Quarterly insights: Pharma IT
How regulation and risk are driving pharma QMS modernization

Pharmaceutical quality management systems ensure product quality, compliance and safety in manufacturing by streamlining processes and managing risks.
Multiple regulatory frameworks underpin quality management systems. These frameworks are evolving and converging around the same expectations: validated systems, risk-based governance and digital traceability. But quality management is no longer just about compliance. It has also become a strategic driver of resilience, efficiency and growth.
As a result, pharma companies continue to invest in cloud-based, AI-enabled electronic quality management systems. We expect eQMS adoption to be strategically important for pharma over the next five to 10 years.
We examine how regulatory demands, scientific complexity and digital transformation are redefining quality management and where the next wave of investment and innovation is headed. We also profile established eQMS leaders and a rising class of specialized, digital-first up-and-comers.
TABLE OF CONTENTS
- Quality management has become a key necessity
- QA and QC: The two pillars of quality management
- Key QMS objectives
- Quality management is evolving along with pharma priorities and regulation
- Regulatory frameworks
- Pharmaceutical regulation is evolving
- AI is entering the quality toolbox, carefully
- Change is driving eQMS investments
- Market landscape: Established leaders and emerging challengers
- Strong eQMS adoption ahead
- Pharma IT indexes lag far behind broader market
- Pharma IT M&A: Notable transactions include Clario and Scientist.com
- Pharma IT private placements: Notable transactions include PhaseV and Ignite Data
Quality management has become a key necessity
If pharmaceutical manufacturing were a symphony, then its quality management system (QMS) would be the conductor, ensuring every process, person and record is perfectly in tune. A QMS lays out clear processes and responsibilities so everyone involved in pharmaceutical production knows what to do and how to do it, and it generates records of these actions that help address any shortcomings. A strong QMS means fewer mistakes, smoother regulatory inspections, and medicines that consistently meet global safety and quality standards. Without it, production slows, errors multiply and regulatory actions loom.
Market dynamics are making quality management systems more critical than ever. Regulators are raising standards for data integrity, risk management and digital maturity. Pharma’s shift to biologics and cell and gene therapies adds complexity, while contract development and manufacturing organizations (CDMOs) and supply chain partners face stricter scrutiny to ensure they are meeting global standards. At the same time, cloud platforms have moved from niche to mainstream, and AI is reshaping how quality issues are predicted and prevented.
Pharmaceutical QMS and its importance
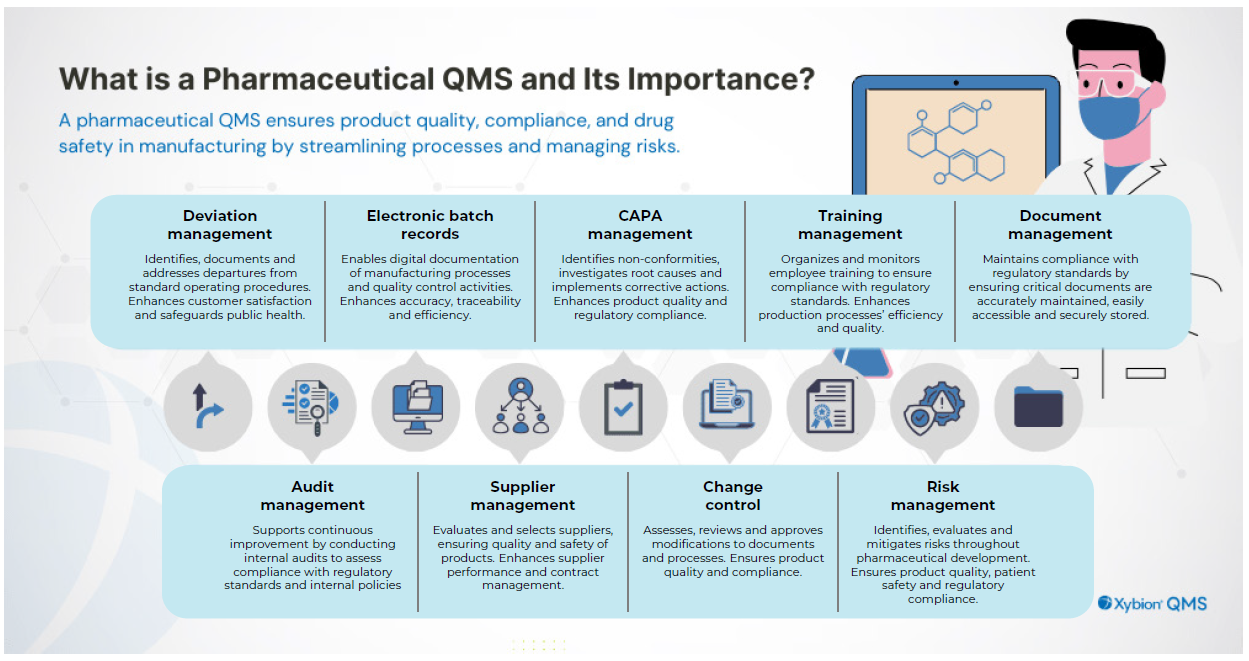
Source: Xybion.
