Quarterly insights: Pharma IT
Clinical trial supply challenges create opportunity for tech

Clinical trial supply chains are deeply interconnected. However, teams throughout the process often work in silos, making clinical trial supply management extremely complex and difficult.
Given the pivotal role of supply chains in patient safety, clinical trial efficiency and overall trial success, pharmaceutical companies are increasingly focused on improving supply chain management, most notably by making greater use of supply chain software.
We examine four areas where clinical trial supply chain management software is having a high-profile impact: randomization and trial supply management and forecasting, sample management, condition monitoring, and cell and gene therapy. We also profile some of the technology solutions innovative companies offer in each of these areas.
TABLE OF CONTENTS
Includes discussion of six private companies
- Pandemic experiences highlighted the need to address trial supply challenges
- Multiple pain points in supplying clinical trials
- Randomization and trial supply management and forecasting
- Sample management
- Condition monitoring
- Cell and gene therapy
- Advanced technology meeting the needs of advanced therapeutics
- Commercialization index in line with markets; eClinical far behind
- Pharma IT M&A: Notable transactions include CorEvitas and Clario
- Pharma IT private placements: Notable transactions include Clinetic, QuantHealth
Pandemic experiences highlighted the need to address trial supply challenges
Clinical trials are the backbone of medical research, serving as a key bridge between drug development and patient care. As we have discussed in our previous reports, clinical trials are expensive and time consuming, and any delay pushes out the timeline for potential approval, meaning lost opportunities to improve patients’ lives and unrecoverable revenue. Supplying clinical trials with trial therapies, or investigational medicinal products (IMPs), is a pivotal part of the clinical trial process that can affect trials’ success, cost and speed. The critical nature of clinical trial supply management became especially apparent during the pandemic, which highlighted supply chain weaknesses in areas such as third-party logistics, forecasting, inventory visibility and staffing. With this in mind, pharmaceutical companies are increasingly turning to technology-based solution providers to handle some or all of their clinical trial supply needs.
Multiple pain points in supplying clinical trials
Clinical trial supplies are the investigational medicinal products and other materials and equipment needed to effectively conduct clinical trials. This includes everything from drugs being tested to patient biological samples to the packaging and labeling used in moving them from one location to another. Managing clinical trial supplies involves a range of activities, from planning and forecasting to procurement and distribution. Clinical trial supply chains are deeply interconnected. Expected enrollment levels at trial sites affect supply and logistics decisions made upstream. Unfortunately, most teams throughout the process work in silos, making clinical trial supply management extremely complex and difficult with challenges that include managing inventory levels, ensuring timely delivery of investigational drugs, and maintaining regulatory compliance and patient safety.
Quite often, the process succumbs to mistakes and delays stemming from lack of visibility and understanding of what’s actually happening in supply chains. A multitude of considerations affect how clinical trial supplies need to move throughout clinical trial processes. Examples include site-level participant enrollment and retention (which can vary widely within a geography), variable dispensing caused by unpredictable titration, schedule switching, and weight-based dosing, all of which can indirectly affect trial-site demand and resupply strategies. Another example is trial protocol amendments, which may add new treatment arms, extend visit schedules, and add countries, all of which affect the supply chain.
Challenges in the clinical supply chain
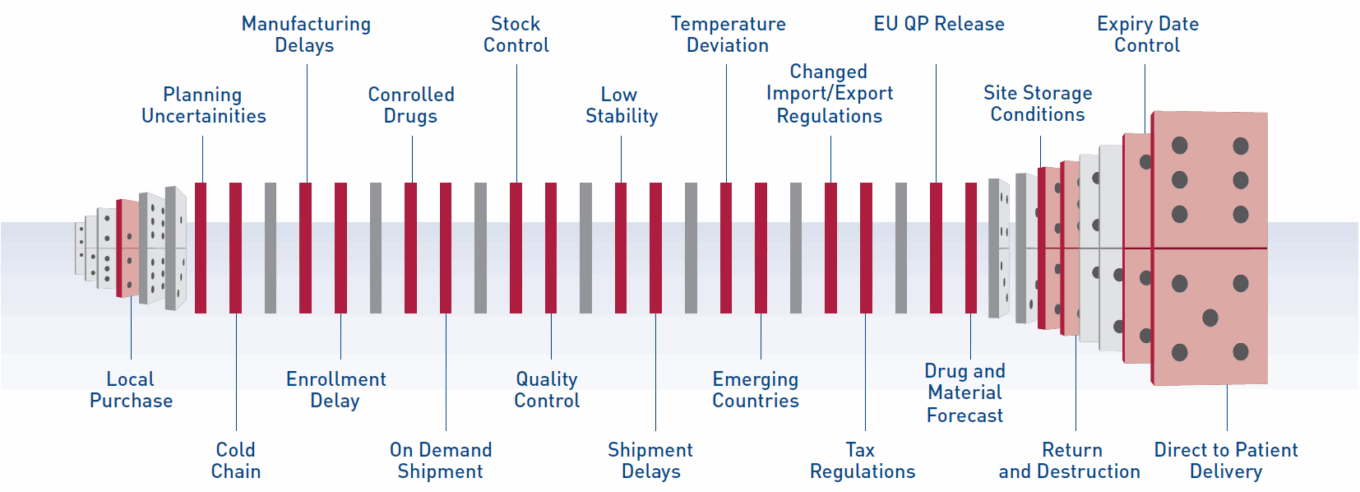
Source: Parexel.
