canada, Mexico and China, citing concerns over illegal immigration and the influx of illicit drugs, particularly fentanyl, into the United States. The tariffs, which were to be effective Feb. 4, included a 25% levy on imports from Canada and Mexico and a 10% levy on imports from China. On March 4, Trump added an additional 10% tariff on all imports from China. Beyond Canada, Mexico and China, Trump has signaled that the European Union may be next in line for 25% tariffs on its exports to the U.S. These tariffs are comprehensive, affecting a wide array of products, including pharmaceuticals.
The tariffs on imported drugs aim to incentivize companies to establish or expand manufacturing facilities in the United States. Although the Biosecure Act, which is intended to block U.S. federal funding for specific Chinese contract manufacturers, has not advanced in Congress, its potential approval coupled with the administrations’ America First agenda could, in our view, lead to renewed interest from both pharma companies and contract development and manufacturing organizations (CDMOs) in moving drug development and manufacturing to the United States.
We note Merck just opened a $1 billion vaccine manufacturing plant in North Carolina to boost U.S. production, while Eli Lilly recently announced plans to invest $27 billion to build four production facilities in the U.S. As the administration continues its push to increase domestic manufacturing and strengthen supply chains, we think U.S.-based CDMOs with excess capacity stand to benefit either from increased volume or by being acquired. Additionally, we think supply-chain logistics software and service providers will see more demand.
Change likely to create new opportunities
Regardless of the merits or specific contours of pharmaceutical industry changes under the Trump administration, one thing seems certain: Innovative and nimble pharma IT companies will likely be able to identify and capitalize on new opportunities for growth. We’re hopeful this means the area will remain a fertile field for creating wealth and improving quality of life through improved healthcare.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