Against this backdrop, pharmaceutical quality management itself is undergoing a fundamental shift. Quality management is no longer just about compliance. It has become a strategic driver of resilience, efficiency and growth. What was once an IT concern is now a board-level priority and performance lever. In this report, we examine how regulatory demands, scientific complexity and digital transformation are redefining quality management and where the next wave of investment and innovation is headed.
BEYOND COMPLIANCE, A QMS STRENGTHENS PERFORMANCE IN SEVERAL CRITICAL AREAS:
- Patient safety: If something goes wrong, the QMS helps you spot it fast, contain it and prevent it from happening again.
- Regulatory trust: Inspectors look for controlled processes and credible records (electronic or paper) that show you do what you say you do.
- Operational performance: Clear procedures, fewer manual handoffs, and “review-by-exception” shorten cycle times and reduce rework.
- Business resilience: When work is standardized and well documented, site expansions, tech transfers, and onboarding CDMOs become manageable.
QA and QC: The two pillars of quality management
In pharmaceutical manufacturing, quality assurance (QA) and quality control (QC) are complementary functions that ensure products are safe, effective and compliant. Both operate under strict regulatory frameworks and depend on each other to create closed-loop systems of continuous improvement.
Quality control, quality assurance and quality management in the pharmaceutical industry
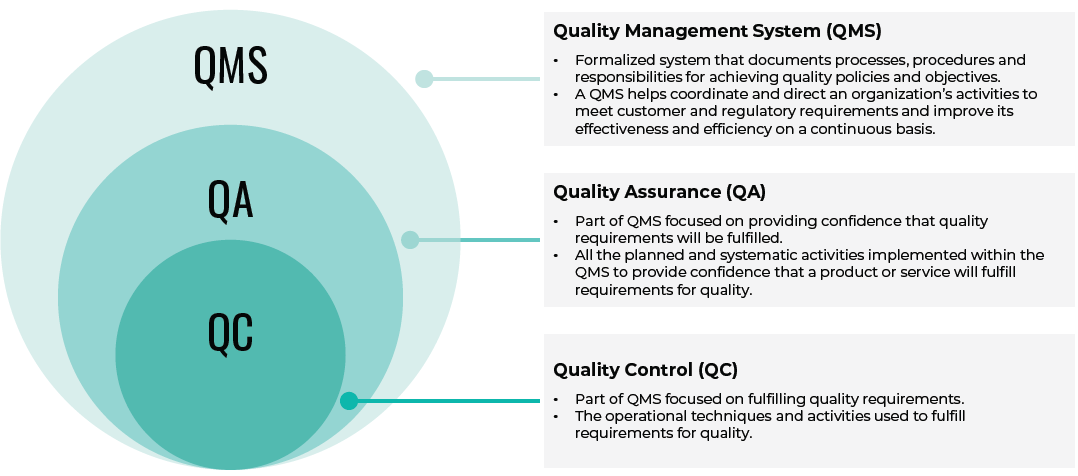
Source: Pharmatalks Community.
QA is proactive, building systems to prevent errors before they occur. This includes writing and maintaining standard operating procedures, training staff, qualifying suppliers, handling deviations and corrective and preventive actions, running audits, and managing change.
QC is reactive. It tests medicines and verifies whether materials meet standards. This includes sampling incoming active pharmaceutical ingredients, running in-process and finished-product tests, validating methods, and managing out-of-specification and out-of-trend investigations and stability programs. Put simply, QC ensures every product released to patients performs as intended.
Crucially, these two functions form a feedback loop. Findings from QC lead to systemic corrective and preventive actions that are incorporated into QA, while QA updates processes to prevent repeat errors discovered by QC. Together, they enable pharma companies to meet regulators’ expectations for a continuous cycle of prevention, detection and improvement underpinned by frameworks such as 21 CFR, EU Annex 11, and ICH Q9. This interdependence is what makes long-term compliance, product integrity and patient safety possible.
