Quarterly insights: Pharma IT
Line of “site” to better drug development: Site-centric clinical trial tech
Pharma sponsors are realizing they must address clinical trial site inefficiencies that have been a byproduct of (or have been neglected by) the otherwise largely successful efforts to make drug development more efficient with clinical trial technology.
Historically, trial sponsors have mandated trial sites use each sponsors’ preferred clinical trial technology stack. As a result, sites’ needs and preferences have been overlooked, and sites have been overwhelmed with multiple disconnected solutions.
To resolve this, sponsors are increasingly focusing on adopting site-centric technology for streamlining workflows, reducing administrative burden and compliance errors, and enhancing sponsor-site relationships. We highlight the key areas where we see site-centric technology making an impact – study start-up, source data collection, and imaging – and some of the innovative companies providing these solutions.
TABLE OF CONTENTS
Includes discussion of eight private companies
- Clinical trials remain inefficient, and trial sites bear the brunt of the pain
- Site-centric solutions address the problem – “We’re in this together”
- Study start-up: Faster path to trial execution
- eSource: More efficient, accurate data collection
- Imaging: Better protocol adherence, faster turnarounds
- Line of “site” to a better future
- First Analysis eClinical and commercialization indices at one-year low
- Pharma IT M&A pace slows
- Pharma IT private placement pace slows
Clinical trials remain inefficient, and trial sites bear the brunt of the pain
Despite a remarkable surge in adoption of eClinical solutions since the onset of the pandemic, particularly decentralized or hybrid solutions to conduct clinical trials remotely, trials remain inefficient as trial costs and protocol complexity increase and timelines tighten. In many ways, the long journey to substantially increase drug development efficiency with technology has just begun. Trials sites bear the brunt of the pain: Factors ranging from staffing shortages to low patient participation rates mean sites must do more with less. At the same time, sites are burdened with more administrative tasks now than ever and still spend an inordinate amount of time on manual processes and administrative work. Solutions aimed at increasing trial site efficiency are key to alleviating the pain.
“Solutions will not only have to be made with technology and personnel usage but also with innovative structural change that will positively affect the sites while driving costs significantly down for sponsors.”
-Dan Perlman, CEO of TPS
Historically, though, trial sponsors have mandated trial sites use each sponsors’ preferred eCinical tech stack. As a result, sites’ needs and preferences have been overlooked, and sites have been overwhelmed with multiple disconnected solutions. According to life science consultant Randall Hein, CEO of Heinsight, “The exciting advancements over the past several years in the tools and technology available for conducting clinical trials have greatly benefited patients, sponsors and sites. However, the proliferation of new technology combined with the lack of integration across various platforms has also created an additional burden for sites, resulting in increased training time, the requirement to use multiple systems for data entry, and potential changes to established workflow and procedures.”
According to a May Sites NOW survey from the Society for Clinical Research Sites (SCRS), 60% of sites polled are using at least 20 systems daily. Furthermore, the 2021 SCRS annual survey shows these systems require about 17 hours in training per study, per site, per month. Ironically, solutions designed to streamline and automate clinical trials have created new bottlenecks for site staff.
“The platform eliminates guesswork for site staff, reduces the potential for protocol deviations, enhances participant diversity, and boosts coordination between sponsors, trial participants and sites by tightly orchestrating the trial tasks within the protocol to the site’s workflow in one platform.”
-Chris Venezia, CEO of ProofPilot
In our view, this is unsustainable. Clinical research personnel are burning out, leading to high turnover. In April 2022, SCRS sent an open letter to sponsors and clinical research organizations (CROs) stating staff turnover rates at site networks have doubled on average relative to prior to the pandemic, from a range of 10% to 37% to a range of 35% to 61%. The letter also stated sites are struggling to retain trained staff members due to sponsors and CROs offering them higher-paying jobs.
Site-centric solutions address the problem – “We’re in this together”
Based on our conversations with industry personnel at the recent Dpharm conference, sponsors are now taking note and seriously listening to the needs of sites, especially when it comes to technology. Sponsors’ “my way or the highway” approach appears to be giving way to a “we’re in this together” approach, with sponsors increasingly focused on site-centric solutions for streamlining workflows, reducing administrative burden and compliance errors, and enhancing sponsor-site relationships.
