Quarterly insights: Process technology
Quants II: Drug discovery “rocket scientists” lifting off
Advanced data analysis, modeling, and model systems – quantitative domains that gained the moniker “rocket science” based on their successful and transformative application in aerospace – have also been successfully applied in several unrelated areas such as finance.
But while these rocket science tools and capabilities have long held great promise in the field of drug discovery, they have generally delivered marginal benefits. We contend that’s because the complexity of the challenges posed by drug discovery and development (the complexity of biology) is orders of magnitude greater than in the areas where “rocket science” has historically seen success.
Today, we see advancements in science and technology we believe will enable the quant approach of using large amounts of quality data and analytics to more fully realize its potential in drug discovery and development. These advancements include large increases in computing power and scale and new tools for engineering and interrogating model biological systems.
We profile a handful of companies successfully applying today’s rocket science to drug discovery and development with business models that extend beyond traditional fee-for-service to taking equity stakes in the therapeutic products they enable with their partners or undertaking proprietary therapeutic product development.
TABLE OF CONTENTS
Includes discussion of BTAI, CDXS, SDGR, SLP, Viva Biotech (1876.HK) and one private company
- Drug discovery much tougher than rocket science, new tools just beginning to tackle the challenge
- Traditional and newer business models capture the value of innovation
- Some pioneers using drug discovery “rocket science”
- Rocket science in drug discovery and development: Transforming our world again
- Process technology index appreciation continues
- M&A activity remains subdued
- Q4 private placements accelerate
Introduction
The extraordinary returns recently seen among a number of our process technology universe public companies suggest something is afoot. A commonality among these companies is they are leveraging experimentation and analytics expertise (their “rocket science”) to participate in the drug discovery and development market with models including:
- Selling their services or access to their platforms on a fee basis
- Taking equity stakes in the therapeutic products their capabilities enable
- In some cases, using their capabilities to develop their own drug pipeline
We believe these companies’ strong stock price performances and associated eye-popping valuations reflect the long-term potential of this drug discovery “rocket science” – potential not captured in simple short-run valuation metrics.
Table 1 shows how the market has ascribed billion-dollar-plus values to discovery technology companies with near-term revenue prospects measured in the tens or hundreds of millions and typically no expectation of near-term profit.
TABLE 1: Drug discovery “rocket scientist” companies financial profile
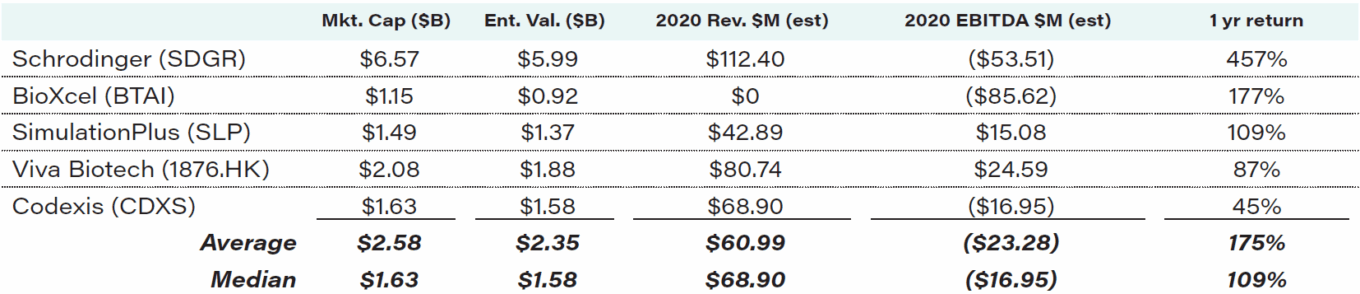
Source: Capital IQ, as of Jan. 21, 2021.
We present a simple framework on how we look at these types of companies and briefly profile some of the participants. We tend to view the field as fruitful for investment and M&A as participants fill gaps and add to white spaces.
Drug discovery much tougher than rocket science, new tools just beginning to tackle the challenge
While “rocket science” traditionally describes complex engineering challenges, such as those encountered in aeronautical engineering, traditional rocket science is simple by today’s standards. The basic equations of motion, including relativistic effects, have long been understood, even in the 1950s and 1960s.
The challenges rocket science addresses are mechanical and deterministic and to a large degree simple and independent. Solutions to a traditional rocket science problem are relatively easy to engineer, dealing with well-defined variables like weight, thrust and velocity. With the advancement of technology, the concept of rocket science has more recently been applied to an arguably more challenging arena: financial systems.
Here, technologies for storing and analyzing large quantities of financial data and building models to describe market or arbitrage dislocations have enabled a proliferation of quantitative financial products. While financial systems’ human element makes them reactive, introducing an extra element of complexity, they exist within a relatively well-defined ecosystem of networks and rules that put boundaries around their complexity, and they have prodigious amounts of high-quality, near-real-time data.
In contrast, the challenges encountered in drug discovery throw some pretty big wrenches into attempts to use traditional quantitative approaches to model biological systems. In general, biological data is very noisy, meaning you need larger amounts of quality data to capture meaningful information.