Key QMS objectives
The core components of a pharmaceutical QMS translate quality principles into day-to-day practice:
- Setting the rules (documents and training): Every QMS starts with the rulebook. Standard operating procedures and controlled forms ensure every operator is following the same method. Training records prove they were qualified. Digitization replaces binders and sign-off sheets with automated version control and dashboards, so when a process changes, the right people are instantly retrained.
- Catching problems before they escalate (deviations, corrective and preventive actions, risk management): Something eventually goes wrong in even the best-managed processes: a mislabeled vial, a failed assay. QC detects such issues, and QA leads the response (root-cause analysis and corrective and preventive action). Risk tools help companies focus resources on what matters most. For example, contamination in sterile areas gets more scrutiny than a temperature excursion in packaging. A QMS speeds this up with workflows, risk scoring and trending to spot repeat issues across sites.
- Proving it works (validation, lab controls, data integrity): Regulators don’t take “trust us” for an answer. Every piece of equipment, process and lab method must be validated and shown to perform consistently. QC labs confirm consistency with release and stability testing, while QA ensures life cycle validation and data integrity. With electronic systems, every action leaves an audit trail, making inspections smoother and reducing the risk of warning letters.
- Making medicines consistently (batch records, supplier and environmental controls): In paper form, missing signatures or transposed numbers delay release. With electronic batch records, operators are guided step-by-step, errors are flagged in real time, and QA can “review by exception.” Quality management systems also encompass qualifying suppliers, testing raw materials, and monitoring cleanrooms to ensure nothing jeopardizes final products.
- Learning from the field (complaints, audits, recalls, management review): Quality management doesn’t stop when drugs leave the factory. Complaints, adverse events, and audits feed back into the system. If a recall is needed, QMS ensures companies can trace exactly which lots are affected and act fast. Management reviews turn this into a board-level conversation, using metrics like “right-first-time” or corrective and preventive action effectiveness to steer investment.
- Adapting and improving (change management and continuous improvement): Pharma is never static. Change control processes ensure every change is evaluated for risk, approved by the right experts, and reflected in updated documents and training. Digitization makes this transparent, linking each change to affected procedures and records. Done well, this isn’t bureaucracy; it’s how companies innovate without falling out of compliance.
Quality management is evolving along with pharma priorities and regulation
Historically, pharmaceutical quality management relied heavily on binders and paper-based documentation. Batch records, deviation reports and standard operating procedures were stored on paper, making compliance a matter of maintaining endless paper trails and preparing binders for inspection. This approach was labor intensive and error prone. Problems were often discovered only after they had already occurred.
Quality oversight is shifting to become more risk-based, data-driven and digitally auditable. Instead of just demonstrating compliance through documents, companies are now expected to show real-time control of their processes through connected, inspection-ready systems. That means:
- Cloud-native electronic quality management systems serve as the inspection-ready backbone that replaces paper trails with centralized, compliant and easily auditable digital systems.
- Integrated analytics and artificial intelligence provide predictive deviation detection, metric-based quality trend analysis, and supplier scorecards to identify risks earlier and manage them more effectively.
- Systems connect with suppliers and CDMOs via portals and data sharing to extend oversight across organizational boundaries, ensuring supply chain resilience and compliance alignment.
Regulatory frameworks
Pharma quality is governed by a patchwork of global and regional frameworks, many of which are modernizing in parallel and collectively raising the bar on validation, risk management, and digital oversight. Their overlap is a key driver of QMS modernization. Table 1 summarizes the principal regimes that are the foundation for compliance in pharmaceutical quality management globally:
Pharmaceutical regulation is evolving
Pharmaceutical quality management is being further reshaped by a series of global regulatory updates.
The revision of the global ICH Q9(R1) regime in the 2023 and 2024 time frame sharpened expectations for quality risk management by clarifying how to choose the level of structure and detail used in quality risk management, reducing subjectivity in risk assessments and explicitly recognizing product-availability risks from quality failures. In practice, mature eQMS platforms now embed these principles directly into changes, deviation management, supplier oversight, and validation, helping companies allocate resources where patient and product risks are highest.
Following pilots in 2024, the FDA extended its Quality Management Maturity (QMM) prototype program in 2025. QMM assessments evaluate sites across practice areas such as management commitment, business continuity and advanced pharmaceutical quality systems. The intent is to incentivize companies to go beyond baseline good manufacturing practices. In practice, QMM pushes firms to equip their QMS with reliable metrics, trend analysis capabilities, and governance tools for continuous improvement.