Traditionally, pharma companies have overproduced, overstocked and overdistributed investigational drugs to minimize the chance shortages put patient safety and overall trial success at risk. They build these supply buffers knowing most of the drug kits will expire before use. While some overage is always necessary, the level of supplies in most clinical trials is much higher than would ever be needed.
According to our conversations with industry executives, conventional wisdom says trials need 40-50% overage just to run a trial. This is due to stocking investigational drugs in the supply chain in anticipation of needs that may never materialize. Whether these levels are enough, too much, or too little is a function of trial design, drug expiry and supply chain logistics. For example, one executive we spoke with described a situation involving a top-ten pharmaceutical company in which about $90 million worth of drug in each year of the trial was never even distributed and was ultimately wasted. Despite all this, supply buffers alone are not sufficient to minimize clinical trials’ vulnerability to supply chain problems.
Given the pivotal role of supply chains in ensuring patient safety (by making sure drugs are available at trial sites when needed) and determining clinical trial speed, efficiency and overall trial success, pharmaceutical companies are increasingly focused on improving supply chain management, most notably by making greater use of supply chain software. We examine four areas where clinical trial supply chain management software is having a high-profile impact: randomization and trial supply management and forecasting, sample management, condition monitoring, and cell and gene therapy.
Randomization and trial supply management and forecasting
Randomized controlled trials are trials in which participants are randomly assigned to one of two or more clinical interventions and/or placebos. Randomized controlled trials are the most scientifically rigorous method of hypothesis testing and are regarded as the gold standard for evaluating the effectiveness of interventions. Randomization in clinical trials is important as it prevents bias in selecting which patients receive investigational products and which receive placebos or comparators. Randomization helps balance allocations between patient groups based on predetermined criteria such as age, sex and smoking.
Most blinded, late-stage, randomized clinical trials package study drugs (active or placebo) into drug kits for distribution to investigational sites. Randomization and trial supply management (RTSM) is a key clinical trial technology that is crucial to conducting randomized controlled trials. RTSM is also known as interactive response technology (IRT). RTSM software connects clinical trial supply management and clinical operations. RTSM software enables critical clinical trial functions, including randomizing patients (who gets the active drug and who gets the placebo), dispensing drugs (ensuring patients receive correct doses), and resupplying trial sites (controlling the flow of drugs from manufacturers to depots to clinical sites).
4G Clinical’s 4C Supply tool
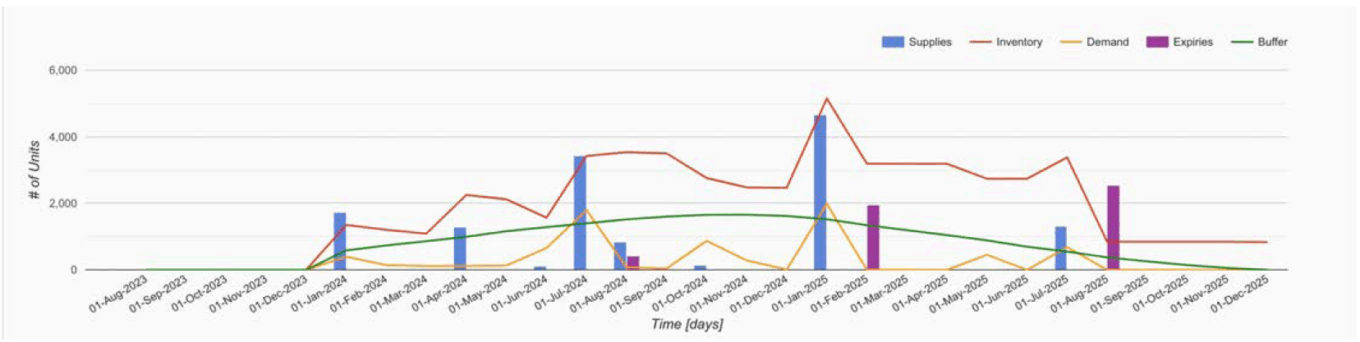
Source: 4G Clinical.
One leading company using innovative technology to enhance RTSM is 4G Clinical. 4G Clinical’s Prancer RTSM platform uses natural language processing (NLP) to accelerate study build and enables trial sponsors to reach first-patient-in status up to 50% faster than with traditional RTSM systems. Instead of using the traditional, weeks-long approach of writing RTSM specifications and handing them to developers to code systems, Prancer reads and understands trial specifications and builds deployable RTSM systems automatically. Advanced algorithms enable 4G Clinical’s supply optimization and forecasting solution, 4C Supply, to run forecasts long before a trial is running and again during execution to develop overall trial supply plans that avoid stock-outs and minimize drug waste.
According to 4G co-founder and CTO Ed Tourtellotte, “One of the hardest things in RTSM is finding and maintaining buffers to cover unpredictable demand at sites. 4G’s Prancer is the only solution on the market that handles this automatically, from the beginning of a trial to the end. This, combined with our next-gen forecaster 4C Supply (including actuals and reforecast), gives our clients quite an edge in launching and smoothly running even the most complex and unpredictable clinical trials.”