Dan Perlman, CEO of First Analysis venture capital eClinical portfolio company Transformative Pharmaceutical Solutions (TPS), explained, “Solutions will not only have to be made with technology and personnel usage but also with innovative structural change that will positively affect the sites while driving costs significantly down for sponsors.” Below we highlight the key areas where we see site-centric solutions making an impact: study start-up, source data collection (or eSource) and imaging.
Study start-up: Faster path to trial execution
It can take up to a year to identify a site and activate it to conduct research. Investments to reduce this timeline can have a high return, as every day a new drug is delayed from reaching market can represent more than $1 million of lost revenue, depending on disease and indication. As a result, study start-up is a key focus for trial sponsors employing a site-centric approach to technology.
Study start-up encompasses several activities performed by sponsors, CROs and trial sites, all of whom need to communicate and share data. These activities include site selection and feasibility assessment, regulatory document submission, contract and budget negotiations, institutional review board and ethics committee approval and planning, and patient consent and enrollment. Today’s increasingly complex study protocols and increased trial decentralization require even greater collaborative efforts on the part of sponsors, CROs and sites to adhere to established timelines and budgets and to ensure proper trial conduct. Any delay in start-up means some combination of higher costs and shortened time available for patient recruitment and the study itself.
One notable company focused on trying to alleviate tech burden at sites and promote collaboration is VersaTrial. VersaTrial’s web-based platform enables site staff to organize and share trial-specific technology links and contacts, automatically complete feasibility questionnaires and provide feedback to sponsors regarding sites’ tech experience. The platform also provides sponsors with direct visibility into the onboarding status of each tech solution used at sites, real-time alerts designed to improve site selection, and site monitoring to detect operational issues before they become problems.
Another company aiming to bridge the gap between sites and sponsors is ProofPilot. ProofPilot provides trial sponsors the ability to digitize text-based trial protocols into task-based digital workflows for trial sites. Additionally, it enables sites to send electronic informed consents (eConsents) to patients remotely to review and sign. This is an important feature given the traditional on-site informed consent process can be a tremendous burden for patients, caregivers and sites, especially for complex trials that may require multiple consents and re-consents.
ProofPilot digital protocol automation platform
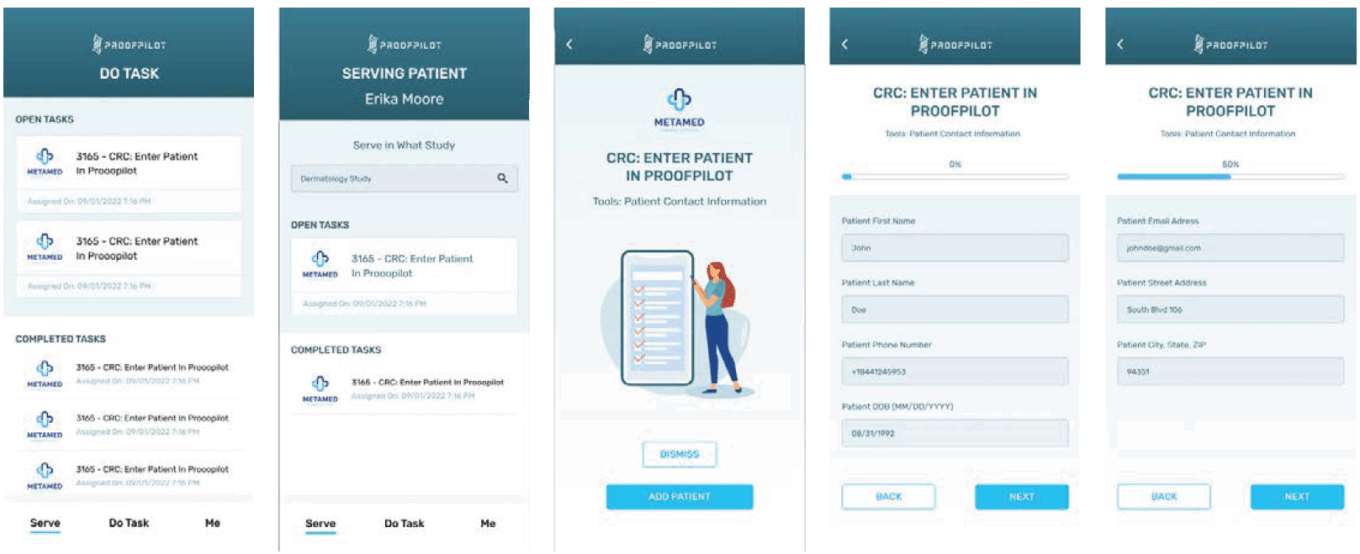
Source: ProofPilot.