TABLE 1: Principal regimes for compliance in pharmaceutical quality management globally
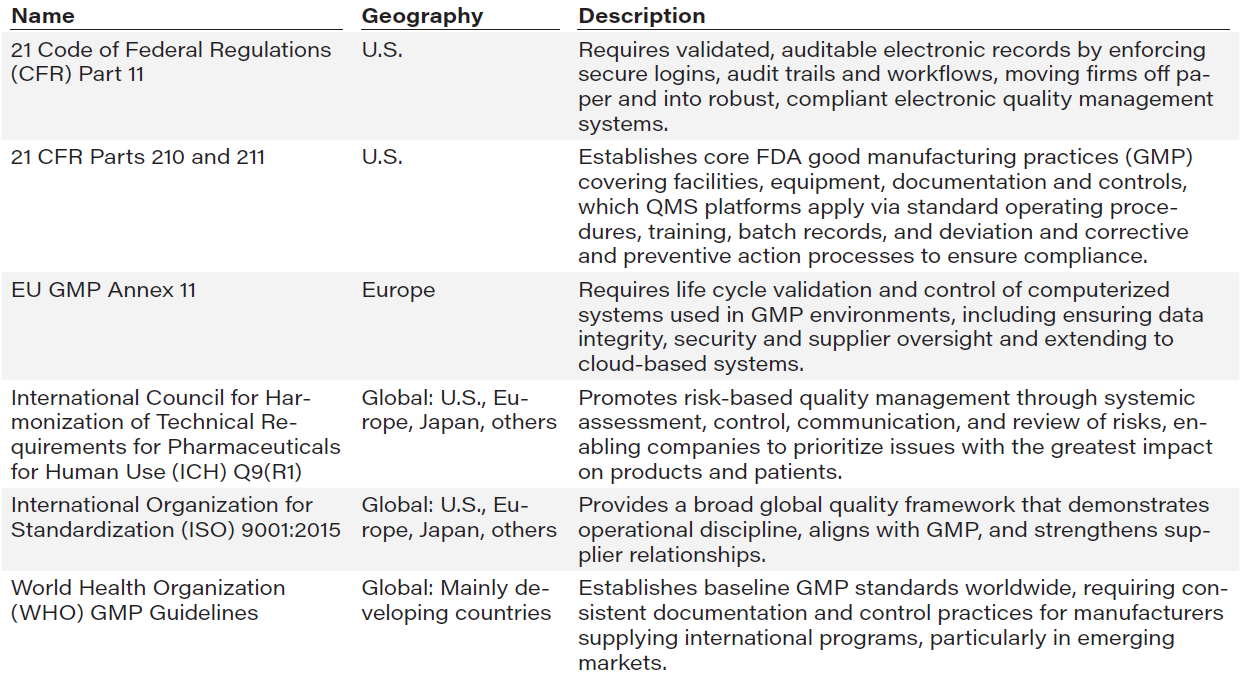
Source: First Analysis.
Together, these updates formalize a differentiated approach to quality: risk-based scaling of rigor under Q9(R1) and maturity-based differentiation under QMM. The result is a landscape where regulators and sponsors distinguish between minimally compliant and best-in-class operations. To demonstrate maturity and resilience, companies are expected to invest in analytics, dashboards, and connected QMS tools that provide real-time visibility and auditable evidence of control.
Europe is modernizing its good manufacturing practice framework for the digital era. Drafts released in 2025 tighten life cycle expectations for computerized systems (Annex 11), introduce the first-ever AI-specific annex covering AI model design, data quality, and human oversight (Annex 22), and refresh documentation rules to reflect digital operations (Chapter 4). For QMS leaders, that means heightened expectations around supplier and cloud oversight, risk-based validation, and governance of AI tools used in manufacturing and quality. Public consultation runs through late 2025, with final texts expected in 2026.
In the United States, FDA’s draft Computer Software Assurance (CSA) framework has become the reference point for validating production and quality software. Rather than exhaustive test scripts, CSA emphasizes critical thinking and intended use. Combined with Annex 11’s life cycle approach, this provides a coherent blueprint for validating electronic quality management systems, laboratory information management systems, manufacturing execution integrations, and automation without wasting resources testing the obvious.
In August 2025, a White House executive order directed the Department of Health and Human Services’ Administration for Strategic Preparedness and Response to create a strategic active pharmaceutical ingredient reserve, identifying roughly two dozen critical drugs, and build repository capacity to strengthen U.S. pharmaceutical manufacturing. Increasing domestic production means more facilities under FDA oversight, which we expect to drive greater eQMS adoption to ensure inspection readiness. For CDMOs and emerging biopharma, this expansion is a structural driver of eQMS adoption, as inspection-ready digital systems become a prerequisite for winning contracts and maintaining licenses.