Another company is N-SIDE, which focuses on clinical trial supply management forecasting. N-SIDE offers a SaaS suite of solutions and professional services to help pharmaceutical companies optimize their clinical supply chains. With over 12,000 clinical trials and 20 years of experience under its belt, the company’s Supply application uses advanced analytics to help pharma companies make data-driven decisions about overage, packaging, sourcing, IRT setup and depot shipments. It also helps them re-evaluate and monitor assumptions about recruitment, site demand, participant drop-out, participant weight and titrations.
N-SIDE supply app workflow
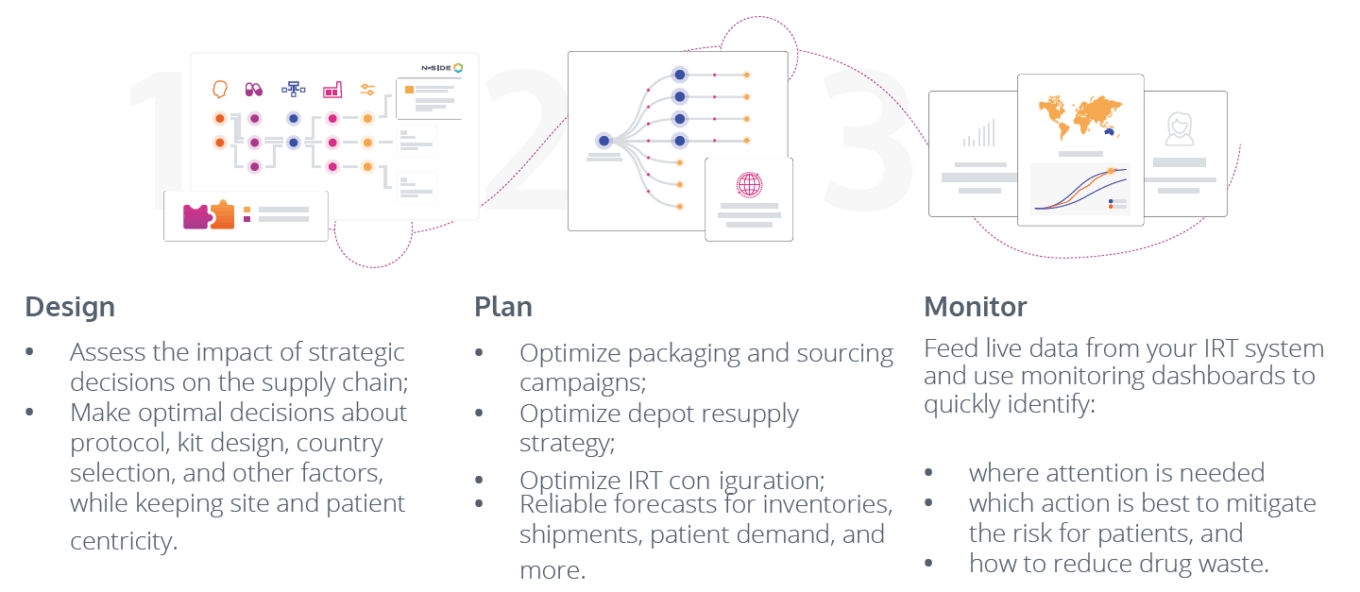
Source: N-SIDE.
Korio, co-founded by RTSM industry veterans Ryan Keane and Chuck Harris, takes a tech-forward approach to RTSM that focuses on reusability, repeatability, system performance and overall system quality. According to CEO Ryan Keane, “Korio’s functionality is nested in a modern platform architecture that enables Korio employees to successfully navigate long-standing quality and scalability issues to ensure our customers’ supply chains run smoothly and patient dosing is uninterrupted in today’s complex and ever-changing clinical trial designs. Korio’s adaptable platform learns each customer’s unique preferences and settings, which in turn enables consistent and predictable RTSM outcomes, creating a virtuous cycle of early and sustainable customer and Korio employee success.”