With ProofPilot’s platform, sponsors and CROs can track participant enrollment status in real time while ensuring the most up-to-date versions of informed consents are reviewed and signed by trial subjects. According to ProofPilot’s CEO Chris Venezia, “The platform eliminates guesswork for site staff, reduces the potential for protocol deviations, enhances participant diversity, and boosts coordination between sponsors, trial participants and sites by tightly orchestrating the trial tasks within the protocol to the site’s workflow in one platform.”
Earlier this month, ProofPilot announced a strategic partnership with Citeline, which offers a patient recruitment and engagement platform called Connect. Citeline will deploy ProofPilot’s Co-Pilot platform to enable patients to instantly schedule calls and on-site visits and communicate directly with site staff. The collaboration also enables patients to share electronic medical records with site staff for screening and enables sites to deliver eConsents to trial participants. We note Citeline announced an agreement to merge with Norstella in June.
ClinOne takes a similar approach. ClinOne’s unified platform features modules for trial awareness, eConsent, patient rides, self-dosing compliance and electronic diaries for quality-of-life questionnaires. Recently hired CEO Andrea Valente commented, “ClinOne’s platform allows patients and families to directly connect with their research site and take better control of their trial experience.” In June, the company announced a partnership with uMotif to provide ClinOne customers direct and seamless access to uMotif’s platform for electronic clinical outcome assessments (eCOAs) and electronic patient reported outcomes (ePROs) to further enhance participant experiences and improve endpoint data quality.
“ClinOne’s platform allows patients and families to directly connect with their research site and take better control of their trial experience.”
-Andrea Valente, CEO of ClinOne
ClinOne eConsent module
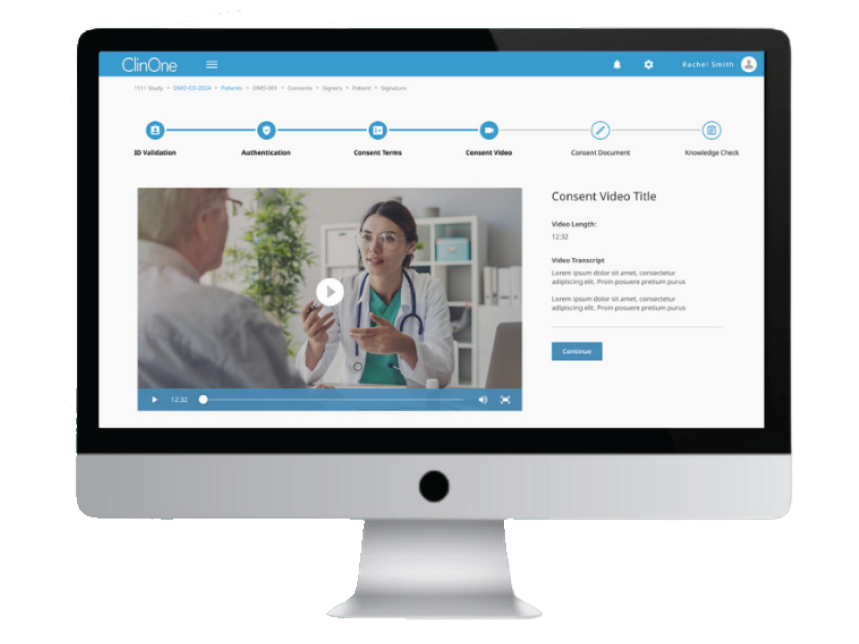
Source: ClinOne.
The study start-up phase is also seeing a new generation of solutions to help sites manage and store regulatory documents during start-up. These documents vary by type of trial and regulatory requirements but typically include study protocol, institutional review board approvals, participant informed consents, copies of study investigator curricula vitae and medical licenses, financial disclosures, Form FDA 1572 (“Statement of Investigator” required by the FDA in drug studies) and investigator agreements (required by the FDA in device studies), and site logs. Companies including Florence Healthcare and Advarra are going beyond traditional eReg binder point solutions to offer sites and sponsors a platform focused on collaboration and transparency.