AI is entering the quality toolbox, carefully
The FDA has released guidance documents signaling formal expectations for AI model life cycle control, data integrity, and supplier oversight. But even as guidance is finalized, AI is already embedded across quality workflows. Teams are using natural language processing and generative AI to accelerate redlines of standard operating procedures and change impact analysis, classify deviations, suggest likely root causes of problems, and pre‑draft corrective and preventive actions. Computer vision systems are being used to verify drug label presence, legibility, lot numbers, and serial numbers, and to prevent mix‑ups. Multivariate and machine learning models are being built to flag emerging drifts in continuous or hybrid manufacturing, enabling earlier interventions and smaller scrap events.
AI shifts quality from “find-and-fix” to “predict-and-prevent” by triaging deviations, prioritizing corrective and preventive actions by risk, and spotting cross‑site patterns that can predict problems and trigger actions to head off recalls. But AI is less likely to replace validated workflows than to augment them under strict controls, and there are still regulatory limits. In the eyes of regulators, AI-driven models must be explainable and traceable. Training data must be representative and controlled for bias. Humans must be involved in critical decisions. AI requires full life cycle management for versioning, drift monitoring, re-training under change control, and validation with auditable records.
Change is driving eQMS investments
Taken together, these regulatory frameworks and their evolution show how fragmented oversight is converging around the same expectations: validated systems, risk-based governance and digital traceability. As a result, pharma companies continue to invest in cloud-based, AI-enabled eQMS platforms. We believe eQMS is increasingly a baseline requirement for modern compliant pharma operations, and we expect eQMS adoption to be a strategically important area for pharma over the next five to ten years.
Market landscape: Established leaders and emerging challengers
The pharmaceutical eQMS market is characterized by a handful of dominant players with broad, integrated platforms, and a rising wave of specialized, digital-first up-and-comers. The leaders command scale and trust with large biopharma, while the smaller innovators bring speed, focus and cloud-native design to underserved segments such as mid-market pharma companies, CDMOs, and biotech companies.
At the enterprise tier, five large-scale platforms anchor pharma quality:
Veeva Systems’ (VEEV) Vault eQMS manages deviations, corrective and preventive actions, change control, complaints, audits and training. Vault’s recent addition of Veeva AI and Direct Data API enable deeper data access and AI capabilities. Veeva’s end-to-end footprint is reinforced by its marquee contact relationship management system, its new, long-term partnership with IQVIA (IQV), and its partnerships with service providers. Together, they tighten integration with quality control and laboratory information management systems. In our view, this strongly positions Veeva to benefit from pharma companies’ pursuit of risk-based, digital-first oversight and better connections to post-market quality information.
Seamless customer experience unifies scientific exchange, content and inquiry management
Source: Veeva.
MasterControl provides cloud-based eQMS and digital manufacturing software for regulated life sciences companies. Its quality suite spans document control, training, deviations and corrective and preventive actions, change management, complaints, and connected shop-floor workflows. Recent enhancements to its offering add regulated-sector readiness, formal AI governance, asset calibration and maintenance, and practical AI assistance.
Closed loop eQMS
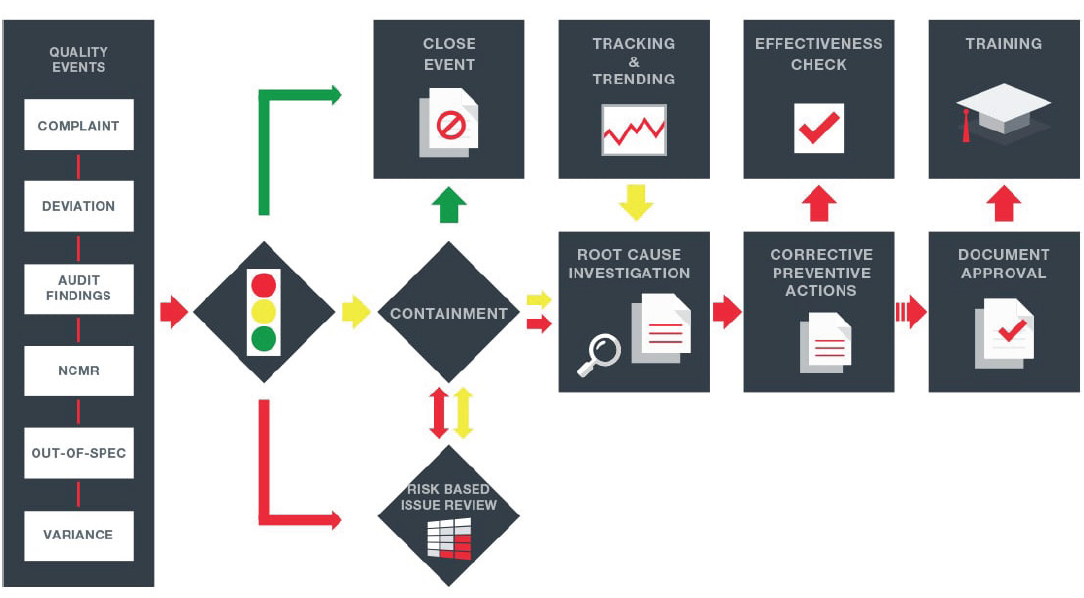
Source: MasterControl.