Korio’s new study intake
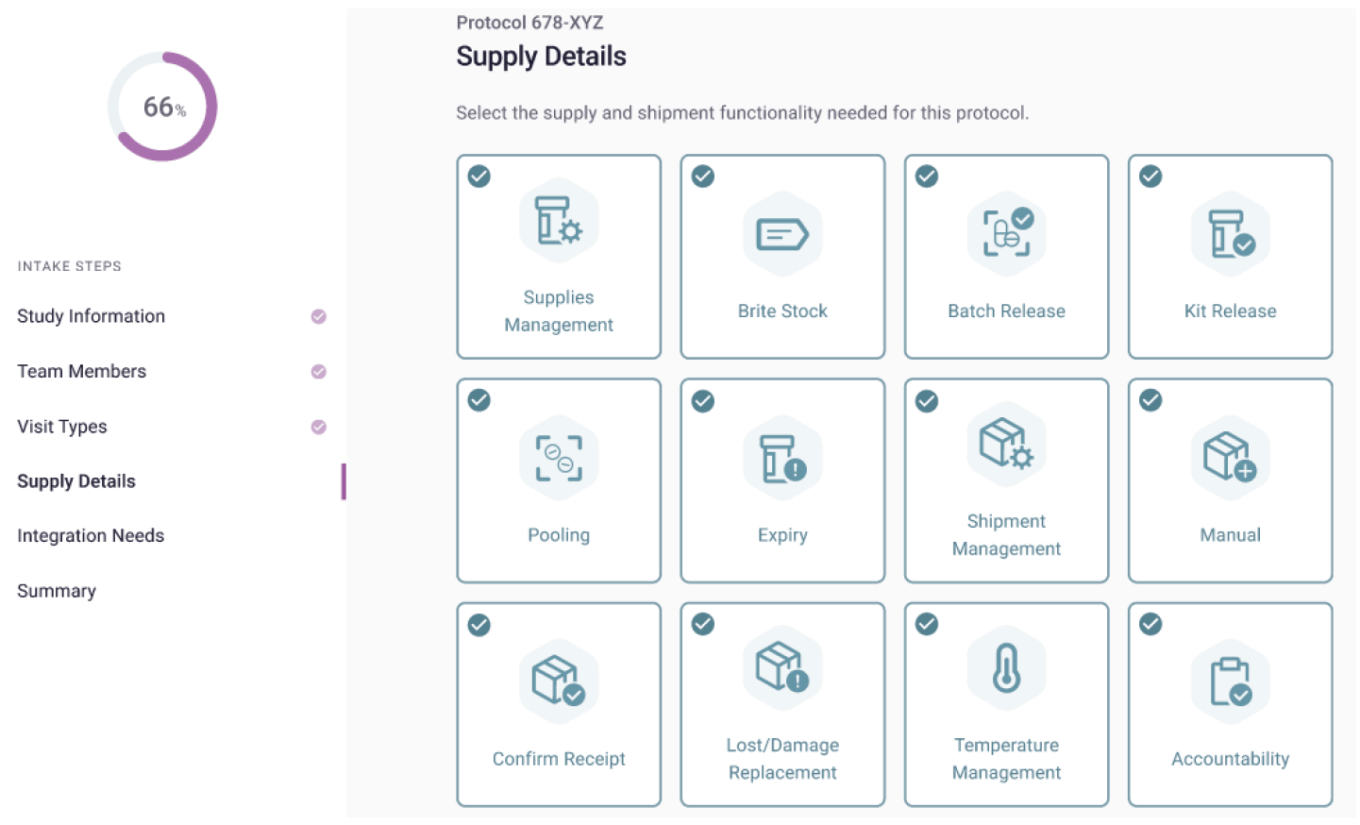
Source: Korio.
Sample management
Clinical trials depend heavily on analyzing biological samples for general screening purposes, such as determining patient eligibility and biomarker profiling. Throughout trials, samples can be analyzed for pharmacokinetic and pharmacodynamic assessments, disease biomarkers, and safety monitoring. Whether blood, hair, urine, saliva, stool or tissues, samples need to go to labs for analysis. Sample management is the process of collecting, processing, storing, shipping, tracking, analyzing and receiving results for clinical trial participant samples. There are many possible paths samples can take depending on trial complexity and requirements. Typically, the journey looks more like a deformed spider web than a linear process. Sponsors must document the whereabouts of all biological samples to prove chain of custody. Gaps in knowledge about samples’ life cycles can lead them to be considered unusable during audits. Manual processes, such as spreadsheets and handwritten notes for handling, storing and coordinating samples, can lead to errors and threaten trial data integrity.
Value chain created with Slope
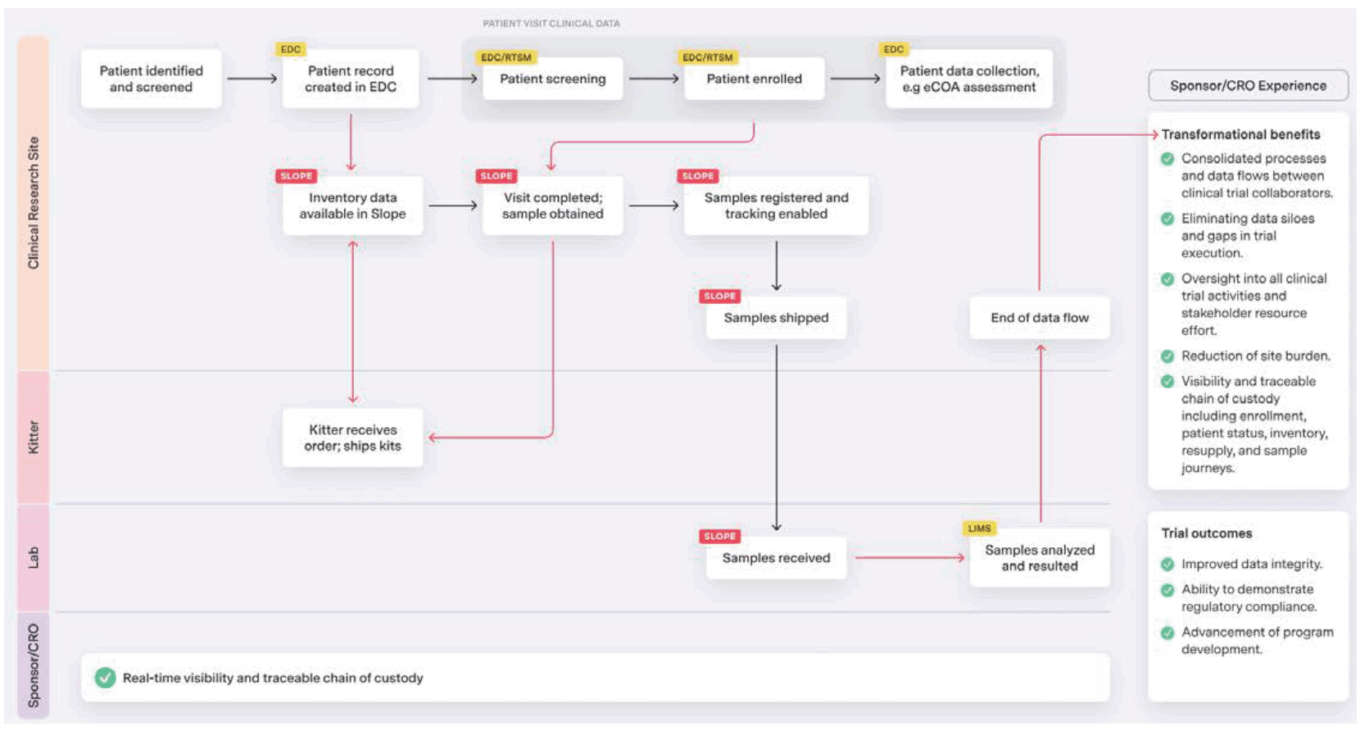
Source: Slope.