Biological systems are extraordinarily complex systems of chemical reactions and components that react, mutate and have high levels of diversity based on genetics and environment. The large, noisy data sets biological systems typically generate require much greater compute infrastructure and more powerful algorithms to make sense of the data. Once you understand the data, engineering biological systems and drugs to drive desired outcomes requires similarly more powerful tools and technology.
These capabilities, which are required to achieve an order of magnitude improvement in drug discovery and development efficiency, are just now coming of age. They include enormous computing power, the ability to accomplish high-throughput experimental biology reproducibly, the ability to engineer biology with tools such as gene editing, and the ability to engineer drug candidates via computational chemistry.
Traditional and newer business models capture the value of innovation
In simplistic terms, the traditional life science tools business model saw life science tools companies sell their products and services to drug companies for cash, and the drug companies developed therapeutics as a captive proprietary pipeline. This model has several limitations: The life science tools company cannot capture the upside of its technology, and the arms-length nature of the vendor-client relationship creates inefficiencies in the discovery and development process that hurt pharma clients by limiting productivity.
Over time, a much more complex but efficient ecosystem has emerged wherein life science tools and technology developers pursue hybrid business models that include participating in the economic reward of the therapeutics they enable. Pharma and biotech companies are willing to share some of the economics of their therapeutics with a technology provider under this more efficient collaborative approach if it benefits the pharma companies and their programs.
Alternatively, life science tools and technology developers can apply their solutions to proprietary drug development efforts. This enables the tools and technology companies to capture all the economic rewards of success but can entail substantial investment and risk. Thus, the role of technology suppliers can be selling technology products for cash (the traditional life science tools model), partnering with pharma in exchange for royalties or equity (collaboration), or using profits to develop their own drug pipelines (the traditional pharma model). Many of the successful players pursue a complex hybrid strategy combining all these roles and compensation forms.
It’s important to note that for technology providers to pursue the partnership elements of this hybrid strategy, they must provide significant value to their drug company partners that pharma can’t access elsewhere or develop internally. Pharma companies are loath to share the economic upside of their therapeutics and provide cash to develop technology they don’t own unless they have no alternative.
We profile a handful of pioneers using their quantitative capabilities and both traditional and new value capture models to bring new, more effective drugs to market much faster than ever before.
Some pioneers using drug discovery “rocket science”
Atomwise (private): Atomwise uses the tag-line of “drug the undruggable.” The basic premise is to use analytical or artificial intelligence to expand the number of genes or proteins that can be targeted via a drug. Today, only a small fraction (less than 1,000) of the genes and proteins in human biology are drug targets. Atomwise uses high-speed and large-scale compute techniques to rapidly scan drug libraries as well as provide tools to medicinal chemists to improve their productivity. It pursues a hybrid strategy of fee-for-service, taking equity in joint ventures with partners, and biotech and pharma deals. Atomwise counts Lilly, Merck, abbvie and Bayer among its pharmaceutical partners.
BioXcel Therapeutics (BTAI): BioXcel uses an artificial intelligence platform to more effectively screen for drug candidates among known compounds that can address disease states other than their primary targets, a process it terms re-innovation. Its current focus areas are neurology and immuno-oncology. Its artificial intelligence platform essentially seeks to aggregate all known data on a universe of potential drugs and mine it for new targets. It has two candidates in Phase 2 clinical studies.
Simulations Plus (SLP): Simulations Plus offers an array of software and services across drug development stages from discovery through clinical. Its toolbox contains a number of model and simulation algorithms that provide in silico analysis to speed up processes and decision making. It has assisted bringing over 40 drugs to market and has participated in numerous development programs.
Viva Biotech (Hong Kong – 1873): Viva was founded in 2008 and has leveraged its beginnings in protein crystallography and synchrotron access to become a player in the field of structure-based drug discovery. Its capabilities now run from early drug discovery through structure and screening to candidate determination. It also pursues a hybrid fee-and-equity model it refers to as cash-for-service (CFS) and equity-for-service (EFS) while at the same time offering its own incubator to assist startup companies in the field. It has over 495 customers, has worked on 1,300 drug candidates, has 17,000 protein structures, and has incubated 56 early stage opportunities.
Codexis (CDXS): We tend to think of Codexis as the protein (enzyme) engineering powerhouse. Its leadership is built on the pillars of high-throughput screening and experimentation and high-performance analytics. It has done an excellent job migrating from fee-for-service to a balanced hybrid model selling access to its platform and pursuing product and drug candidates in partnership and on its own account. In contrast to many in the area, it also has commercial-scale manufacturing and process optimization capabilities.
Schrödinger (SDGR): Schrödinger specializes in the physics-based approach to new molecule discovery and screening. It works across chemicals, materials and drugs (a special subcategory of chemicals). Its claim to fame (no it isn’t the wave equation, that’s a different guy) is high-speed evaluation of new compounds. For example, classic synthesis might produce and screen 1,000 compounds per year, while Schrödinger might achieve a million times more than that in a day. It is pursuing a number of cancer indications on its own as well as in partnership with Bristol Myers Squibb, and it works with several other pharma partners in other diseases.
Rocket science in drug discovery and development: Transforming our world again
The rocket science of aerospace transformed our world in ways that were unimaginable at its advent, from enabling global personal mobility to enabling satellite communication, imagery, and geolocation. We expect the rocket science of drug discovery will likewise transform our world in ways we can only begin to imagine today.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