Notes: NCMR means non-conforming material report.
Honeywell (HON) acquired Sparta Systems and its TrackWise offering in 2021. Sparta Systems’ long-established, on-premise TrackWise QMS and the cloud-based TrackWise Digital QMS form the core of the offering and are complemented by the broader TrackWise life sciences platform, including recent additions such as TrackWise Manufacturing (for AI-assisted, cloud-native manufacturing optimization) and TrackWise Recall Management (for end-to-end recall orchestration). Given Honeywell’s strength in industrial automation, we believe TrackWise remains well positioned to connect plant data with quality management. This positioning aligns well with today’s focus on drug life cycle quality management and the shift from binder-driven compliance to integrated, cross-plant dashboards.
ETQ, part of Hexagon’s Manufacturing Intelligence division, offers the cloud-native ETQ Reliance QMS, which includes more than 40 applications. Recent enhancements include AI-driven anomaly detection, the ability to generate recommendations for core workflows via its Predictive Quality Analytics solution, and updates to its Reliance NXG data access solution along with a Snowflake (SNOW) powered data layer that strengthen analytics and data lineage. (Data lineage is the process of tracking the flow of data over time, providing a clear understanding of where data originated, how it has changed and its ultimate destination within data pipelines.) ETQ’s annual Pulse of Quality survey underscores macro drivers – labor constraints and rising recall exposure – that favor proactive, data-driven quality management programs.
ComplianceQuest’s eQMS is a modern, cloud-based platform powered by Salesforce that connects every stage of the product life cycle – from quality and safety to supplier oversight – within a unified digital environment. It streamlines essential quality processes such as corrective and preventive actions, audits, deviation handling, document control, and training management while delivering real-time visibility and actionable analytics to foster continuous improvement. Designed specifically for highly regulated sectors like pharmaceuticals and biotechnology, the system includes built-in audit trails, secure electronic signatures, and scalable integrations to ensure full compliance and operational agility.
ComplainceQuest’s enhanced next best action
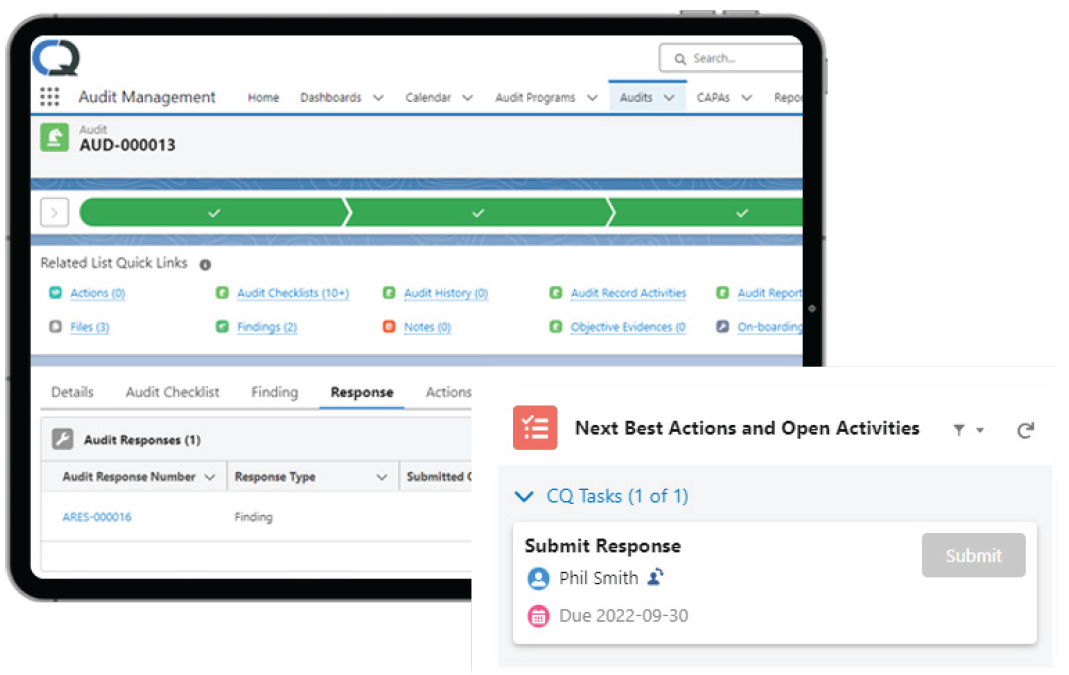
Source: ComplianceQuest.