One company looking to bring order to the chaotic sample management process in clinical trials is Slope. Slope’s eClinical supply chain management platform aims to reduce clinical trial risks, rein in costs, and promote collaboration among sponsors, sites, distributors, labs, biorepositories and couriers by replacing manual, spreadsheet-driven, and error-prone processes traditionally used to manage samples with a digital platform that enables real-time traceable chain-of-custody information, including use, transit and storage. According to CEO Rust Felix, “There are too many gaps from beginning to end. Sponsors lack sample visibility. Slope is filling in existing gaps, connecting disparate systems and processes, and delivering a fully integrated solution that creates a whole new value chain with real-time visibility for sample management.”
Condition monitoring
An increasing number of drugs used in clinical trials must be transported and stored under very specific conditions, such as low temperatures. These are often high-value specialty drugs being developed in areas such as oncology, neurology, and rare disease. Trial sponsors want to ensure these conditions are maintained throughout these drugs’ journeys through supply chains to ensure smooth trial operation and integrity of results. Additionally, regulatory directives in multiple regions of the world require comprehensive and compliant monitoring of all temperature-sensitive IMPs to ensure products remain safe.
Paxafe’s Contxt platform
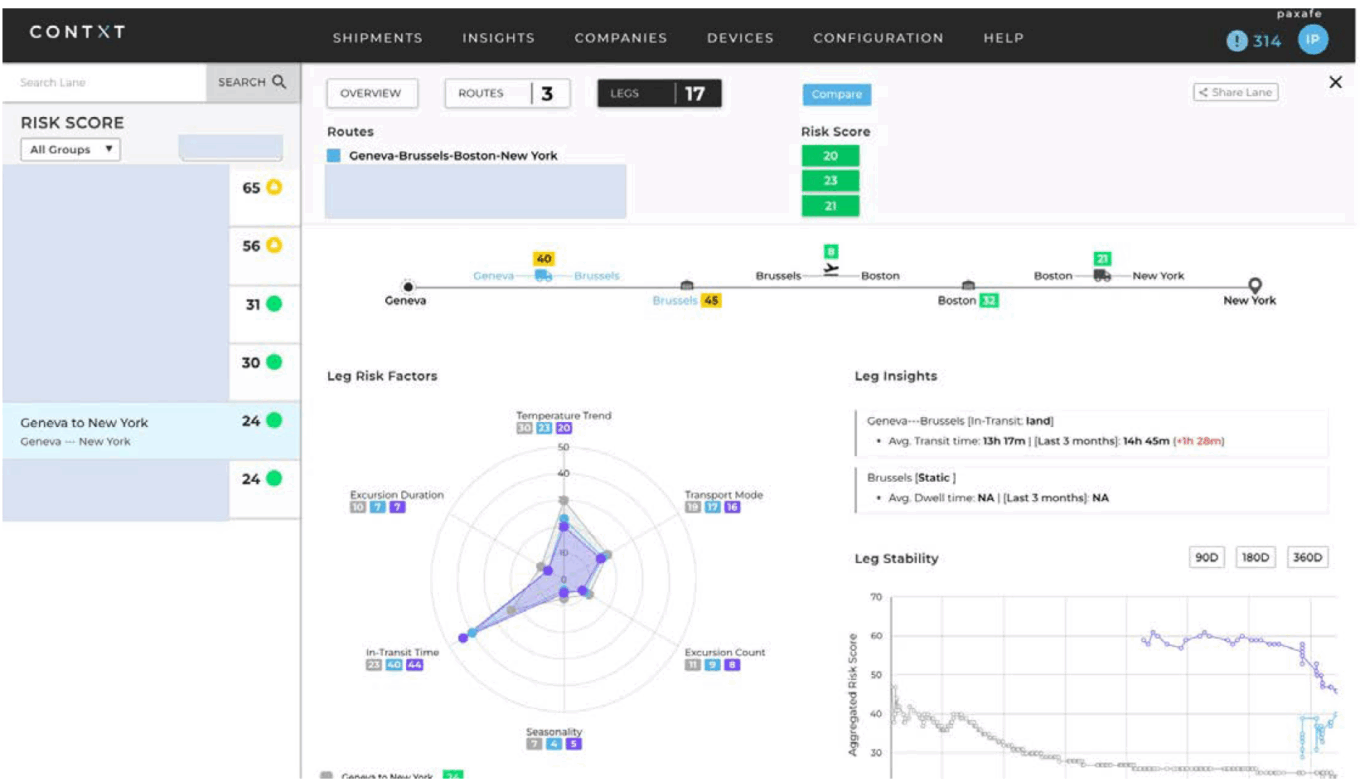
Source: Paxafe.