Florence’s site enablement platform
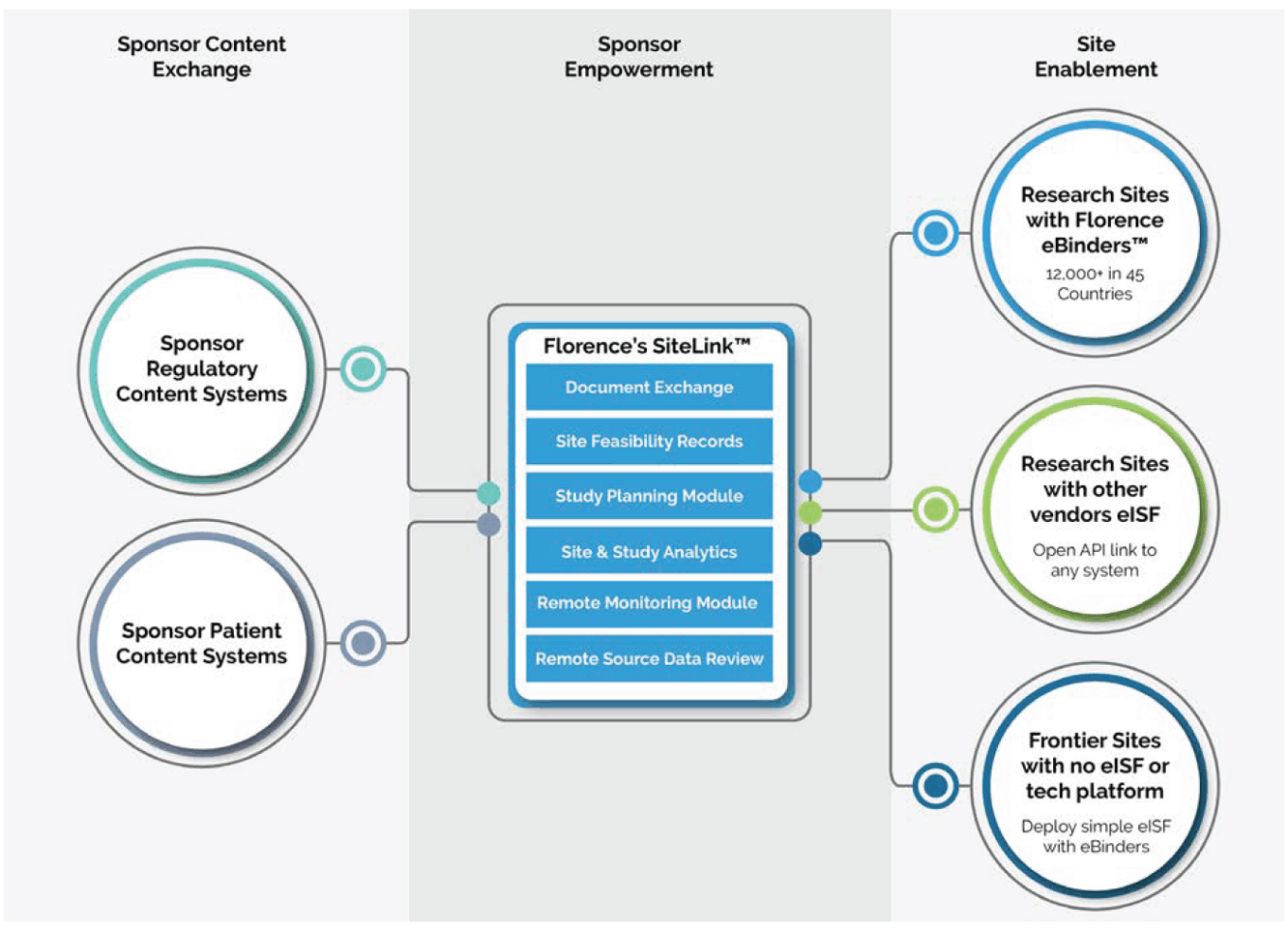
Source: Florence Healthcare.