Following are some of the more specialized, digital-first up-and-comers.
Founded in 2015 and headquartered in Phoenix, Arizona, Dot Compliance delivers a Salesforce-native, AI-powered eQMS tailored for pharmaceutical, biotech and medical device companies. With modular offerings like eQMS Xpress, Compliance Xpand and Enterprise Xact, the company enables rapid adoption of core quality workflows alongside advanced AI insights through its Dottie AI assistant, which was recently upgraded with generative and predictive capabilities. In April 2024, Dot Compliance announced a Series B extension to scale its AI roadmap amid growing customer momentum (over 500 customers across 30 countries), reinforcing its alignment with regulatory shifts that demand digital, validated and inspection-ready quality systems.
Dot Compliance dashboard
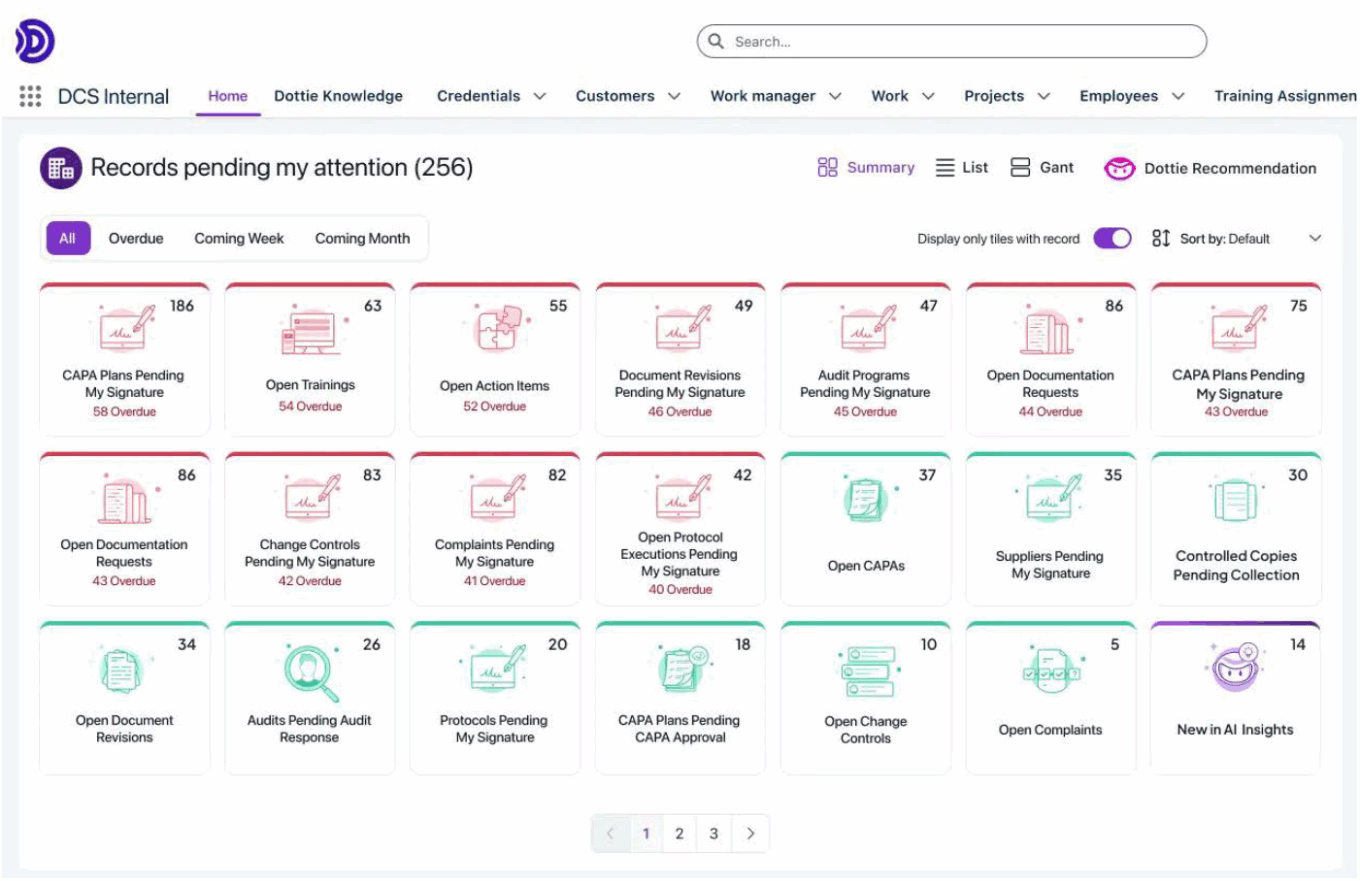
Source: Dot Compliance.