For temperature-sensitive IMPs, just one unexpected event can lead to a temperature excursion that jeopardizes IMPs’ stability. For example, heavy highway traffic on a hot Friday afternoon between an airport and a trial site could result in a delayed delivery that causes IMPs to reach temperatures that render them ineffective. Or customs officials might open IMP packaging and remove or damage temperature monitors. When IMP temperature excursions are discovered, the IMPs are immediately quarantined, and if stability data for the IMP indicates the excursion exceeds relevant thresholds, the IMP is rejected, potentially resulting in treatment delays and related trial disruptions.
To minimize these risks, trial supply managers use supply strategies that take into account the risks of temperature excursions associated with different modes of transportation, shipping lanes, product packaging, and storage. Visibility is a key part of such strategies. For example, data logging devices have traditionally been used to monitor shipments’ temperatures throughout their journeys and provide records of temperatures when shipments reach their destinations. Today, telemetry-enabled monitoring devices can transmit origin-to-destination shipment condition data to cloud-based software platforms in real time or near real time to provide much greater visibility into products’ supply chain journeys. Companies offering supply chain visibility technology for the pharmaceutical industry include Berlinger, Cargosense, Controlant, FourKites, OnAsset, ParkourSC, project44, and Roambee.
One notable up-and-comer in the space is Paxafe. Paxafe’s platform provides pharma supply chain managers with enhanced real-time visbility and artificial-intelligence-driven decision support for temperature controlled logistics.
CAR-T cell therapy (combined gene and cell therapy)
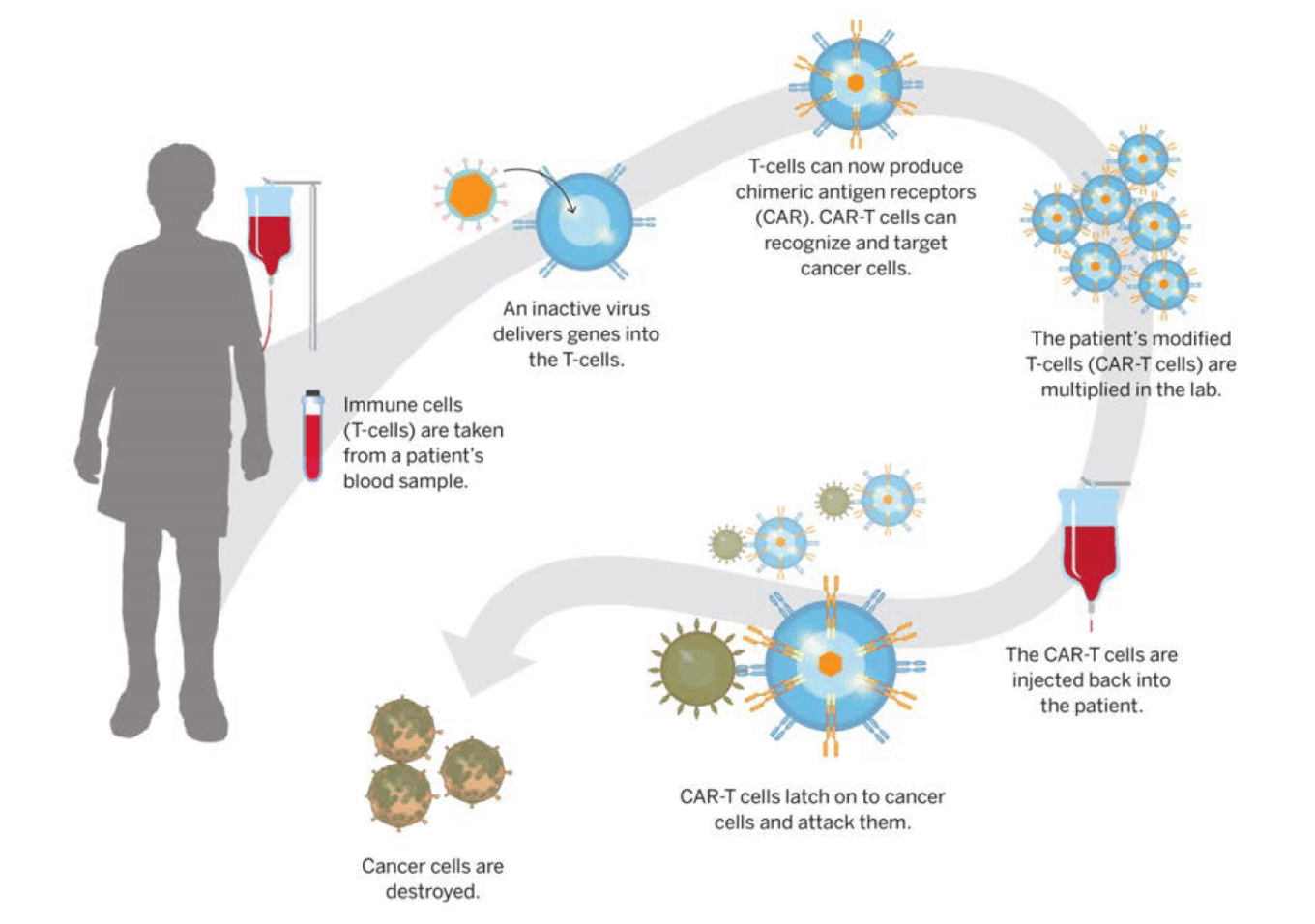
Source: Speeding Cell, Gene Therapy Development. Harvard Gazette, Nov 2019.