Earlier this month at the Dpharm conference, Florence Healthcare announced that SiteLink, the company’s unified document workflow platform, can integrate with any site-based electronic investigator site file (eISF) or eRegulatory system. SiteLink also integrates sites’ eISFs directly with sponsors’ electronic trial master files (eTMF) for quality-controlled document exchange, whether sponsors use Florence’s eTMF solution or another vendor’s. According to the company, the platform facilitates over 5.5 million research workflows monthly across activities including remote start-up, site management, monitoring and source data review. “SiteLink is a new approach to solving an old problem – linking sites and sponsors. The key difference is our focus on site centricity and open integrations, two of the barriers that held back other solutions,” commented Florence’s CEO Ryan Jones.
“SiteLink is a new approach to solving an old problem – linking sites and sponsors. The key difference is our focus on site centricity and open integrations, two of the barriers that held back other solutions.”
-Ryan Jones, CEO of Florence Healthcare
In July, Advarra launched cloud-based end-to-end secure document exchange aimed at accelerating study start-up. All regulatory documents originating with sponsors or at sites can be managed, signed and securely exchanged to correct parties for long-term storage in sponsors’ eTMFs and sites’ eISFs. These new capabilities connect eReg with Advarra’s Longboat Platform.
Advarra’s secure document exchange
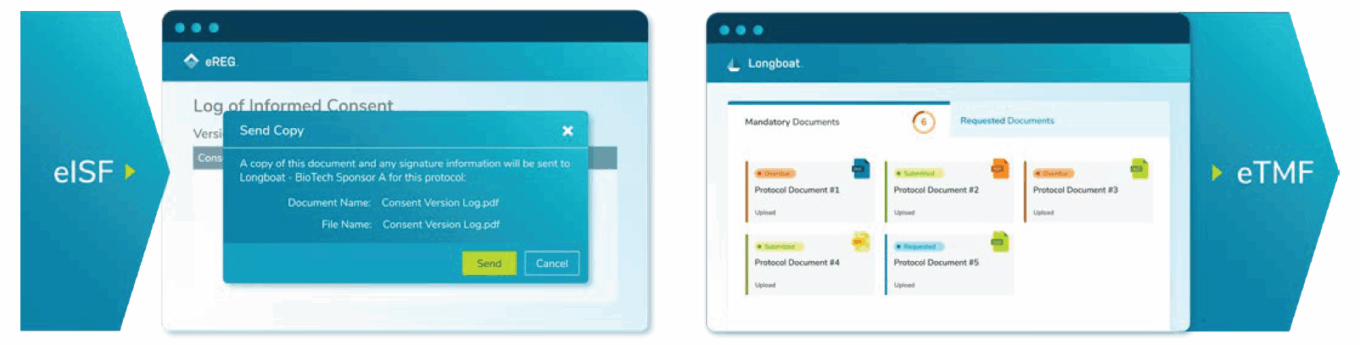
Source: Advarra.
eSource: More efficient, accurate data collection
eSource is a solution that enables trial sites to collect data digitally at the source without having to record it on a piece of paper first and then transcribe it to an electronic data capture (EDC) or eCOA system. With eSource, information flows directly into studies’ databases in real time or near real time, enabling earlier visibility and a faster start to data analysis relative to traditional data collection methods. The information collected includes clinical findings, observations and evaluations conducted on participants. eSource reduces site workloads by eliminating data entry and the need for quality control and quality assurance for data entry.
Before the advent of eSource, the pharmaceutical industry mainly relied on on-site monitoring, including frequent source data review (SDR) and source data verification (SDV), to help ensure sites maintained processes, procedures and records in accordance with protocols, standard operating procedures, good clinical practice and applicable regulations. Clinical research associates (CRAs) have traditionally conducted site visits every four to eight weeks to achieve 100% SDV. SDV can be particularly laborious because it involves validating data presented in case report forms against original source data such as laboratory notes, pharmacy dispensing records and consent forms. Additionally, it can be costly. The industry spends an estimated $8 billion each year on SDV monitoring, accounting for 15% to 30% of trial costs. According to Clinical Research.IO’s 2022 Clinical Research Associate Survey, CRAs spend about 60% of their time on SDR and SDV.