Founded in 2016 and headquartered in Austin, Texas, QbDVision provides a Digital CMC (chemistry, manufacturing, and controls) platform that centralizes product and process knowledge, integrates risk management, and supports control-strategy development and tech transfer across product life cycles. We think QbDVision is interesting because it sits at the CMC-to-quality handoff. By structuring development knowledge and risks up front, it shortens tech transfer processes, improves data lineage for quality management and validation downstream, and addresses regulators’ push for risk-based, digitally auditable control strategies.
QbDVision offers software tools to manage everything from a single login: process evolution, risk evaluation and data analytics
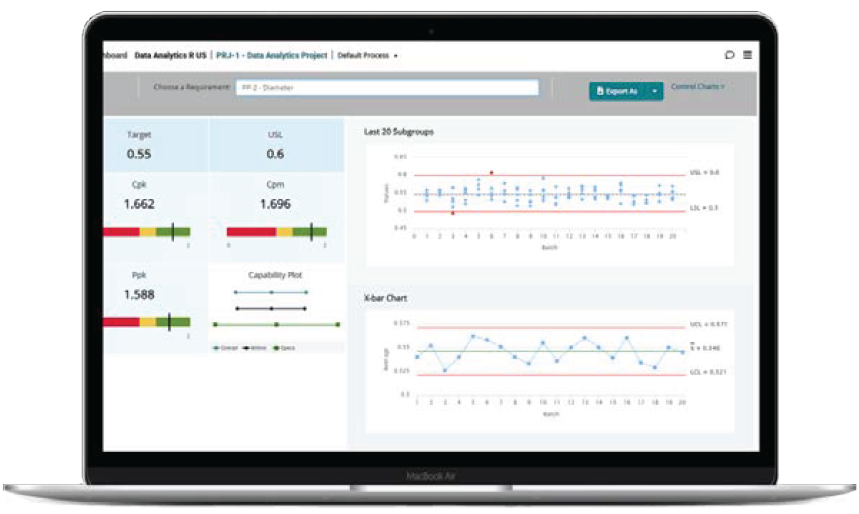
Source: QbDVision.
Founded in 2012 and headquartered in San Francisco, Qualio offers a cloud-based QMS designed to streamline quality and compliance processes for life science companies. Its centralized platform automates tasks such as document control, training, corrective and preventive actions, audits, and supplier management, ensuring adherence to FDA, ISO and good practice standards. The system features AI-powered gap analysis, pre-built templates and digital workflows to enhance efficiency and maintain constant audit readiness. In June 2025, Qualio unveiled an upgraded design control module, offering improved traceability, flexibility and user experience. This enhancement includes a new Traceability tab that visualizes relationships across requirements, tests and risks, as well as an AI-powered gap analysis tool to identify compliance gaps. In October 2025, Qualio announced the general availability of Compliance Intelligence, an AI-driven tool that streamlines audit preparation and ensure continuous compliance.
Qualio’s medical device QMS demo
Source: Qualio.
Founded in 2010 and headquartered in Ardmore, Pennsylvania, ZenQMS offers a cloud eQMS for good-practice-regulated organizations covering document control, training, corrective and preventive actions, audits, and supplier oversight. Over the last two years ZenQMS has also added validation capabilities. In our view, this has resulted in a mid-market, focused platform that maps cleanly to regulatory expectations and positions ZenQMS as an agile alternative to the enterprise-focused providers.
ZenQMS’ flexible, all-in-one eQMS for life sciences and good-practice-regulated companies
Source: ZenQMS.
Strong eQMS adoption ahead
In today’s increasingly complex and regulated pharmaceutical landscape, the importance of a robust, modern electronic quality management system cannot be overstated. eQMS platforms have become indispensable for ensuring compliance, driving operational excellence and enabling continuous improvement across product life cycles. With accelerating regulatory scrutiny, expanding global operations and rapid advances in cloud and AI technologies, we believe eQMS adoption will remain strong over the next several years.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