Cell and gene therapy
Optimally managing supply chains for cell and gene therapy clinical trials can be especially challenging. Cell therapies aim to treat diseases by restoring or altering certain sets of cells or by using cells to carry a therapy through the body. With cell therapy, cells are cultivated or modified outside the body before being injected into patients. The cells may originate from the patient (autologous cells) or a donor (allogeneic cells). Gene therapy aims to treat diseases by replacing, inactivating or introducing genes into cells either inside the body (in vivo) or outside the body (ex vivo).
The FDA’s 2017 approval of Yescarta and Kymriah, two autologous T-cell oncology therapies, was a landmark moment for the industry and led to an explosion of research and development in cell and gene therapies. At the end of the 2023 first quarter, there were over 1,600 cell and gene therapy clinical trials underway worldwide.
Ongoing cell and gene therapy clinical trials by phase and therapeutic approach
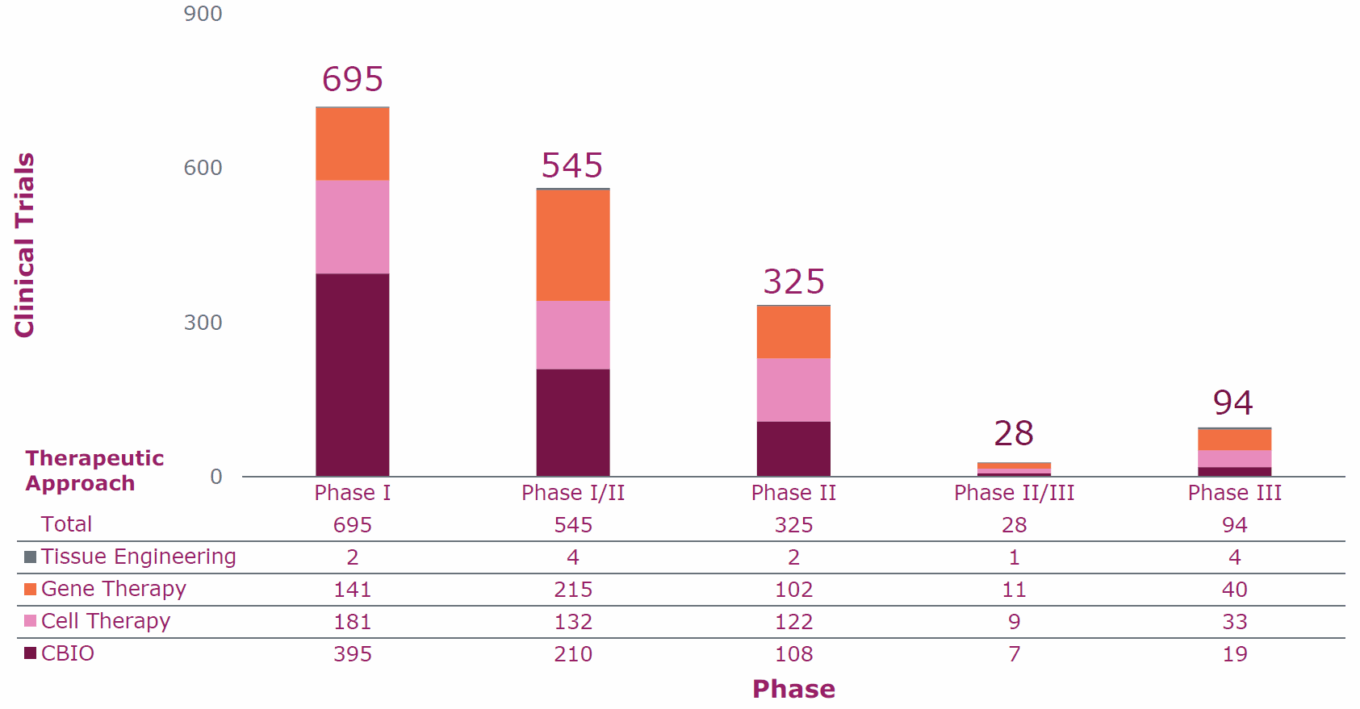
Source: Clinical Trial Data – 2023 Q1. Alliance for Regenrative Medicine, June 2023.