This traditional SDR and SDV approach leaves substantial room for problems to arise. Sites commonly miss critical study data points during patient enrollment, and the potential for something to go wrong is compounded by the fact sites often enter data into EDC systems several days after patient visits occur. This delay, plus the time intervals between CRA visits that can uncover such errors, means it can be a long time before research sites become aware of problems, which may affect all a site’s participants in the interim. eSource technology eliminates much of this risk by obviating the need for SDR and SDV. It also reduces travel costs for CRAs and frees up their time to focus on other trial tasks. Likewise, it reduces the amount of time site-dedicated staff spend on data entry.
Currently, most eSource solutions are built on mobile devices such as tablets designed to look like paper but enable site staff to record clinical trial data electronically while they are seeing and caring for patients. One notable vendor, CRIO, provides direct connectivity between sites and sponsors. Sites use CRIO’s eSource solution to transmit clinical trial data automatically to CRIO’s Reviewer module or to legacy EDC systems so sponsors can review, query, code, lock and extract the data. CRIO’s eSource solution eliminates data transcription errors by sites and the need for SDV, and it enables real-time access to site data, which reduces travel costs for CRAs and alleviates the laborious and time consuming task of data entry by site staff.
CRIO eSource solution
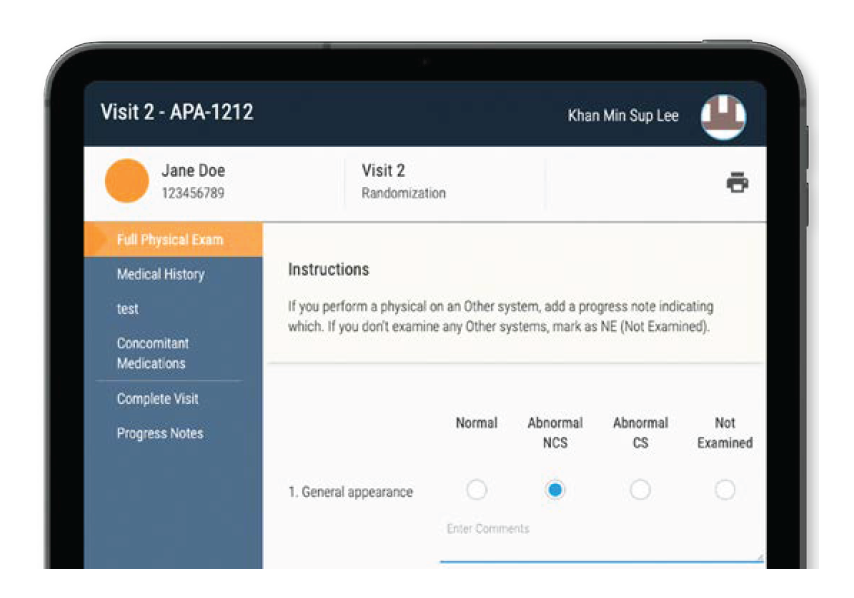
Source: Clinical Research.IO
Imaging: Better protocol adherence, faster turnarounds
The use of medical imaging in clinical trials has increased dramatically over the past decade, fueled by advances in science, technology and software. Clinical trials use a variety of imaging modalities, mainly X-rays, ultrasounds, computed tomography scans, magnetic resonance imaging and positron emission tomography scans. Trials use medical imaging for patient eligibility screening and enrollment, disease progression tracking and therapy monitoring. According to Medpace and Medidata, about 50% of all clinical trials use medical imaging as an endpoint or for eligibility criteria.
In oncology, about 90% of trials rely on medical imaging as a trial component. Oncology trials use quantitative imaging as a key tool for assessing patient eligibility, response to cancer therapies and overall treatment efficacy. For example, patients with cancer may have lesions that require serial imaging for objective assessments defined by specific response criteria and based on tumor metrics. The discovery of targeted drugs and immunotherapies as well as advances in imaging technology have led to a multitude of complex and varied tumor metrics criteria. Using the correct metrics is particularly critical in cancer trials, where these assessments can mean the difference between inclusion or exclusion in a trial.
In addition to using correct metrics, the timing of images and their assessment is particularly critical in oncology trials. Studies often require tumor metrics at the time of patient consults to determine patient eligibility to stay in trials and continue investigational therapies. This can be especially challenging when it entails rapid turnaround of complex assessments using varied response criteria. Further, workflows for completing standardized imaging assessments for trials vary by site, and site staff often struggle to balance clinical trial demands with their daily clinical practice responsibilities. Sites frequently have few resources to address the continuously evolving needs of clinical trials. Making matters worse, the FDA has been urging trial sponsors to incorporate more rigorous, controlled imaging methods and objectives into their studies.
Typically, trial sponsors have viewed and engaged sites as siloed organizations with respect to imaging, relying on sites with somewhat varied expertise to translate protocols’ imaging requirements into measurements without consistency or standardization across stakeholders and sites. Further, conventional picture archiving and communication systems (PACS), the current gold standard for clinical care, lack metrics flexibility and cross-departmental workflows needed for efficient and effective clinical trials and also lack trial-specific security features and administrative functions needed for site staff reimbursement.
First Analysis venture capital portfolio company Yunu has a novel approach to addressing these challenges in a site-centric manner. Yunu’s integrated, cloud-hosted clinical trial imaging assessment and workflow management platform, recently spun out from the Tumor Imaging Metrics Core (TIMC) at the Dana-Farber/Harvard Cancer Center, was designed and developed to improve clinical trial protocol adherence by eliminating common discrepancies that affect patient and trial decisions at sites. It also accelerates tumor measurement turnarounds so radiologists can provide same-day results. Other key features include support for high-level trial analytics, fiscal tracking, trial auditing capabilities, and a standardized training application designed to train site staff on the different tumor response criteria and how to apply these criteria during trials with the goal of reducing variability and increasing compliance across clinical trial sites.
“Clinical sites cannot scale or standardize qualityacross sites the way their pharma sponsors need them to without better research-specific technology solutions that provide the process, collaboration and best practices required.”
-Jeff Sorenson, CEO of Yunu
Yunu’s platform
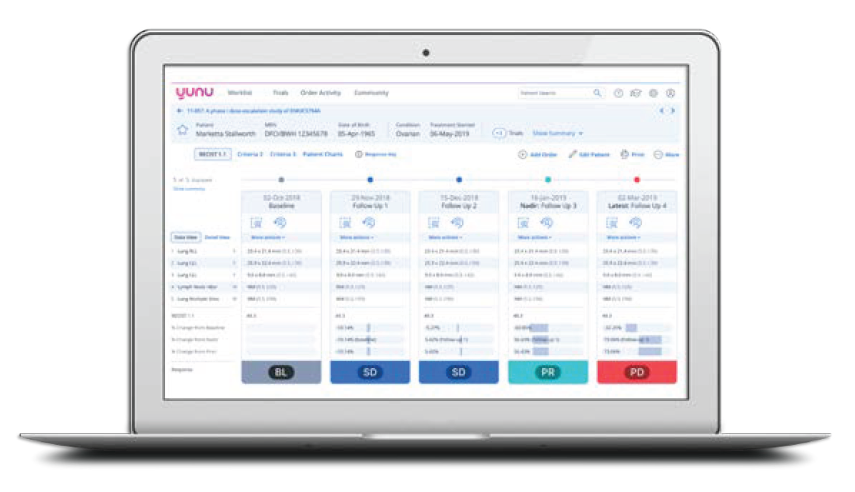
Source: Yunu.
“One of the most effective ways for pharma sponsors to increase patient enrollment in clinical trials is to expand trials to a larger number of clinical sites. Unfortunately, today clinical sites each make different interpretations about what needs to be done and apply different tools and manual processes, causing inconsistency, errors and frustration,” said Jeff Sorenson, CEO of Yunu. “Clinical sites cannot scale or standardize quality across sites the way their pharma sponsors need them to without better research-specific technology solutions that provide the process, collaboration and best practices required.”
Line of “site” to a better future
Pharma sponsors are realizing they must address the trial site inefficiencies that have been a byproduct of (or that have been neglected by) the otherwise largely successful efforts to make drug development more efficient with technology, a process that has accelerated with the onset of the pandemic and the increased adoption of decentralized and hybrid trial technologies fueled by the pandemic. The emerging shift in clinical trial automation technology to better encompass and address the needs of clinical trial sites eliminates key pain points in the process for bringing new therapies to market. We expect the companies providing site-centric trial solutions to see substantial demand and success, ultimately making a significant contribution to improving the quality and availability of healthcare.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