Clinical trials for these therapies require trial personnel to properly and safely manage participant samples, especially since those samples could ultimately be used to create the therapy. For example, a cell therapy might be produced from cells that are collected from a healthy donor and shipped to a clinical site to treat a patient. Or, the cells might be collected from a trial participant, sent to a biomanufacturing site to be genetically modified, and then returned and administered to the same trial participant. Given the complex nature and sensitivity of cell and gene therapies and the samples being shipped, the stakes are even higher than with conventional drugs. A delay, chain-of-custody error, process deviations, or temperature excursion could lead to an immediate life-or-death situation.
These challenges and the growing number of cell and gene therapies under development suggest there will be strong demand for clinical trial supply chain management solutions that can address the special needs of cell and gene therapy clinical trials.
TrakCel, established in 2012, is a leading provider of integrated cell and gene therapy software for orchestrating and precisely managing, controlling and tracking cell and gene therapies from sample collection through manufacturing to treatment delivery, while maintaining “needle-to-needle compliance and traceability.” TrakCel’s OCELLOS is a configurable, integrated, advanced therapy orchestration platform built on Salesforce and designed to meet the needs of cell and gene therapy supply chains. It includes key integrations with processes for shipping, labeling and manufacturing and for establishing and safeguarding chain of custody and chain of identity while capturing full audit trails from every patient.
TrakCel’s OCELLOS Lite platform
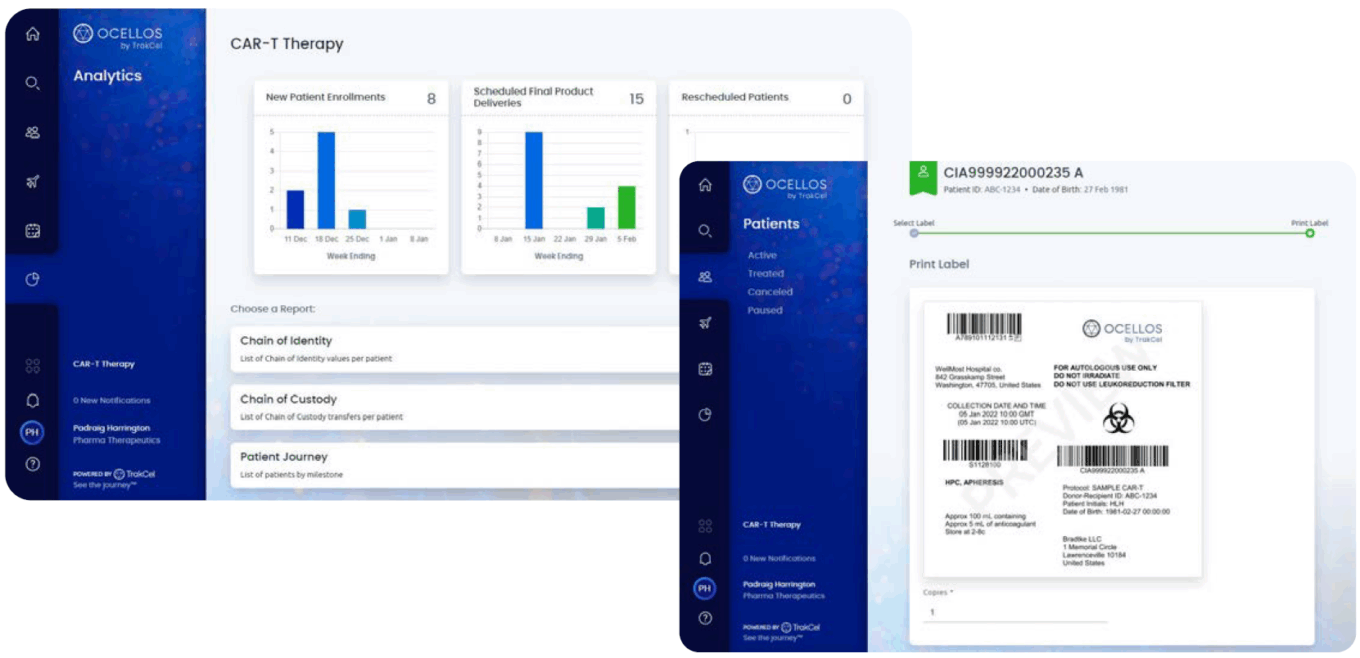
Source: TrakCel.
Advanced technology meeting the needs of advanced therapeutics
As society develops ever more effective healthcare interventions based on new scientific discoveries and advancements, the processes used to manage development and delivery must keep pace. Once mundane elements of development and delivery processes, including making sure the right materials are in the right place at the right time, are now critical and sensitive links in a highly sophisticated healthcare system. Many innovative companies are rising to the challenge, often focusing on clinical trials – the key step between development and commercialization. We see a bright future for these companies and the therapies they help enable.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
